Gamma Radiation on:
[Wikipedia]
[Google]
[Amazon]
 A gamma ray, also known as gamma radiation (symbol γ or ), is a penetrating form of
A gamma ray, also known as gamma radiation (symbol γ or ), is a penetrating form of

 One example of gamma ray production due to radionuclide decay is the decay scheme for cobalt-60, as illustrated in the accompanying diagram. First, decays to excited by
One example of gamma ray production due to radionuclide decay is the decay scheme for cobalt-60, as illustrated in the accompanying diagram. First, decays to excited by

 Due to their penetrating nature, gamma rays require large amounts of shielding mass to reduce them to levels which are not harmful to living cells, in contrast to
Due to their penetrating nature, gamma rays require large amounts of shielding mass to reduce them to levels which are not harmful to living cells, in contrast to
 When a gamma ray passes through matter, the probability for absorption is proportional to the thickness of the layer, the density of the material, and the absorption cross section of the material. The total absorption shows an exponential decrease of intensity with distance from the incident surface:
:
where x is the thickness of the material from the incident surface, μ= ''n''σ is the absorption coefficient, measured in cm−1, ''n'' the number of atoms per cm3 of the material (atomic density) and σ the absorption cross section in cm2.
As it passes through matter, gamma radiation ionizes via three processes:
*The photoelectric effect: This describes the case in which a gamma photon interacts with and transfers its energy to an atomic electron, causing the ejection of that electron from the atom. The kinetic energy of the resulting photoelectron is equal to the energy of the incident gamma photon minus the energy that originally bound the electron to the atom (binding energy). The photoelectric effect is the dominant energy transfer mechanism for X-ray and gamma ray photons with energies below 50 keV (thousand electronvolts), but it is much less important at higher energies.
* Compton scattering: This is an interaction in which an incident gamma photon loses enough energy to an atomic electron to cause its ejection, with the remainder of the original photon's energy emitted as a new, lower energy gamma photon whose emission direction is different from that of the incident gamma photon, hence the term "scattering". The probability of Compton scattering decreases with increasing photon energy. It is thought to be the principal absorption mechanism for gamma rays in the intermediate energy range 100 keV to 10 MeV. It is relatively independent of the
When a gamma ray passes through matter, the probability for absorption is proportional to the thickness of the layer, the density of the material, and the absorption cross section of the material. The total absorption shows an exponential decrease of intensity with distance from the incident surface:
:
where x is the thickness of the material from the incident surface, μ= ''n''σ is the absorption coefficient, measured in cm−1, ''n'' the number of atoms per cm3 of the material (atomic density) and σ the absorption cross section in cm2.
As it passes through matter, gamma radiation ionizes via three processes:
*The photoelectric effect: This describes the case in which a gamma photon interacts with and transfers its energy to an atomic electron, causing the ejection of that electron from the atom. The kinetic energy of the resulting photoelectron is equal to the energy of the incident gamma photon minus the energy that originally bound the electron to the atom (binding energy). The photoelectric effect is the dominant energy transfer mechanism for X-ray and gamma ray photons with energies below 50 keV (thousand electronvolts), but it is much less important at higher energies.
* Compton scattering: This is an interaction in which an incident gamma photon loses enough energy to an atomic electron to cause its ejection, with the remainder of the original photon's energy emitted as a new, lower energy gamma photon whose emission direction is different from that of the incident gamma photon, hence the term "scattering". The probability of Compton scattering decreases with increasing photon energy. It is thought to be the principal absorption mechanism for gamma rays in the intermediate energy range 100 keV to 10 MeV. It is relatively independent of the
 Gamma rays provide information about some of the most energetic phenomena in the universe; however, they are largely absorbed by the Earth's atmosphere. Instruments aboard high-altitude balloons and satellites missions, such as the Fermi Gamma-ray Space Telescope, provide our only view of the universe in gamma rays.
Gamma-induced molecular changes can also be used to alter the properties of semi-precious stones, and is often used to change white topaz into blue topaz.
