Oxocarbenium on:
[Wikipedia]
[Google]
[Amazon]
 An oxocarbenium ion (or oxacarbenium ion) is a chemical species characterized by a central sp2-hybridized carbon, an oxygen substituent, and an overall positive charge that is delocalized between the central carbon and oxygen atoms. An oxocarbenium ion is represented by two limiting resonance structures, one in the form of a
An oxocarbenium ion (or oxacarbenium ion) is a chemical species characterized by a central sp2-hybridized carbon, an oxygen substituent, and an overall positive charge that is delocalized between the central carbon and oxygen atoms. An oxocarbenium ion is represented by two limiting resonance structures, one in the form of a
 This is also the order of electrophilicity for species containing C=X (X = O, NR) bonds. This order is synthetically significant and explains, for example, why
This is also the order of electrophilicity for species containing C=X (X = O, NR) bonds. This order is synthetically significant and explains, for example, why

 The
The 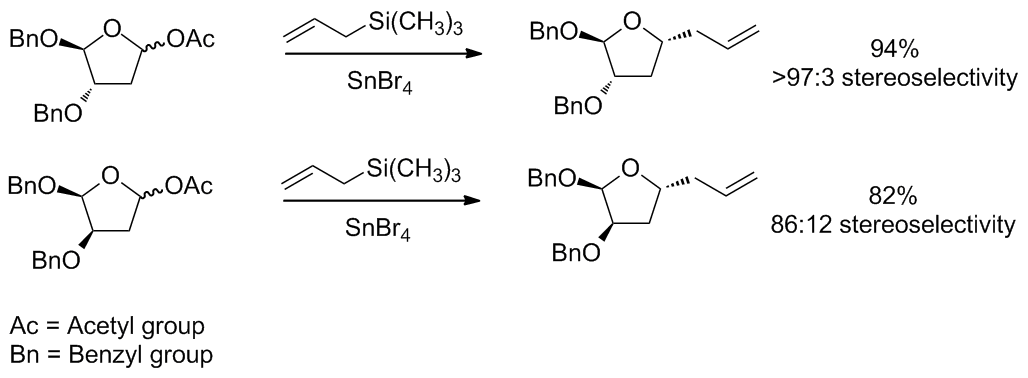
 The transition state model for a six-membered oxocarbenium ring was proposed earlier in 1992 by Woods et al. The general strategy for determining the stereochemistry of a nucleophilic addition to a six-membered ring follows a similar procedure to the case of the five-membered ring. The assumption that one makes for this analysis is that the ring is in the same conformation as
The transition state model for a six-membered oxocarbenium ring was proposed earlier in 1992 by Woods et al. The general strategy for determining the stereochemistry of a nucleophilic addition to a six-membered ring follows a similar procedure to the case of the five-membered ring. The assumption that one makes for this analysis is that the ring is in the same conformation as 


 A second example is seen in the key step of the synthesis of (−)-neopeltolide, which uses another six-membered oxocarbenium ring reduction for a diastereoselective hydride addition.
A second example is seen in the key step of the synthesis of (−)-neopeltolide, which uses another six-membered oxocarbenium ring reduction for a diastereoselective hydride addition.

 An oxocarbenium ion (or oxacarbenium ion) is a chemical species characterized by a central sp2-hybridized carbon, an oxygen substituent, and an overall positive charge that is delocalized between the central carbon and oxygen atoms. An oxocarbenium ion is represented by two limiting resonance structures, one in the form of a
An oxocarbenium ion (or oxacarbenium ion) is a chemical species characterized by a central sp2-hybridized carbon, an oxygen substituent, and an overall positive charge that is delocalized between the central carbon and oxygen atoms. An oxocarbenium ion is represented by two limiting resonance structures, one in the form of a carbenium ion
A carbenium ion is a positive ion with the structure RR′R″C+, that is, a chemical species with a trivalent carbon that bears a +1 formal charge.
