Ozonolysis on:
[Wikipedia]
[Google]
[Amazon]
In
 An example is the ozonolysis of eugenol converting the terminal alkene to an aldehyde:
:
An example is the ozonolysis of eugenol converting the terminal alkene to an aldehyde:
: By controlling the reaction/workup conditions, unsymmetrical products can be generated from symmetrical alkenes:
* Using TsOH; sodium bicarbonate (NaHCO3); dimethyl sulfide (DMS) gives an aldehyde and a dimethyl
By controlling the reaction/workup conditions, unsymmetrical products can be generated from symmetrical alkenes:
* Using TsOH; sodium bicarbonate (NaHCO3); dimethyl sulfide (DMS) gives an aldehyde and a dimethyl
 In the generally accepted mechanism proposed by Rudolf Criegee in 1953, the alkene and ozone form an intermediate
In the generally accepted mechanism proposed by Rudolf Criegee in 1953, the alkene and ozone form an intermediate  Evidence for this mechanism is found in
Evidence for this mechanism is found in

\underset + 4O3 -> \underset + \underset
 The method was used to confirm the structural
The method was used to confirm the structural
organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the scientific study of the structure, properties, and reactions of organic compounds and organic materials, i.e., matter in its various forms that contain carbon atoms.Clayden, ...
, ozonolysis is an organic reaction where the unsaturated bonds of alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s (), alkynes (), or azo compound
Azo compounds are organic compounds bearing the functional group diazenyl (, in which R and R′ can be either aryl or alkyl groups).
IUPAC defines azo compounds as: "Derivatives of diazene (diimide), , wherein both hydrogens are substituted ...
s () are cleaved with ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the lo ...
(). Alkenes and alkynes form organic compound
In chemistry, organic compounds are generally any chemical compounds that contain carbon-hydrogen or carbon-carbon bonds. Due to carbon's ability to catenate (form chains with other carbon atoms), millions of organic compounds are known. T ...
s in which the multiple carbon–carbon bond has been replaced by a carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containi ...
() group while azo compounds form nitrosamines (). The outcome of the reaction depends on the type of multiple bond being oxidized and the work-up conditions.
Ozonolysis of alkenes
Alkenes can be oxidized withozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the lo ...
to form alcohols, aldehyde
In organic chemistry, an aldehyde () is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl grou ...
s or ketones, or carboxylic acids. In a typical procedure, ozone is bubbled through a solution of the alkene in methanol at −78 °C until the solution takes on a characteristic blue color, which is due to unreacted ozone. This indicates complete consumption of the alkene. Alternatively, various other chemicals can be used as indicators of this endpoint by detecting the presence of ozone. If ozonolysis is performed by bubbling a stream of ozone-enriched oxygen through the reaction mixture, the gas that bubbles out can be directed through a potassium iodide
Potassium iodide is a chemical compound, medication, and dietary supplement. It is a medication used for treating hyperthyroidism, in radiation emergencies, and for protecting the thyroid gland when certain types of radiopharmaceuticals are u ...
solution. When the solution has stopped absorbing ozone, the ozone in the bubbles oxidizes the iodide to iodine, which can easily be observed by its violet color. For closer control of the reaction itself, an indicator such as Sudan Red III
Sudan III is a lysochrome (fat-soluble dye) diazo dye. It is structurally related to azobenzene.
Uses
It is used to color nonpolar substances such as oils, fats, waxes, greases, various hydrocarbon products, and acrylic emulsions. Its main ...
can be added to the reaction mixture. Ozone reacts with this indicator more slowly than with the intended ozonolysis target. The ozonolysis of the indicator, which causes a noticeable color change, only occurs once the desired target has been consumed. If the substrate has two alkenes that react with ozone at different rates, one can choose an indicator whose own oxidation rate is intermediate between them, and therefore stop the reaction when only the most susceptible alkene in the substrate has reacted. Otherwise, the presence of unreacted ozone in solution (seeing its blue color) or in the bubbles (via iodide detection) only indicates when all alkenes have reacted.