Non-contact industrial sensors commonly use sources of gamma radiation in refining, mining, chemicals, food, soaps and detergents, and pulp and paper industries, for the measurement of levels, density, and thicknesses. Gamma-ray sensors are also used for measuring the fluid levels in water and oil industries. Typically, these use Co-60 or Cs-137 isotopes as the radiation source.
In the US, gamma ray detectors are beginning to be used as part of the
Gamma rays provide information about some of the most energetic phenomena in the universe; however, they are largely absorbed by the Earth's atmosphere. Instruments aboard high-altitude balloons and satellites missions, such as the Fermi Gamma-ray Space Telescope, provide our only view of the universe in gamma rays.
Gamma-induced molecular changes can also be used to alter the properties of semi-precious stones, and is often used to change white topaz into blue topaz.
Non-contact industrial sensors commonly use sources of gamma radiation in refining, mining, chemicals, food, soaps and detergents, and pulp and paper industries, for the measurement of levels, density, and thicknesses. Gamma-ray sensors are also used for measuring the fluid levels in water and oil industries. Typically, these use Co-60 or Cs-137 isotopes as the radiation source.
In the US, gamma ray detectors are beginning to be used as part of the
 The conventional distinction between X-rays and gamma rays has changed over time. Originally, the electromagnetic radiation emitted by X-ray tubes almost invariably had a longer
The conventional distinction between X-rays and gamma rays has changed over time. Originally, the electromagnetic radiation emitted by X-ray tubes almost invariably had a longer  For example, modern high-energy X-rays produced by
For example, modern high-energy X-rays produced by
Basic reference on several types of radiation
Radiation Q & ARadiation information
The Lund/LBNL Nuclear Data Search
– Contains information on gamma-ray energies from isotopes.
The LIVEChart of Nuclides – IAEA
with filter on gamma-ray energy
Health Physics Society Public Education Website
{{Authority control Electromagnetic spectrum IARC Group 1 carcinogens Nuclear physics Radiation Radioactivity Articles containing video clips
 A gamma ray, also known as gamma radiation (symbol γ or ), is a penetrating form of
A gamma ray, also known as gamma radiation (symbol γ or ), is a penetrating form of electromagnetic radiation
In physics, electromagnetic radiation (EMR) consists of waves of the electromagnetic (EM) field, which propagate through space and carry momentum and electromagnetic radiant energy. It includes radio waves, microwaves, infrared, (visib ...
arising from the radioactive decay of atomic nuclei
The atomic nucleus is the small, dense region consisting of protons and neutrons at the center of an atom, discovered in 1911 by Ernest Rutherford based on the 1909 Geiger–Marsden gold foil experiment. After the discovery of the neutron ...
. It consists of the shortest wavelength electromagnetic waves, typically shorter than those of X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
s. With frequencies above 30 exahertz (), it imparts the highest photon energy
Photon energy is the energy carried by a single photon. The amount of energy is directly proportional to the photon's electromagnetic frequency and thus, equivalently, is inversely proportional to the wavelength. The higher the photon's frequency, ...
. Paul Villard, a French chemist
A chemist (from Greek ''chēm(ía)'' alchemy; replacing ''chymist'' from Medieval Latin ''alchemist'') is a scientist trained in the study of chemistry. Chemists study the composition of matter and its properties. Chemists carefully describe th ...
and physicist
A physicist is a scientist who specializes in the field of physics, which encompasses the interactions of matter and energy at all length and time scales in the physical universe.
Physicists generally are interested in the root or ultimate cau ...
, discovered gamma radiation in 1900 while studying radiation
In physics, radiation is the emission or transmission of energy in the form of waves or particles through space or through a material medium. This includes:
* ''electromagnetic radiation'', such as radio waves, microwaves, infrared, visi ...
emitted by radium
Radium is a chemical element with the symbol Ra and atomic number 88. It is the sixth element in group 2 of the periodic table, also known as the alkaline earth metals. Pure radium is silvery-white, but it readily reacts with nitrogen (rat ...