In older literature the name carbonium ion was used for this class, but now it refers exclusivel ...
with the positive charge on carbon and the other in the form of an oxonium species with the formal charge on oxygen. As a resonance hybrid, the true structure falls between the two. Compared to neutral carbonyl compounds like ketones or esters, the carbenium ion form is a larger contributor to the structure. They are common reactive intermediates in the hydrolysis of glycosidic bond
A glycosidic bond or glycosidic linkage is a type of covalent bond that joins a carbohydrate (sugar) molecule to another group, which may or may not be another carbohydrate.
A glycosidic bond is formed between the hemiacetal or hemiketal group ...
s, and are a commonly used strategy for chemical glycosylation
A chemical glycosylation reaction involves the coupling of a glycosyl donor, to a glycosyl acceptor forming a glycoside. If both the donor and acceptor are sugars, then the product is an oligosaccharide. The reaction requires activation with a ...
. These ions have since been proposed as reactive intermediates in a wide range of chemical transformations, and have been utilized in the total synthesis of several natural products. In addition, they commonly appear in mechanisms of enzyme-catalyzed biosynthesis and hydrolysis of carbohydrates in nature. Anthocyanin
Anthocyanins (), also called anthocyans, are water-soluble vacuolar pigments that, depending on their pH, may appear red, purple, blue, or black. In 1835, the German pharmacist Ludwig Clamor Marquart gave the name Anthokyan to a chemical com ...
s are natural flavylium
Pyrylium is a cation (positive ion) with formula , consisting of a six-membered ring of five carbon atoms, each with one hydrogen atom, and one positively charged oxygen atom. The bonds in the ring are conjugated as in benzene, giving it an arom ...
dyes, which are stabilized oxocarbenium compounds. Anthocyanins are responsible for the colors of a wide variety of common flowers such as pansies
The garden pansy (''Viola'' × ''wittrockiana'') is a type of large-flowered hybrid plant cultivated as a garden flower. It is derived by hybridization from several species in the section ''Melanium'' ("the pansies") of the genus ''Viola'', p ...
and edible plants such as eggplant
Eggplant ( US, Canada), aubergine ( UK, Ireland) or brinjal (Indian subcontinent, Singapore, Malaysia, South Africa) is a plant species in the nightshade family Solanaceae. ''Solanum melongena'' is grown worldwide for its edible fruit.
Mo ...
and blueberry
Blueberries are a widely distributed and widespread group of perennial flowering plants with blue or purple berries. They are classified in the section ''Cyanococcus'' within the genus '' Vaccinium''. ''Vaccinium'' also includes cranberries ...
.
Electron distribution and reactivity
The best Lewis structure for an oxocarbenium ion contains an oxygen–carbondouble bond
In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist betwee ...
, with the oxygen atom attached to an additional group and consequently taking on a formal positive charge. In the language of canonical structures (or "resonance"), the polarization of the π bond is described by a secondary carbocationic resonance form, with a formal positive charge on carbon (see above). In terms of frontier molecular orbital theory In chemistry, frontier molecular orbital theory is an application of MO theory describing HOMO/LUMO interactions.
History
In 1952, Kenichi Fukui published a paper in the ''Journal of Chemical Physics'' titled "A molecular theory of reactivity i ...
, the Lowest Unoccupied Molecular Orbital
In chemistry, HOMO and LUMO are types of molecular orbitals. The acronyms stand for ''highest occupied molecular orbital'' and ''lowest unoccupied molecular orbital'', respectively. HOMO and LUMO are sometimes collectively called the ''frontie ...
(LUMO) of the oxocarbenium ion is a π* orbital that has the large lobe on the carbon atom; the more electronegative oxygen contributes less to the LUMO. Consequently, in an event of a nucleophilic attack, the carbon is the electrophilic site. Compared to a ketone
In organic chemistry, a ketone is a functional group with the structure R–C(=O)–R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group –C(=O)– (which contains a carbon-oxygen double b ...
, the polarization of an oxocarbenium ion is accentuated: they more strongly resemble a "true" carbocation, and they are more reactive toward nucleophiles. In organic reactions, ketones are commonly activated by the coordination of a Lewis acid
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any sp ...
or Brønsted acid to the oxygen to generate an oxocarbenium ion as an intermediate.
