neutron absorption on:
[Wikipedia]
[Google]
[Amazon]
 Neutron capture is a
Neutron capture is a
 At small
At small
 In engineering, the most important neutron absorber is 10 B, used as boron carbide in nuclear reactor
In engineering, the most important neutron absorber is 10 B, used as boron carbide in nuclear reactor
XSPlot an online neutron cross section plotter
at the International Atomic Energy Agency {{Authority control Nuclear physics Capture
 Neutron capture is a
Neutron capture is a nuclear reaction
In nuclear physics and nuclear chemistry, a nuclear reaction is a process in which two nuclei, or a nucleus and an external subatomic particle, collide to produce one or more new nuclides. Thus, a nuclear reaction must cause a transformation o ...
in which an atomic nucleus and one or more neutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the nuclei of atoms. Since protons and neutrons behav ...
s collide and merge to form a heavier nucleus. Since neutrons have no electric charge, they can enter a nucleus more easily than positively charged proton
A proton is a stable subatomic particle, symbol , H+, or 1H+ with a positive electric charge of +1 ''e'' elementary charge. Its mass is slightly less than that of a neutron and 1,836 times the mass of an electron (the proton–electron mass ...
s, which are repelled electrostatically.
Neutron capture plays a significant role in the cosmic nucleosynthesis
Nucleosynthesis is the process that creates new atomic nuclei from pre-existing nucleons (protons and neutrons) and nuclei. According to current theories, the first nuclei were formed a few minutes after the Big Bang, through nuclear reactions i ...
of heavy elements. In stars it can proceed in two ways: as a rapid process (r-process
In nuclear astrophysics, the rapid neutron-capture process, also known as the ''r''-process, is a set of nuclear reactions that is responsible for the creation of approximately half of the atomic nuclei heavier than iron, the "heavy elements", ...
) or a slow process (s-process
The slow neutron-capture process, or ''s''-process, is a series of reactions in nuclear astrophysics that occur in stars, particularly asymptotic giant branch stars. The ''s''-process is responsible for the creation (nucleosynthesis) of approximat ...
). Nuclei of masses greater than 56 cannot be formed by thermonuclear reaction
Nuclear fusion is a reaction in which two or more atomic nuclei are combined to form one or more different atomic nuclei and subatomic particles (neutrons or protons). The difference in mass between the reactants and products is manifest ...
s (i.e., by nuclear fusion
Nuclear fusion is a reaction in which two or more atomic nuclei are combined to form one or more different atomic nuclei and subatomic particles (neutrons or protons). The difference in mass between the reactants and products is manife ...
) but can be formed by neutron capture.
Neutron capture on protons yields a line at 2.223 MeV predicted and commonly observed in solar flares
A solar flare is an intense localized eruption of electromagnetic radiation in the Sun's atmosphere. Flares occur in active regions and are often, but not always, accompanied by coronal mass ejections, solar particle events, and other solar phe ...
.
Neutron capture at small neutron flux
neutron flux
The neutron flux, φ, is a scalar quantity used in nuclear physics and nuclear reactor physics. It is the total length travelled by all free neutrons per unit time and volume. Equivalently, it can be defined as the number of neutrons travelling ...
, as in a nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat from n ...
, a single neutron is captured by a nucleus. For example, when natural gold
Gold is a chemical element with the symbol Au (from la, aurum) and atomic number 79. This makes it one of the higher atomic number elements that occur naturally. It is a bright, slightly orange-yellow, dense, soft, malleable, and ductile m ...
(197Au) is irradiated by neutrons (n), the isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers ( mass numbers ...
198Au is formed in a highly excited state, and quickly decays to the ground state of 198Au by the emission of gamma ray
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically s ...
s (). In this process, the mass number increases by one. This is written as a formula in the form , or in short form . If thermal neutrons
The neutron detection temperature, also called the neutron energy, indicates a free neutron's kinetic energy, usually given in electron volts. The term ''temperature'' is used, since hot, thermal and cold neutrons are moderated in a medium with ...
are used, the process is called thermal capture.
The isotope 198Au is a beta emitter
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For exa ...
that decays into the mercury isotope 198Hg. In this process, the atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of every ...
rises by one.