. In 1903, Ernest Rutherford named this radiation ''gamma rays'' based on their relatively strong penetration of matter
In classical physics and general chemistry, matter is any substance that has mass and takes up space by having volume. All everyday objects that can be touched are ultimately composed of atoms, which are made up of interacting subatomic part ...
; in 1900 he had already named two less penetrating types of decay radiation (discovered by Henri Becquerel) alpha rays and beta rays
A beta particle, also called beta ray or beta radiation (symbol β), is a high-energy, high-speed electron or positron emitted by the radioactive decay of an atomic nucleus during the process of beta decay. There are two forms of beta decay, β� ...
in ascending order of penetrating power.
Gamma rays from radioactive decay are in the energy range from a few kiloelectronvolts ( keV) to approximately 8 megaelectronvolts ( MeV), corresponding to the typical energy levels in nuclei with reasonably long lifetimes. The energy spectrum of gamma rays can be used to identify the decaying radionuclides using gamma spectroscopy
Gamma-ray spectroscopy is the quantitative study of the energy spectra of gamma-ray sources, such as in the nuclear industry, geochemical investigation, and astrophysics.
Most radioactive sources produce gamma rays, which are of various energie ...
. Very-high-energy gamma rays in the 100–1000 teraelectronvolt (TeV
TEV may refer to:
* Transient Earth Voltage: a term for voltages appearing on the metal work of switchgear due to internal partial discharges
* TeV, or teraelectronvolt or trillion electron volt, a measure of energy
* Total Enterprise Value, a ...
) range have been observed from sources such as the Cygnus X-3 microquasar.
Natural sources of gamma rays originating on Earth are mostly a result of radioactive decay and secondary radiation from atmospheric interactions with cosmic ray
Cosmic rays are high-energy particles or clusters of particles (primarily represented by protons or atomic nuclei) that move through space at nearly the speed of light. They originate from the Sun, from outside of the Solar System in our own ...
particles. However, there are other rare natural sources, such as terrestrial gamma-ray flashes, which produce gamma rays from electron action upon the nucleus. Notable artificial sources of gamma rays include fission, such as that which occurs in nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
s, and high energy physics experiments, such as neutral pion decay
In particle physics, a pion (or a pi meson, denoted with the Greek letter pi: ) is any of three subatomic particles: , , and . Each pion consists of a quark and an antiquark and is therefore a meson. Pions are the lightest mesons and, more gene ...
and nuclear fusion.
Gamma rays and X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
s are both electromagnetic radiation, and since they overlap in the electromagnetic spectrum
The electromagnetic spectrum is the range of frequencies (the spectrum) of electromagnetic radiation and their respective wavelengths and photon energies.
The electromagnetic spectrum covers electromagnetic waves with frequencies ranging fro ...
, the terminology varies between scientific disciplines. In some fields of physics, they are distinguished by their origin: Gamma rays are created by nuclear decay while X-rays originate outside the nucleus. In astrophysics
Astrophysics is a science that employs the methods and principles of physics and chemistry in the study of astronomical objects and phenomena. As one of the founders of the discipline said, Astrophysics "seeks to ascertain the nature of the h ...
, gamma rays are conventionally defined as having photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they alwa ...
energies above 100 keV and are the subject of gamma ray astronomy, while radiation below 100 keV is classified as X-rays and is the subject of X-ray astronomy.
Gamma rays are ionizing radiation and are thus hazardous to life. Due to their high penetration power, they can damage bone marrow and internal organs. Unlike alpha and beta rays, they easily pass through the body and thus pose a formidable radiation protection
Radiation protection, also known as radiological protection, is defined by the International Atomic Energy Agency (IAEA) as "The protection of people from harmful effects of exposure to ionizing radiation, and the means for achieving this". Expos ...
challenge, requiring shielding made from dense materials such as lead or concrete. On Earth
Earth is the third planet from the Sun and the only astronomical object known to harbor life. While large volumes of water can be found throughout the Solar System, only Earth sustains liquid surface water. About 71% of Earth's sur ...