Numerically, a typical partial charge (derived from Hartree-Fock computations) for the carbonyl carbon of a ketone R2C=O (like acetone) is ''δ+'' = 0.51. With the addition of an acidic hydrogen to the oxygen atom to produce 2C=OHsup>+, the partial charge increases to ''δ+'' = 0.61. In comparison, the nitrogen analogues of ketones and oxocarbenium ions, imines (R2C=NR) and iminium ions ( 2C=NRHsup>+), respectively, have partial charges of ''δ+'' = 0.33 and ''δ+'' = 0.54, respectively. The order of partial positive charge on the carbonyl carbon is therefore imine < ketone < iminium < oxocarbenium.
 This is also the order of electrophilicity for species containing C=X (X = O, NR) bonds. This order is synthetically significant and explains, for example, why
This is also the order of electrophilicity for species containing C=X (X = O, NR) bonds. This order is synthetically significant and explains, for example, why reductive amination
Reductive amination (also known as reductive alkylation) is a form of amination that involves the conversion of a carbonyl group to an amine via an intermediate imine. The carbonyl group is most commonly a ketone or an aldehyde. It is considered ...
s are often best carried out at pH = 5 to 6 using sodium cyanoborohydride
Sodium cyanoborohydride is the chemical compound with the formula Na B H3 CN. It is a colourless salt, but commercial samples can appear tan. It is widely used in organic synthesis for the reduction of imines. The salt tolerates aqueous conditions ...
(Na+ 3B(CN)sup>−) or sodium triacetoxyborohydride (Na+ B(OAc)3sup>−) as a reagent. Bearing an electron-withdrawing group, sodium cyanoborohydride and sodium triacetoxyborohydride are poorer reducing agents than sodium borohydride, and their direct reaction with ketones is generally a slow and inefficient process. However, the iminium ion (but not the imine itself) formed ''in situ'' during a reductive amination reaction is a stronger electrophile than the ketone starting material and will react with the hydride source at a synthetically useful rate. Importantly, the reaction is conducted under mildly acidic conditions that protonate the imine intermediate to a significant extent, forming the iminium ion, while not being strongly acidic enough to protonate the ketone, which would form the even more electrophilic oxocarbenium ion. Thus, the reaction conditions and reagent ensure that amine is formed selectively from iminium reduction, instead of direct reduction of the carbonyl group (or its protonated form) to form an alcohol.
Formation
Formation of oxocarbenium ions can proceed through several different pathways. Most commonly, the oxygen of a ketone will bind to aLewis Acid
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any sp ...
, which activates the ketone, making it a more effective electrophile. The Lewis acid can be a wide range of molecules, from a simple hydrogen atom to metal complexes. The remainder of this article will focus on alkyl oxocarbenium ions, however, where the atom added to the oxygen is a carbon. One way that this sort of ion will form is the elimination of a leaving group In chemistry, a leaving group is defined by the IUPAC as an atom or group of atoms that detaches from the main or residual part of a substrate during a reaction or elementary step of a reaction. However, in common usage, the term is often limited ...
. In carbohydrate chemistry, this leaving group is often an ether
In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula , where R and R′ represent the alkyl or aryl groups. Ethers can again ...
or ester
In chemistry, an ester is a compound derived from an oxoacid (organic or inorganic) in which at least one hydroxyl group () is replaced by an alkoxy group (), as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides ...
. An alternative to elimination is direct deprotonation of the molecule to form the ion, however, this can be difficult and require strong bases to achieve.

Applications to synthesis
5-membered rings
 The
The stereochemistry
Stereochemistry, a subdiscipline of chemistry, involves the study of the relative spatial arrangement of atoms that form the structure of molecules and their manipulation. The study of stereochemistry focuses on the relationships between stereoi ...