After completing the addition, a reagent is then added to convert the intermediate ozonide to a carbonyl derivative. Reductive work-up conditions are far more commonly used than oxidative conditions. The use of triphenylphosphine, thiourea
Thiourea () is an organosulfur compound with the formula and the structure . It is structurally similar to urea (), except that the oxygen atom is replaced by a sulfur atom (as implied by the ''thio-'' prefix); however, the properties of urea a ...
, zinc
Zinc is a chemical element with the symbol Zn and atomic number 30. Zinc is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodi ...
dust, or dimethyl sulfide produces aldehydes or ketones while the use of sodium borohydride
Sodium borohydride, also known as sodium tetrahydridoborate and sodium tetrahydroborate, is an inorganic compound with the formula Na BH4. This white solid, usually encountered as an aqueous basic solution, is a reducing agent that finds applica ...
produces alcohols. The use of hydrogen peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%� ...
produces carboxylic acids. Recently, the use of amine ''N''-oxides has been reported to produce aldehydes directly. Other functional group
In organic chemistry, a functional group is a substituent or moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions regardless of the re ...
s, such as benzyl
In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure . Benzyl features a benzene ring () attached to a methylene group () group.
Nomenclature
In IUPAC nomenclature, the prefix benzyl refers to a substi ...
ether
In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula , where R and R′ represent the alkyl or aryl groups. Ethers can again be ...
s, can also be oxidized by ozone. It has been proposed that small amounts of acid may be generated during the reaction from oxidation of the solvent, so pyridine
Pyridine is a basic heterocyclic organic compound with the chemical formula . It is structurally related to benzene, with one methine group replaced by a nitrogen atom. It is a highly flammable, weakly alkaline, water-miscible liquid with a ...
is sometimes used to buffer
Buffer may refer to:
Science
* Buffer gas, an inert or nonflammable gas
* Buffer solution, a solution used to prevent changes in pH
* Buffering agent, the weak acid or base in a buffer solution
* Lysis buffer, in cell biology
* Metal ion buffer
* ...
the reaction. Dichloromethane is often used as a 1:1 cosolvent to facilitate timely cleavage of the ozonide. Azelaic acid and pelargonic acid
Pelargonic acid, also called nonanoic acid, is an organic compound with structural formula CH3(CH2)7CO2H. It is a nine-carbon fatty acid. Nonanoic acid is a colorless oily liquid with an unpleasant, rancid odor. It is nearly insoluble in water, ...
s are produced from ozonolysis of oleic acid
Oleic acid is a fatty acid that occurs naturally in various animal and vegetable fats and oils. It is an odorless, colorless oil, although commercial samples may be yellowish. In chemical terms, oleic acid is classified as a monounsaturated omeg ...
on an industrial scale.
: An example is the ozonolysis of eugenol converting the terminal alkene to an aldehyde:
:
An example is the ozonolysis of eugenol converting the terminal alkene to an aldehyde:
: By controlling the reaction/workup conditions, unsymmetrical products can be generated from symmetrical alkenes:
* Using TsOH; sodium bicarbonate (NaHCO3); dimethyl sulfide (DMS) gives an aldehyde and a dimethyl
By controlling the reaction/workup conditions, unsymmetrical products can be generated from symmetrical alkenes:
* Using TsOH; sodium bicarbonate (NaHCO3); dimethyl sulfide (DMS) gives an aldehyde and a dimethyl acetal
In organic chemistry, an acetal is a functional group with the connectivity . Here, the R groups can be organic fragments (a carbon atom, with arbitrary other atoms attached to that) or hydrogen, while the R' groups must be organic fragments n ...
* Using acetic anhydride (Ac2O), triethylamine (Et3N) gives a methyl ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides a ...
and an aldehyde
* Using TsOH; Ac2O, Et3N, gives a methyl ester and a dimethyl acetal.