, the magnetosphere
In astronomy and planetary science, a magnetosphere is a region of space surrounding an astronomical object in which charged particles are affected by that object's magnetic field. It is created by a celestial body with an active interior d ...
protects life from most types of lethal cosmic radiation other than gamma rays, which are absorbed by 0.53 bars of atmosphere as they penetrate the atmosphere.
Gamma rays cannot be reflected by a mirror and their wavelengths are so small that they will pass between the atoms in a detector.
History of discovery
The first gamma ray source to be discovered was the radioactive decay process called ''gamma decay''. In this type of decay, an excited nucleus emits a gamma ray almost immediately upon formation.It is now understood that a nuclear isomeric transition, however, can produce inhibited gamma decay with a measurable and much longer half-life. Paul Villard, a French chemist and physicist, discovered gamma radiation in 1900, while studying radiation emitted fromradium
Radium is a chemical element with the symbol Ra and atomic number 88. It is the sixth element in group 2 of the periodic table, also known as the alkaline earth metals. Pure radium is silvery-white, but it readily reacts with nitrogen (rat ...
. Villard knew that his described radiation was more powerful than previously described types of rays from radium, which included beta rays, first noted as "radioactivity" by Henri Becquerel in 1896, and alpha rays, discovered as a less penetrating form of radiation by Rutherford, in 1899. However, Villard did not consider naming them as a different fundamental type. Later, in 1903, Villard's radiation was recognized as being of a type fundamentally different from previously named rays by Ernest Rutherford, who named Villard's rays "gamma rays" by analogy with the beta and alpha rays that Rutherford had differentiated in 1899. The "rays" emitted by radioactive elements were named in order of their power to penetrate various materials, using the first three letters of the Greek alphabet: alpha rays as the least penetrating, followed by beta rays, followed by gamma rays as the most penetrating. Rutherford also noted that gamma rays were not deflected (or at least, not deflected) by a magnetic field, another property making them unlike alpha and beta rays.
Gamma rays were first thought to be particles with mass, like alpha and beta rays. Rutherford initially believed that they might be extremely fast beta particles, but their failure to be deflected by a magnetic field indicated that they had no charge. In 1914, gamma rays were observed to be reflected from crystal surfaces, proving that they were electromagnetic radiation. Rutherford and his co-worker Edward Andrade measured the wavelengths of gamma rays from radium, and found they were similar to X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
s, but with shorter wavelengths and thus, higher frequency. This was eventually recognized as giving them more energy per photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless, so they alwa ...
, as soon as the latter term became generally accepted. A gamma decay was then understood to usually emit a gamma photon.
Sources
Natural sources of gamma rays on Earth include gamma decay from naturally occurring radioisotopes such as potassium-40, and also as a secondary radiation from various atmospheric interactions withcosmic ray
Cosmic rays are high-energy particles or clusters of particles (primarily represented by protons or atomic nuclei) that move through space at nearly the speed of light. They originate from the Sun, from outside of the Solar System in our own ...
particles. Some rare terrestrial natural sources that produce gamma rays that are not of a nuclear origin, are lightning strike
A lightning strike or lightning bolt is an electric discharge between the atmosphere and the ground. Most originate in a cumulonimbus cloud and terminate on the ground, called cloud-to-ground (CG) lightning. A less common type of strike, ground- ...
s and terrestrial gamma-ray flashes, which produce high energy emissions from natural high-energy voltages. Gamma rays are produced by a number of astronomical processes in which very high-energy electrons are produced. Such electrons produce secondary gamma rays by the mechanisms of ''bremsstrahlung
''Bremsstrahlung'' (), from "to brake" and "radiation"; i.e., "braking radiation" or "deceleration radiation", is electromagnetic radiation produced by the deceleration of a charged particle when deflected by another charged particle, typical ...