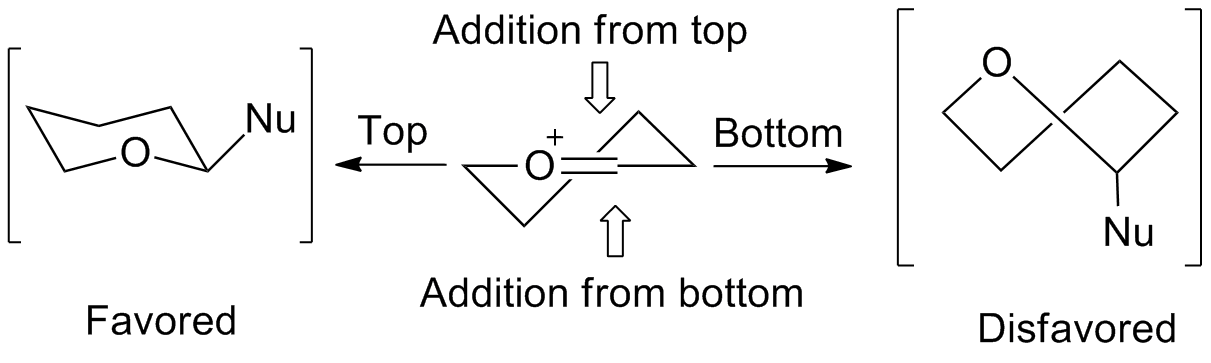
involved in the reactions of five-membered rings can be predicted by an envelope transition state model. Nucleophiles favor addition from the "inside" of the envelope, or from the top of the figure on the right. The "inside" addition produces a results in a staggered conformation
In organic chemistry, a staggered conformation is a chemical conformation of an ethane-like moiety abcX–Ydef in which the substituents a, b, and c are at the maximum distance from d, e, and f; this requires the torsion angles to be 60°. It ...
, rather than the eclipsed conformation
In chemistry an eclipsed conformation is a conformation in which two substituents X and Y on adjacent atoms A, B are in closest proximity, implying that the torsion angle X–A–B–Y is 0°. Such a conformation can exist in any open chain ...
that results from the "outside" addition.

6-membered rings
 The transition state model for a six-membered oxocarbenium ring was proposed earlier in 1992 by Woods et al. The general strategy for determining the stereochemistry of a nucleophilic addition to a six-membered ring follows a similar procedure to the case of the five-membered ring. The assumption that one makes for this analysis is that the ring is in the same conformation as
The transition state model for a six-membered oxocarbenium ring was proposed earlier in 1992 by Woods et al. The general strategy for determining the stereochemistry of a nucleophilic addition to a six-membered ring follows a similar procedure to the case of the five-membered ring. The assumption that one makes for this analysis is that the ring is in the same conformation as cyclohexene
Cyclohexene is a hydrocarbon with the formula C6H10. This cycloalkene is a colorless liquid with a sharp smell. It is an intermediate in various industrial processes. Cyclohexene is not very stable upon long term storage with exposure to light ...
, with three carbons and the oxygen in a plane with the two other carbon atome puckered out of the plane, with one above and one below (see the figure to the right). Based on the substituients present on the ring, the lowest energy conformation is determined, keeping in mind steric and stereoelectronic effects (see the section below for a discussion of stereoelectronic effects in oxocarbenium rings). Once this conformation is established, one can consider the nucleophilic addition. The addition will proceed through the low energy chair transition state, rather than the relatively high energy twist-boat. An example of this type of reaction can be seen below. The example also highlights how the stereoelectronic effect exerted by an electronegative substituent flips the lowest energy conformation and leads to opposite selectivity.

Stereoelectronic effects
In analkene
In organic chemistry, an alkene is a hydrocarbon containing a carbon–carbon double bond.
Alkene is often used as synonym of olefin, that is, any hydrocarbon containing one or more double bonds.H. Stephen Stoker (2015): General, Organic ...
ring that does not contain an oxygen atom, any large substituent prefers to be in an equatorial position, in order to minimize steric effects
Steric effects arise from the spatial arrangement of atoms. When atoms come close together there is a rise in the energy of the molecule. Steric effects are nonbonding interactions that influence the shape ( conformation) and reactivity of ions ...