Neutron capture at high neutron flux
Ther-process
In nuclear astrophysics, the rapid neutron-capture process, also known as the ''r''-process, is a set of nuclear reactions that is responsible for the creation of approximately half of the atomic nuclei heavier than iron, the "heavy elements", ...
happens inside stars if the neutron flux density is so high that the atomic nucleus has no time to decay via beta emission between neutron captures. The mass number therefore rises by a large amount while the atomic number (i.e., the element) stays the same. When further neutron capture is no longer possible, the highly unstable nuclei decay via many β− decays to beta-stable
Beta-decay stable isobars are the set of nuclides which cannot undergo beta decay, that is, the transformation of a neutron to a proton or a proton to a neutron within the nucleus. A subset of these nuclides are also stable with regards to doubl ...
isotopes of higher-numbered elements.
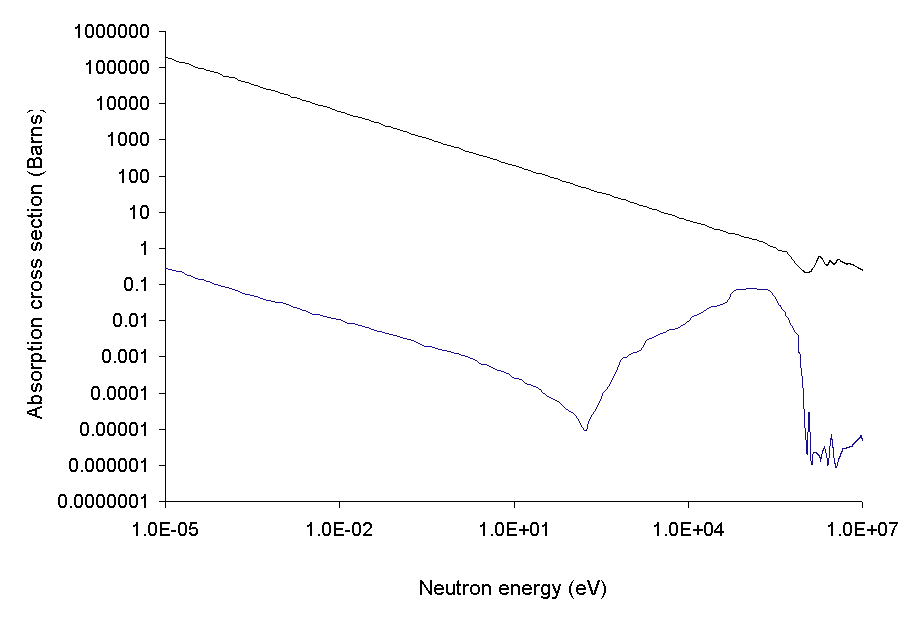
Capture cross section
The absorptionneutron cross section
In nuclear physics, the concept of a neutron cross section is used to express the likelihood of interaction between an incident neutron and a target nucleus. The neutron cross section σ can be defined as the area in cm2 for which the number of ...
of an isotope of a chemical element
A chemical element is a species of atoms that have a given number of protons in their nuclei, including the pure substance consisting only of that species. Unlike chemical compounds, chemical elements cannot be broken down into simpler sub ...
is the effective cross-sectional area that an atom of that isotope presents to absorption and is a measure of the probability of neutron capture. It is usually measured in barns
A barn is an agricultural building usually on farms and used for various purposes. In North America, a barn refers to structures that house livestock, including cattle and horses, as well as equipment and fodder, and often grain.Allen G. N ...
.
Absorption cross section is often highly dependent on neutron energy
The neutron detection temperature, also called the neutron energy, indicates a free neutron's kinetic energy, usually given in electron volts. The term ''temperature'' is used, since hot, thermal and cold neutrons are moderated in a medium with ...
. In general, the likelihood of absorption is proportional to the time the neutron is in the vicinity of the nucleus. The time spent in the vicinity of the nucleus is inversely proportional to the relative velocity between the neutron and nucleus. Other more specific issues modify this general principle. Two of the most specified measures are the cross section for thermal neutron
The neutron detection temperature, also called the neutron energy, indicates a free neutron's kinetic energy, usually given in electron volts. The term ''temperature'' is used, since hot, thermal and cold neutrons are moderated in a medium with ...
absorption and the resonance integral, which considers the contribution of absorption peaks at certain neutron energies specific to a particular nuclide
A nuclide (or nucleide, from nucleus, also known as nuclear species) is a class of atoms characterized by their number of protons, ''Z'', their number of neutrons, ''N'', and their nuclear energy state.