'', inverse Compton scattering and synchrotron radiation. A large fraction of such astronomical gamma rays are screened by Earth's atmosphere. Notable artificial sources of gamma rays include fission, such as occurs in nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
s, as well as high energy physics experiments, such as neutral pion decay
In particle physics, a pion (or a pi meson, denoted with the Greek letter pi: ) is any of three subatomic particles: , , and . Each pion consists of a quark and an antiquark and is therefore a meson. Pions are the lightest mesons and, more gene ...
and nuclear fusion.
A sample of gamma ray-emitting material that is used for irradiating or imaging is known as a gamma source. It is also called a radioactive source, isotope source, or radiation source, though these more general terms also apply to alpha and beta-emitting devices. Gamma sources are usually sealed to prevent radioactive contamination, and transported in heavy shielding.
Radioactive decay (gamma decay)
Gamma rays are produced during gamma decay, which normally occurs after other forms of decay occur, such asalpha
Alpha (uppercase , lowercase ; grc, ἄλφα, ''álpha'', or ell, άλφα, álfa) is the first letter of the Greek alphabet. In the system of Greek numerals, it has a value of one. Alpha is derived from the Phoenician letter aleph , whi ...
or beta
Beta (, ; uppercase , lowercase , or cursive ; grc, βῆτα, bē̂ta or ell, βήτα, víta) is the second letter of the Greek alphabet. In the system of Greek numerals, it has a value of 2. In Modern Greek, it represents the voiced labiod ...
decay. A radioactive nucleus can decay by the emission of an or particle. The daughter nucleus that results is usually left in an excited state. It can then decay to a lower energy state by emitting a gamma ray photon, in a process called gamma decay.
The emission of a gamma ray from an excited nucleus typically requires only 10−12 seconds. Gamma decay may also follow nuclear reaction
In nuclear physics and nuclear chemistry, a nuclear reaction is a process in which two nuclei, or a nucleus and an external subatomic particle, collide to produce one or more new nuclides. Thus, a nuclear reaction must cause a transformatio ...
s such as neutron capture
Neutron capture is a nuclear reaction in which an atomic nucleus and one or more neutrons collide and merge to form a heavier nucleus. Since neutrons have no electric charge, they can enter a nucleus more easily than positively charged protons ...
, nuclear fission, or nuclear fusion. Gamma decay is also a mode of relaxation of many excited states of atomic nuclei following other types of radioactive decay, such as beta decay, so long as these states possess the necessary component of nuclear spin. When high-energy gamma rays, electrons, or protons bombard materials, the excited atoms emit characteristic "secondary" gamma rays, which are products of the creation of excited nuclear states in the bombarded atoms. Such transitions, a form of nuclear gamma fluorescence
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation. It is a form of luminescence. In most cases, the emitted light has a longer wavelength, and therefore a lower photon energy, tha ...
, form a topic in nuclear physics
Nuclear physics is the field of physics that studies atomic nuclei and their constituents and interactions, in addition to the study of other forms of nuclear matter.
Nuclear physics should not be confused with atomic physics, which studies t ...
called gamma spectroscopy
Gamma-ray spectroscopy is the quantitative study of the energy spectra of gamma-ray sources, such as in the nuclear industry, geochemical investigation, and astrophysics.
Most radioactive sources produce gamma rays, which are of various energie ...
. Formation of fluorescent gamma rays are a rapid subtype of radioactive gamma decay.
In certain cases, the excited nuclear state that follows the emission of a beta particle or other type of excitation, may be more stable than average, and is termed a metastable excited state, if its decay takes (at least) 100 to 1000 times longer than the average 10−12 seconds. Such relatively long-lived excited nuclei are termed nuclear isomers, and their decays are termed isomeric transitions. Such nuclei have half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable ...
s that are more easily measurable, and rare nuclear isomers are able to stay in their excited state for minutes, hours, days, or occasionally far longer, before emitting a gamma ray. The process of isomeric transition is therefore similar to any gamma emission, but differs in that it involves the intermediate metastable excited state(s) of the nuclei. Metastable states are often characterized by high nuclear spin
In atomic physics, the spin quantum number is a quantum number (designated ) which describes the intrinsic angular momentum (or spin angular momentum, or simply spin) of an electron or other particle. The phrase was originally used to describe ...