. It has been observed in rings containing oxocarbenium ions that electronegative
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the ...
substituents prefer the axial
Axial may refer to:
* one of the anatomical directions describing relationships in an animal body
* In geometry:
:* a geometric term of location
:* an axis of rotation
* In chemistry, referring to an axial bond
* a type of modal frame, in music
* ...
or pseudo-axial positions. When the electronegative atom is in the axial position, its electron density can be donated through space to the positively charged oxygen atom in the ring. This electronic interaction stabilizes the axial conformation. Hydroxyl
In chemistry, a hydroxy or hydroxyl group is a functional group with the chemical formula and composed of one oxygen atom covalently bonded to one hydrogen atom. In organic chemistry, alcohols and carboxylic acids contain one or more hydrox ...
groups, ethers
In organic chemistry, ethers are a class of compounds that contain an ether group—an oxygen atom connected to two alkyl or aryl groups. They have the general formula , where R and R′ represent the alkyl or aryl groups. Ethers can again be ...
and halogens
The halogens () are a group in the periodic table consisting of five or six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). In the modern IUPAC nomenclature, this group i ...
are examples of substituents that exhibit this phenomenon. Stereoelectronic effects must be taken into consideration when determining the lowest energy conformation in the analysis for nucleophilic addition
In organic chemistry, a nucleophilic addition reaction is an addition reaction where a chemical compound with an electrophilic double or triple bond reacts with a nucleophile, such that the double or triple bond is broken. Nucleophilic additions d ...
to an oxocarbenium ion.

Cycloadditions
In organic synthesis, vinyl oxocarbenium ions (structure on right) can be utilized in a wide range ofcycloaddition
In organic chemistry, a cycloaddition is a chemical reaction in which "two or more unsaturated molecules (or parts of the same molecule) combine with the formation of a cyclic adduct in which there is a net reduction of the bond multiplicity" ...