The word ''nuclide'' was coined by Truman ...
, usually above the thermal range, but encountered as neutron moderation slows the neutron from an original high energy.
The thermal energy of the nucleus also has an effect; as temperatures rise, Doppler broadening
In atomic physics, Doppler broadening is broadening of spectral lines due to the Doppler effect caused by a distribution of velocities of atoms or molecules. Different velocities of the emitting (or absorbing) particles result in different Dop ...
increases the chance of catching a resonance peak. In particular, the increase in uranium-238
Uranium-238 (238U or U-238) is the most common isotope of uranium found in nature, with a relative abundance of 99%. Unlike uranium-235, it is non-fissile, which means it cannot sustain a chain reaction in a thermal-neutron reactor. However, it ...
's ability to absorb neutrons at higher temperatures (and to do so without fissioning) is a negative feedback
Feedback occurs when outputs of a system are routed back as inputs as part of a chain of cause-and-effect that forms a circuit or loop. The system can then be said to ''feed back'' into itself. The notion of cause-and-effect has to be handled c ...
mechanism that helps keep nuclear reactors under control.
Thermochemical significance
Neutron capture is involved in the formation of isotopes of chemical elements. The energy of neutron capture thus intervenes in the standard enthalpy of formation of isotopes.Uses
Neutron activation analysis
Neutron activation analysis (NAA) is the nuclear process used for determining the concentrations of elements in many materials. NAA allows discrete sampling of elements as it disregards the chemical form of a sample, and focuses solely on atomic ...
can be used to remotely detect the chemical composition of materials. This is because different elements release different characteristic radiation when they absorb neutrons. This makes it useful in many fields related to mineral exploration and security.
Neutron absorbers
 In engineering, the most important neutron absorber is 10 B, used as boron carbide in nuclear reactor
In engineering, the most important neutron absorber is 10 B, used as boron carbide in nuclear reactor control rod
Control rods are used in nuclear reactors to control the rate of fission of the nuclear fuel – uranium or plutonium. Their compositions include chemical elements such as boron, cadmium, silver, hafnium, or indium, that are capable of absorbing ...
s or as boric acid
Boric acid, more specifically orthoboric acid, is a compound of boron, oxygen, and hydrogen with formula . It may also be called hydrogen borate or boracic acid. It is usually encountered as colorless crystals or a white powder, that dissolves ...
as a coolant water additive in pressurized water reactor
A pressurized water reactor (PWR) is a type of light-water nuclear reactor. PWRs constitute the large majority of the world's nuclear power plants (with notable exceptions being the UK, Japan and Canada). In a PWR, the primary coolant (water) is ...
s. Other neutron absorbers used in nuclear reactors are xenon
Xenon is a chemical element with the symbol Xe and atomic number 54. It is a dense, colorless, odorless noble gas found in Earth's atmosphere in trace amounts. Although generally unreactive, it can undergo a few chemical reactions such as the ...
, cadmium
Cadmium is a chemical element with the symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12, zinc and mercury. Like zinc, it demonstrates oxidation state +2 in most of ...
, hafnium
Hafnium is a chemical element with the Symbol (chemistry), symbol Hf and atomic number 72. A lustre (mineralogy), lustrous, silvery gray, tetravalence, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirco ...
, gadolinium
Gadolinium is a chemical element with the symbol Gd and atomic number 64. Gadolinium is a silvery-white metal when oxidation is removed. It is only slightly malleable and is a ductile rare-earth element. Gadolinium reacts with atmospheric oxygen ...
, cobalt
Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, pro ...
, samarium
Samarium is a chemical element with symbol Sm and atomic number 62. It is a moderately hard silvery metal that slowly oxidizes in air. Being a typical member of the lanthanide series, samarium usually has the oxidation state +3. Compounds of samar ...
, titanium
Titanium is a chemical element with the symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resistant to corrosion in ...
, dysprosium
Dysprosium is the chemical element with the symbol Dy and atomic number 66. It is a rare-earth element in the lanthanide series with a metallic silver luster. Dysprosium is never found in nature as a free element, though, like other lanthanides, ...
, erbium
Erbium is a chemical element with the symbol Er and atomic number 68. A silvery-white solid metal when artificially isolated, natural erbium is always found in chemical combination with other elements. It is a lanthanide, a rare-earth element, orig ...