, requiring a change in spin of several units or more with gamma decay, instead of a single unit transition that occurs in only 10−12 seconds. The rate of gamma decay is also slowed when the energy of excitation of the nucleus is small.
An emitted gamma ray from any type of excited state may transfer its energy directly to any electron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have n ...
s, but most probably to one of the K shell electrons of the atom, causing it to be ejected from that atom, in a process generally termed the photoelectric effect (external gamma rays and ultraviolet rays may also cause this effect). The photoelectric effect should not be confused with the internal conversion process, in which a gamma ray photon is not produced as an intermediate particle (rather, a "virtual gamma ray" may be thought to mediate the process).
Decay schemes
beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which a beta particle (fast energetic electron or positron) is emitted from an atomic nucleus, transforming the original nuclide to an isobar of that nuclide. For ...
emission of an electron of . Then the excited decays to the ground state (see nuclear shell model
In nuclear physics, atomic physics, and nuclear chemistry, the nuclear shell model is a model of the atomic nucleus which uses the Pauli exclusion principle to describe the structure of the nucleus in terms of energy levels. The first shell ...
) by emitting gamma rays in succession of 1.17 MeV followed by . This path is followed 99.88% of the time:
:
Another example is the alpha decay of to form ; which is followed by gamma emission. In some cases, the gamma emission spectrum of the daughter nucleus is quite simple, (e.g. /) while in other cases, such as with (/ and /), the gamma emission spectrum is complex, revealing that a series of nuclear energy levels exist.
Particle physics
Gamma rays are produced in many processes ofparticle physics
Particle physics or high energy physics is the study of fundamental particles and forces that constitute matter and radiation. The fundamental particles in the universe are classified in the Standard Model as fermions (matter particles) an ...
. Typically, gamma rays are the products of neutral systems which decay through electromagnetic interactions (rather than a weak
Weak may refer to:
Songs
* "Weak" (AJR song), 2016
* "Weak" (Melanie C song), 2011
* "Weak" (SWV song), 1993
* "Weak" (Skunk Anansie song), 1995
* "Weak", a song by Seether from '' Seether: 2002-2013''
Television episodes
* "Weak" (''Fear t ...
or strong
Strong may refer to:
Education
* The Strong, an educational institution in Rochester, New York, United States
* Strong Hall (Lawrence, Kansas), an administrative hall of the University of Kansas
* Strong School, New Haven, Connecticut, United S ...
interaction). For example, in an electron–positron annihilation, the usual products are two gamma ray photons. If the annihilating electron and positron are at rest, each of the resulting gamma rays has an energy of ~ 511 keV and frequency of ~ . Similarly, a neutral pion most often decays into two photons. Many other hadron
In particle physics, a hadron (; grc, ἁδρός, hadrós; "stout, thick") is a composite subatomic particle made of two or more quarks held together by the strong interaction. They are analogous to molecules that are held together by the ...
s and massive bosons also decay electromagnetically. High energy physics experiments, such as the Large Hadron Collider
The Large Hadron Collider (LHC) is the world's largest and highest-energy particle collider. It was built by the European Organization for Nuclear Research (CERN) between 1998 and 2008 in collaboration with over 10,000 scientists and hundr ...
, accordingly employ substantial radiation shielding. Because subatomic particle
In physical sciences, a subatomic particle is a particle that composes an atom. According to the Standard Model of particle physics, a subatomic particle can be either a composite particle, which is composed of other particles (for example, a p ...
s mostly have far shorter wavelengths than atomic nuclei, particle physics gamma rays are generally several orders of magnitude more energetic than nuclear decay gamma rays. Since gamma rays are at the top of the electromagnetic spectrum in terms of energy, all extremely high-energy photons are gamma rays; for example, a photon having the Planck energy would be a gamma ray.