reactions. They are commonly employed as dienophiles in the Diels–Alder reaction
In organic chemistry, the Diels–Alder reaction is a chemical reaction between a conjugated diene and a substituted alkene, commonly termed the dienophile, to form a substituted cyclohexene derivative. It is the prototypical example of a peric ...
. An electron withdrawing ketone is often added to the dienophile to increase the rate of the reaction, and these ketones are often converted to vinyl oxocarbenium ions during the reaction It is not clear that an oxocarbenium ion necessarily will form, but Roush and co-workers demonstrated the oxocarbenium intermediate in the cyclization shown below. Two products were observed in this reaction, which could only form if the oxocarbenium ring is present as an intermediate. +3 +2 +2and +2cycloadditions with oxocarbenium intermediates have also been reported.

Aldol reaction
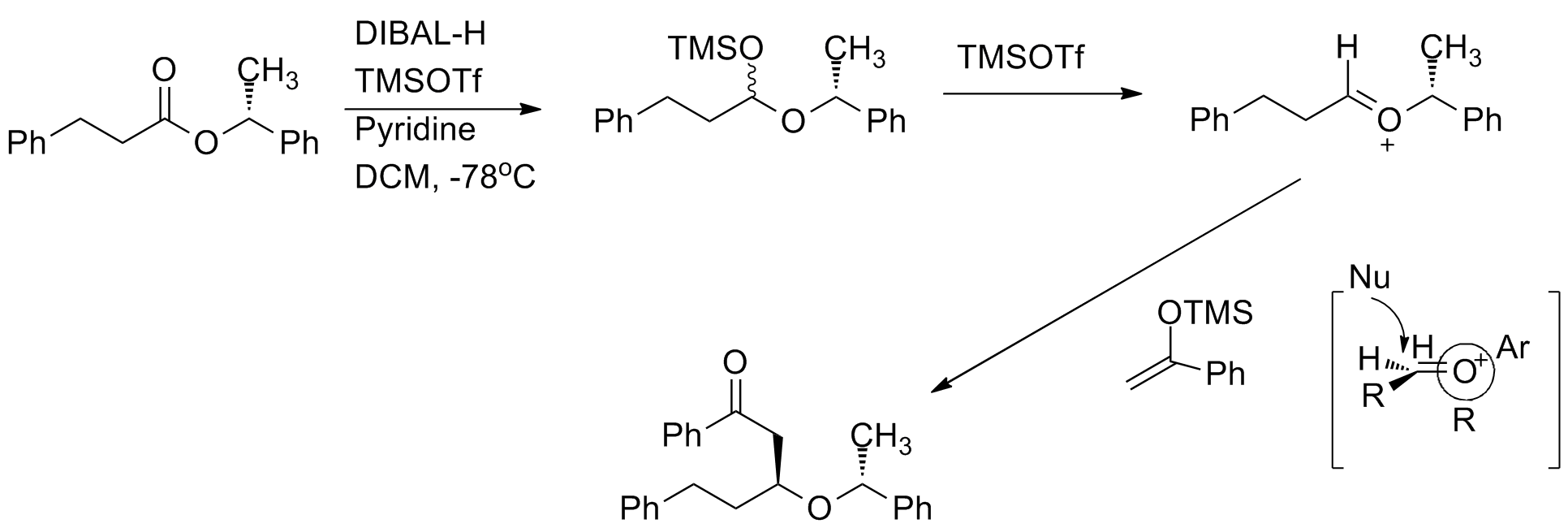
Chiral oxocarbenium ions have been exploited to carry out highly diastereoselective and enantioselective acetate aldol addition reactions. The oxocarbenium ion is used as anelectrophile
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carrie ...
in the reaction. When the methyl group increases in size, the diastereoselevtivity increases.

Examples from total synthesis
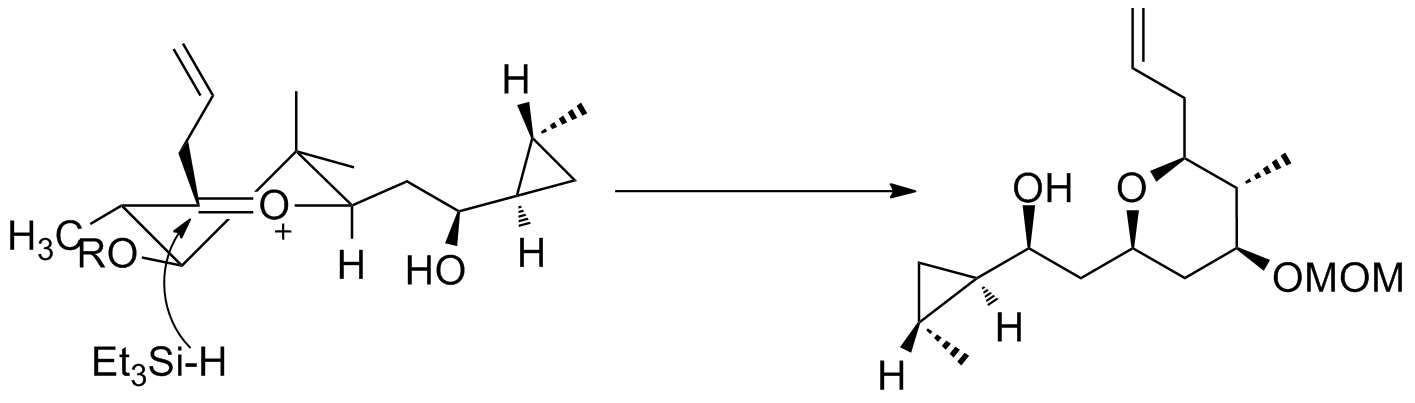
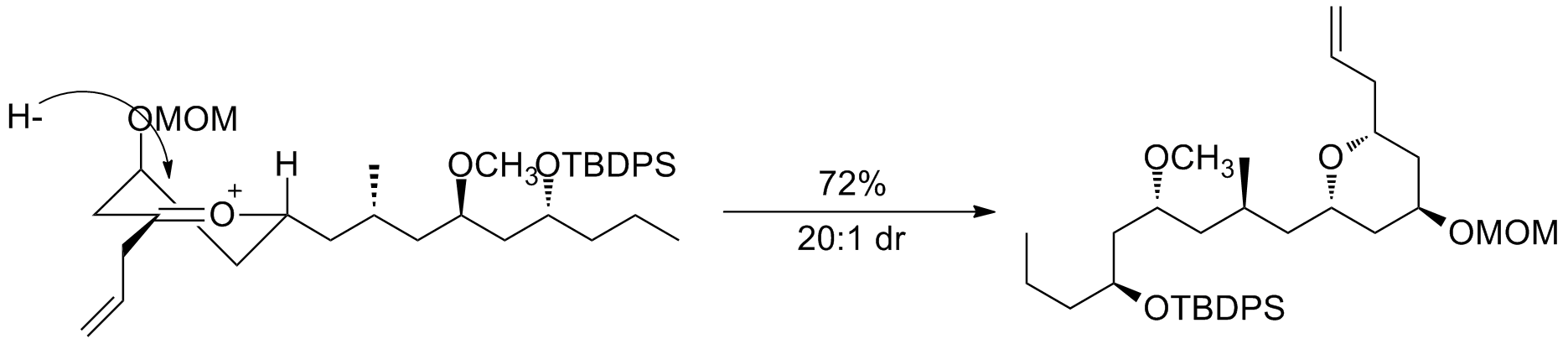
Oxocarbenium ions have been utilized in total synthesis on several occasions. A major subunit of (+)-clavosolide was synthesized with a reduction of a six-membered oxocarbenium ring. All the large substituents were found in an equatorial position, and the transformation went through the chair transition state, as predicted. A second example is seen in the key step of the synthesis of (−)-neopeltolide, which uses another six-membered oxocarbenium ring reduction for a diastereoselective hydride addition.
A second example is seen in the key step of the synthesis of (−)-neopeltolide, which uses another six-membered oxocarbenium ring reduction for a diastereoselective hydride addition.