, europium
Europium is a chemical element with the symbol Eu and atomic number 63. Europium is the most reactive lanthanide by far, having to be stored under an inert fluid to protect it from atmospheric oxygen or moisture. Europium is also the softest lanth ...
, molybdenum
Molybdenum is a chemical element with the symbol Mo and atomic number 42 which is located in period 5 and group 6. The name is from Neo-Latin ''molybdaenum'', which is based on Ancient Greek ', meaning lead, since its ores were confused with l ...
and ytterbium
Ytterbium is a chemical element with the symbol Yb and atomic number 70. It is a metal, the fourteenth and penultimate element in the lanthanide series, which is the basis of the relative stability of its +2 oxidation state. However, like the ...
. All of these occur in nature as mixtures of various isotopes, some of which are excellent neutron absorbers. They may occur in compounds such as molybdenum boride, hafnium diboride
Hafnium diboride belongs to the class of ultra-high-temperature ceramics, a type of ceramic composed of hafnium and boron. It has a melting temperature of about 3250 °C. It is an unusual ceramic, having relatively high thermal and electrical c ...
, titanium diboride
Titanium diboride (TiB2) is an extremely hard ceramic which has excellent heat conductivity, oxidation stability and wear resistance. TiB2 is also a reasonable electrical conductor,J. Schmidt et al. "Preparation of titanium diboride TiB2 by spark ...
, dysprosium titanate and gadolinium titanate.
Hafnium
Hafnium is a chemical element with the Symbol (chemistry), symbol Hf and atomic number 72. A lustre (mineralogy), lustrous, silvery gray, tetravalence, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirco ...
absorbs neutrons avidly and it can be used in reactor control rod
Control rods are used in nuclear reactors to control the rate of fission of the nuclear fuel – uranium or plutonium. Their compositions include chemical elements such as boron, cadmium, silver, hafnium, or indium, that are capable of absorbing ...
s. However, it is found in the same ores as zirconium
Zirconium is a chemical element with the symbol Zr and atomic number 40. The name ''zirconium'' is taken from the name of the mineral zircon, the most important source of zirconium. The word is related to Persian '' zargun'' (zircon; ''zar-gun'', ...
, which shares the same outer electron shell configuration and thus has similar chemical properties. Their nuclear properties are profoundly different: hafnium absorbs neutrons 600 times better than zirconium. The latter, being essentially transparent to neutrons, is prized for internal reactor parts, including the metallic cladding of the fuel rod
Nuclear fuel is material used in nuclear power stations to produce heat to power turbines. Heat is created when nuclear fuel undergoes nuclear fission.
Most nuclear fuels contain heavy fissile actinide elements that are capable of undergoing ...
s. To use these elements in their respective applications, the zirconium must be separated from the naturally co-occurring hafnium. This can be accomplished economically with ion-exchange resin
An ion-exchange resin or ion-exchange polymer is a resin or polymer that acts as a medium for ion exchange. It is an insoluble matrix (or support structure) normally in the form of small (0.25–1.43 mm radius) microbeads, usually white or ye ...
s.
See also
*Beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For e ...
*Induced radioactivity
Induced radioactivity, also called artificial radioactivity or man-made radioactivity, is the process of using radiation to make a previously stable material radioactive. The husband and wife team of Irène Joliot-Curie and Frédéric Joliot-Curie ...
*List of particles
This is a list of known and hypothesized particles.
Elementary particles
Elementary particles are particles with no measurable internal structure; that is, it is unknown whether they are composed of other particles. They are the fundamental ob ...
*Neutron emission
Neutron emission is a mode of radioactive decay in which one or more neutrons are ejected from a nucleus. It occurs in the most neutron-rich/proton-deficient nuclides, and also from excited states of other nuclides as in photoneutron emission and ...
*Radioactive decay
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is consi ...
*Rays: α — β — γ — δ — ε
*p-process
The term p-process (''p'' for proton) is used in two ways in the scientific literature concerning the astrophysical origin of the elements ( nucleosynthesis). Originally it referred to a proton capture process which is the source of certain, nat ...
(proton capture)
References
External links
XSPlot an online neutron cross section plotter
at the International Atomic Energy Agency {{Authority control Nuclear physics Capture