Other sources
A few gamma rays in astronomy are known to arise from gamma decay (see discussion of SN1987A), but most do not. Photons from astrophysical sources that carry energy in the gamma radiation range are often explicitly called gamma-radiation. In addition to nuclear emissions, they are often produced by sub-atomic particle and particle-photon interactions. Those include electron-positron annihilation,neutral pion decay
In particle physics, a pion (or a pi meson, denoted with the Greek letter pi: ) is any of three subatomic particles: , , and . Each pion consists of a quark and an antiquark and is therefore a meson. Pions are the lightest mesons and, more gene ...
, bremsstrahlung
''Bremsstrahlung'' (), from "to brake" and "radiation"; i.e., "braking radiation" or "deceleration radiation", is electromagnetic radiation produced by the deceleration of a charged particle when deflected by another charged particle, typical ...
, inverse Compton scattering, and synchrotron radiation.
Laboratory sources
In October 2017, scientists from various European universities proposed a means for sources of GeV photons using lasers as exciters through a controlled interplay between the cascade and anomalous radiative trapping.Terrestrial thunderstorms
Thunderstorm
A thunderstorm, also known as an electrical storm or a lightning storm, is a storm characterized by the presence of lightning and its acoustic effect on the Earth's atmosphere, known as thunder. Relatively weak thunderstorms are some ...
s can produce a brief pulse of gamma radiation called a terrestrial gamma-ray flash. These gamma rays are thought to be produced by high intensity static electric fields accelerating electrons, which then produce gamma rays by bremsstrahlung
''Bremsstrahlung'' (), from "to brake" and "radiation"; i.e., "braking radiation" or "deceleration radiation", is electromagnetic radiation produced by the deceleration of a charged particle when deflected by another charged particle, typical ...
as they collide with and are slowed by atoms in the atmosphere. Gamma rays up to 100 MeV can be emitted by terrestrial thunderstorms, and were discovered by space-borne observatories. This raises the possibility of health risks to passengers and crew on aircraft flying in or near thunderclouds.
Solar flares
The most effusive solar flares emit across the entire EM spectrum, including γ-rays. The first confident observation occurred in 1972.Cosmic rays
Extraterrestrial, high energy gamma rays include the gamma ray background produced when cosmic rays (either high speed electrons or protons) collide with ordinary matter, producing pair-production gamma rays at 511 keV. Alternatively,bremsstrahlung
''Bremsstrahlung'' (), from "to brake" and "radiation"; i.e., "braking radiation" or "deceleration radiation", is electromagnetic radiation produced by the deceleration of a charged particle when deflected by another charged particle, typical ...
are produced at energies of tens of MeV or more when cosmic ray electrons interact with nuclei of sufficiently high atomic number (see gamma ray image of the Moon near the end of this article, for illustration).

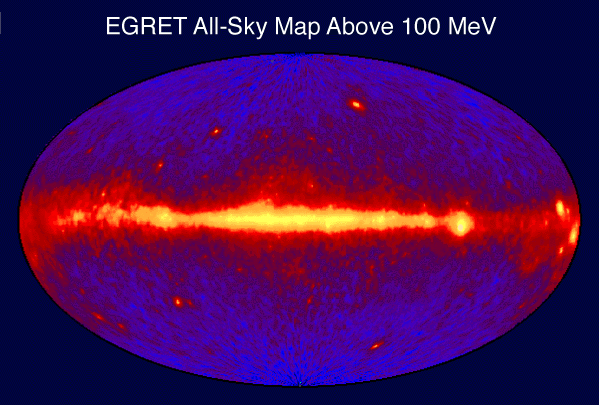
Pulsars and magnetars
The gamma ray sky (see illustration at right) is dominated by the more common and longer-term production of gamma rays that emanate from pulsars within the Milky Way. Sources from the rest of the sky are mostly quasars. Pulsars are thought to be neutron stars with magnetic fields that produce focused beams of radiation, and are far less energetic, more common, and much nearer sources (typically seen only in our own galaxy) than are quasars or the rarer gamma-ray burst sources of gamma rays. Pulsars have relatively long-lived magnetic fields that produce focused beams of relativistic speed charged particles, which emit gamma rays (bremsstrahlung) when those strike gas or dust in their nearby medium, and are decelerated. This is a similar mechanism to the production of high-energy photons inmegavoltage
Megavoltage X-rays are produced by linear accelerators ("linacs") operating at voltages in excess of 1000 kV (1 MV) range, and therefore have an energy in the MeV range. The voltage in this case refers to the voltage used to accelerat ...