Applications to biology
In biological systems, oxocarbenium ions are mostly seen during reactions ofcarbohydrate
In organic chemistry, a carbohydrate () is a biomolecule consisting of carbon (C), hydrogen (H) and oxygen (O) atoms, usually with a hydrogen–oxygen atom ratio of 2:1 (as in water) and thus with the empirical formula (where ''m'' may o ...
s. Since sugars are present in the structure of nucleic acids
Nucleic acids are biopolymers, macromolecules, essential to all known forms of life. They are composed of nucleotides, which are the monomers made of three components: a 5-carbon sugar, a phosphate group and a nitrogenous base. The two main cl ...
, with a ribose
Ribose is a simple sugar and carbohydrate with molecular formula C5H10O5 and the linear-form composition H−(C=O)−(CHOH)4−H. The naturally-occurring form, , is a component of the ribonucleotides from which RNA is built, and so this com ...
sugar
Sugar is the generic name for sweet-tasting, soluble carbohydrates, many of which are used in food. Simple sugars, also called monosaccharides, include glucose, fructose, and galactose. Compound sugars, also called disaccharides or do ...
present in RNA and a deoxyribose
Deoxyribose, or more precisely 2-deoxyribose, is a monosaccharide with idealized formula H−(C=O)−(CH2)−(CHOH)3−H. Its name indicates that it is a deoxy sugar, meaning that it is derived from the sugar ribose by loss of a hydroxy group. D ...
present in the structure of DNA, their chemistry plays an important role in wide range of cellular functions of nucleic acids. In addition to their functions in nucleotides, sugars are also used for structural components of organisms, as energy storage molecules, cell signaling molecules, protein modification and play key roles in the immune system
The immune system is a network of biological processes that protects an organism from diseases. It detects and responds to a wide variety of pathogens, from viruses to parasitic worms, as well as cancer cells and objects such as wood splinte ...