Reaction mechanism
molozonide
A molozonide (or "molecular ozonide") is a 1,2,3-trioxolane, which can also be thought of a cyclic dialkyl trioxidane. Molozonides are formed by cycloaddition of ozone and an alkene during ozonolysis, as a transient intermediate which quickly rearr ...
in a 1,3-dipolar cycloaddition. Next, the molozonide reverts to its corresponding carbonyl oxide (also called the Criegee intermediate
A Criegee intermediate (also called a Criegee zwitterion or Criegee biradical) is a carbonyl oxide with two charge centres. These chemicals may react with sulfur dioxide and nitrogen oxides in the earth's atmosphere, and are implicated in the f ...
or Criegee zwitterion
In chemistry, a zwitterion ( ; ), also called an inner salt or dipolar ion, is a molecule that contains an equal number of positively- and negatively-charged functional groups.
: With amino acids, for example, in solution a chemical equilibrium wil ...
) and aldehyde or ketone in a retro-1,3-dipolar cycloaddition. The oxide and aldehyde or ketone react again in a 1,3-dipolar cycloaddition or produce a relatively stable ozonide
Ozonide is the polyatomic anion . Cyclic organic compounds formed by the addition of ozone () to an alkene are also called ozonides.
Ionic ozonides
Inorganic ozonides are dark red salts. The anion has the bent shape of the ozone molecule.
Ino ...
intermediate (a trioxolane).
: Evidence for this mechanism is found in
Evidence for this mechanism is found in isotopic labeling
Isotopic labeling (or isotopic labelling) is a technique used to track the passage of an isotope (an atom with a detectable variation in neutron count) through a reaction, metabolic pathway, or cell. The reactant is 'labeled' by replacing specific ...
. When 17O-labelled benzaldehyde reacts with carbonyl oxides, the label ends up exclusively in the ether linkage of the ozonide. There is still dispute over whether the molozonide collapses via a concerted or radical process; this may also exhibit a substrate dependence.
History
Christian Friedrich Schönbein, who discovered ozone in 1840, also did the first ozonolysis: in 1845, he reported that ethylene reacts with ozone – after the reaction, neither the smell of ozone nor the smell of ethylene was perceivable. The ozonolysis of alkenes is sometimes referred to as "Harries ozonolysis", because some attribute this reaction toCarl Dietrich Harries
Carl Dietrich Harries (5 August 1866 – 3 November 1923) was a German chemist born in Luckenwalde, Brandenburg, Prussia. He received his doctorate in 1892. In 1900, he married Hertha von Siemens, daughter of the electrical genius Werner von Sie ...
.
Before the advent of modern spectroscopic techniques, the ozonolysis was an important method for determining the structure of organic molecules. Chemists would ozonize an unknown alkene to yield smaller and more readily identifiable fragments.
Ozonolysis of alkynes
Ozonolysis of alkynes generally gives anacid anhydride An acid anhydride is a type of chemical compound derived by the removal of water molecules from an acid.
In organic chemistry, organic acid anhydrides contain the functional group R(CO)O(CO)R'. Organic acid anhydrides often form when one equivale ...
or diketone
In organic chemistry, a dicarbonyl is a molecule containing two carbonyl () groups. Although this term could refer to any organic compound containing two carbonyl groups, it is used more specifically to describe molecules in which both carbonyls ...
product, not complete fragmentation as for alkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic, an ...
s. A reducing agent is not needed for these reactions. The exact mechanism is not completely known. If the reaction is performed in the presence of water, the anhydride hydrolyzes to give two carboxylic acids.
:
Ozonolysis of other substrates
Ozonolysis ofoleic acid
Oleic acid is a fatty acid that occurs naturally in various animal and vegetable fats and oils. It is an odorless, colorless oil, although commercial samples may be yellowish. In chemical terms, oleic acid is classified as a monounsaturated omeg ...
is an important route to azelaic acid. The coproduct is nonanoic acid:
:Ozonolysis of elastomers
 The method was used to confirm the structural
The method was used to confirm the structural repeat unit
In polymer chemistry, a repeat unit or repeating unit (or mer) is a part of a polymer whose repetition would produce the complete polymer chain (except for the end-groups) by linking the repeat units together successively along the chain, like the ...
in natural rubber
Rubber, also called India rubber, latex, Amazonian rubber, ''caucho'', or ''caoutchouc'', as initially produced, consists of polymers of the organic compound isoprene, with minor impurities of other organic compounds. Thailand, Malaysia, and ...