, fertilization
Fertilisation or fertilization (see spelling differences), also known as generative fertilisation, syngamy and impregnation, is the fusion of gametes to give rise to a new individual organism or offspring and initiate its development. Pro ...
, preventing pathogenesis
Pathogenesis is the process by which a disease or disorder develops. It can include factors which contribute not only to the onset of the disease or disorder, but also to its progression and maintenance. The word comes from Greek πάθος ''pat ...
, blood clotting
Coagulation, also known as clotting, is the process by which blood changes from a liquid to a gel, forming a blood clot. It potentially results in hemostasis, the cessation of blood loss from a damaged vessel, followed by repair. The mechan ...
, and development
Development or developing may refer to:
Arts
*Development hell, when a project is stuck in development
*Filmmaking, development phase, including finance and budgeting
*Development (music), the process thematic material is reshaped
* Photograph ...
. The abundance of sugar chemistry in biological processes leads many reaction mechanisms to proceed through oxocarbenium ions. Several important biological reactions that utilize oxocarbenium ions are outlined in this section.
Nucleotide biosynthesis
Nucleotides can undergo enzyme-catalyzed intramolecular cyclization in order to produce several important biological molecules. These cyclizations typically proceed through an oxocarbenium intermediate. An example of this reaction can be seen in the cyclization cyclic ADP ribose, which is an important molecule for intracellular calcium signaling.Glycosidases
Aglycosidase
Glycoside hydrolases (also called glycosidases or glycosyl hydrolases) catalyze the hydrolysis of glycosidic bonds in complex sugars. They are extremely common enzymes with roles in nature including degradation of biomass such as cellulose ...
is an enzyme that catalyzes the breakdown of a glycosidic linkage
A glycosidic bond or glycosidic linkage is a type of covalent bond that joins a carbohydrate (sugar) molecule to another group, which may or may not be another carbohydrate.
A glycosidic bond is formed between the hemiacetal or hemiketal group ...
to produce two smaller sugars. This process has important implications in the utilization of stored energy, like glycogen
Glycogen is a multibranched polysaccharide of glucose that serves as a form of energy storage in animals, fungi, and bacteria. The polysaccharide structure represents the main storage form of glucose in the body.
Glycogen functions as one of ...
in animals, as well as in the breakdown of cellulose
Cellulose is an organic compound with the formula , a polysaccharide consisting of a linear chain of several hundred to many thousands of β(1→4) linked D-glucose units. Cellulose is an important structural component of the primary cell wa ...
by organisms that feed on plants. In general, aspartic or glutamic acid residues in the active site of the enzyme catalyze the hydrolysis of the glycosidic bond. The mechanism of these enzymes involves an oxocarbenium ion intermediate, a general example of which is shown below.

See also
*Carbocation
A carbocation is an ion with a positively charged carbon atom. Among the simplest examples are the methenium , methanium and vinyl cations. Occasionally, carbocations that bear more than one positively charged carbon atom are also encount ...
* Chemical glycosylation
A chemical glycosylation reaction involves the coupling of a glycosyl donor, to a glycosyl acceptor forming a glycoside. If both the donor and acceptor are sugars, then the product is an oligosaccharide. The reaction requires activation with a ...
* Glycosyl donor A glycosyl donor is a carbohydrate mono- or oligosaccharide that will react with a suitable glycosyl acceptor to form a new glycosidic bond. By convention, the donor is the member of this pair that contains the resulting anomeric carbon of the new ...
* Glycosidase
Glycoside hydrolases (also called glycosidases or glycosyl hydrolases) catalyze the hydrolysis of glycosidic bonds in complex sugars. They are extremely common enzymes with roles in nature including degradation of biomass such as cellulose ...
* Oxocarbon anion
In chemistry, an oxocarbon anion is a negative ion consisting solely of carbon and oxygen atoms, and therefore having the general formula for some integers ''x'', ''y'', and ''n''.
The most common oxocarbon anions are carbonate, , and oxalate, ...
References
{{reflist Carbohydrate chemistry Organic reactions Reactive intermediates Carbocations Oxycations