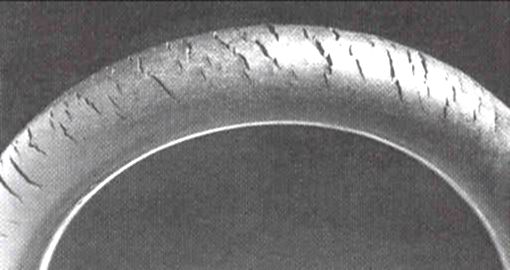
as isoprene. It is also a serious problem, known as ozone cracking
Cracks can be formed in many different elastomers by ozone attack, and the characteristic form of attack of vulnerable rubbers is known as ozone cracking. The problem was formerly very common, especially in tires, but is now rarely seen in those ...
where traces of the gas in an atmosphere cause degradation in elastomers
An elastomer is a polymer with viscoelasticity (i.e. both viscosity and elasticity) and with weak intermolecular forces, generally low Young's modulus and high failure strain compared with other materials. The term, a portmanteau of ''elastic p ...
, such as natural rubber
Rubber, also called India rubber, latex, Amazonian rubber, ''caucho'', or ''caoutchouc'', as initially produced, consists of polymers of the organic compound isoprene, with minor impurities of other organic compounds. Thailand, Malaysia, and ...
, polybutadiene, styrene-butadiene
Styrene-butadiene or styrene-butadiene rubber (SBR) describe families of synthetic rubbers derived from styrene and butadiene (the version developed by Goodyear is called Neolite). These materials have good abrasion resistance and good aging st ...
and nitrile rubber. Ozonolysis creates surface ketone groups which can cause further gradual degradation via Norrish reactions if the polymer is exposed to light.
Ozone cracking is a form of stress corrosion cracking where active chemical species attack products of a susceptible material. The rubber product must be under tension
Tension may refer to:
Science
* Psychological stress
* Tension (physics), a force related to the stretching of an object (the opposite of compression)
* Tension (geology), a stress which stretches rocks in two opposite directions
* Voltage or el ...
for crack growth to occur. Ozone cracking was once commonly seen in the sidewalls of tire
A tire (American English) or tyre (British English) is a ring-shaped component that surrounds a wheel's rim to transfer a vehicle's load from the axle through the wheel to the ground and to provide traction on the surface over which t ...
s, where it could expand to cause a dangerous blowout, but is now rare owing to the use of modern antiozonant
An antiozonant, also known as anti-ozonant, is an organic compound that prevents or retards damage caused by ozone. The most important antiozonants are those which prevent degradation of elastomers like rubber. A number of research projects study ...
s. Other means of prevention include replacing susceptible rubbers with resistant elastomers such as polychloroprene, EPDM or Viton.
See also
*Polymer degradation
Polymer degradation is the reduction in the physical properties of a polymer, such as strength, caused by changes in its chemical composition. Polymers and particularly plastics are subject to degradation at all stages of their product life cycl ...
*Lemieux–Johnson oxidation The Lemieux–Johnson or Malaprade–Lemieux–Johnson oxidation is a chemical reaction in which an olefin undergoes oxidative cleavage to form two aldehyde or ketone units. The reaction is named after its inventors, Raymond Urgel Lemieux and Will ...
– an alternative system using periodate and osmium tetroxide
*''Trametes hirsuta
''Trametes hirsuta'', commonly known as hairy bracket, is a fungal plant pathogen. It is found on dead wood of deciduous trees, especially beechwood. It is found all year round and persists due to its leathery nature.Phillips, Roger (2006), Mushr ...
'', a biotechnological
Biotechnology is the integration of natural sciences and engineering sciences in order to achieve the application of organisms, cells, parts thereof and molecular analogues for products and services. The term ''biotechnology'' was first used ...
alternative to ozonolysis.
References
{{Organic reactions Organic oxidation reactions Cycloadditions