radiation therapy
Radiation therapy or radiotherapy, often abbreviated RT, RTx, or XRT, is a therapy using ionizing radiation, generally provided as part of cancer treatment to control or kill malignant cells and normally delivered by a linear accelerator. Rad ...
machines (see bremsstrahlung
''Bremsstrahlung'' (), from "to brake" and "radiation"; i.e., "braking radiation" or "deceleration radiation", is electromagnetic radiation produced by the deceleration of a charged particle when deflected by another charged particle, typical ...
). Inverse Compton scattering
Compton scattering, discovered by Arthur Holly Compton, is the scattering of a high frequency photon after an interaction with a charged particle, usually an electron. If it results in a decrease in energy (increase in wavelength) of the photon ...
, in which charged particles (usually electrons) impart energy to low-energy photons boosting them to higher energy photons. Such impacts of photons on relativistic charged particle beams is another possible mechanism of gamma ray production. Neutron stars with a very high magnetic field ( magnetars), thought to produce astronomical soft gamma repeaters, are another relatively long-lived star-powered source of gamma radiation.
Quasars and active galaxies
More powerful gamma rays from very distant quasars and closer active galaxies are thought to have a gamma ray production source similar to a particle accelerator. High energy electrons produced by the quasar, and subjected to inverse Compton scattering, synchrotron radiation, or bremsstrahlung, are the likely source of the gamma rays from those objects. It is thought that a supermassive black hole at the center of such galaxies provides the power source that intermittently destroys stars and focuses the resulting charged particles into beams that emerge from their rotational poles. When those beams interact with gas, dust, and lower energy photons they produce X-rays and gamma rays. These sources are known to fluctuate with durations of a few weeks, suggesting their relatively small size (less than a few light-weeks across). Such sources of gamma and X-rays are the most commonly visible high intensity sources outside our galaxy. They shine not in bursts (see illustration), but relatively continuously when viewed with gamma ray telescopes. The power of a typical quasar is about 1040 watts, a small fraction of which is gamma radiation. Much of the rest is emitted as electromagnetic waves of all frequencies, including radio waves.
Gamma-ray bursts
The most intense sources of gamma rays, are also the most intense sources of any type of electromagnetic radiation presently known. They are the "long duration burst" sources of gamma rays in astronomy ("long" in this context, meaning a few tens of seconds), and they are rare compared with the sources discussed above. By contrast, "short" gamma-ray bursts of two seconds or less, which are not associated with supernovae, are thought to produce gamma rays during the collision of pairs of neutron stars, or a neutron star and a black hole. The so-called ''long-duration'' gamma-ray bursts produce a total energy output of about 1044 joules (as much energy as our Sun will produce in its entire life-time) but in a period of only 20 to 40 seconds. Gamma rays are approximately 50% of the total energy output. The leading hypotheses for the mechanism of production of these highest-known intensity beams of radiation, are inverse Compton scattering and synchrotron radiation from high-energy charged particles. These processes occur as relativistic charged particles leave the region of the event horizon of a newly formed black hole created during supernova explosion. The beam of particles moving at relativistic speeds are focused for a few tens of seconds by the magnetic field of the exploding hypernova. The fusion explosion of the hypernova drives the energetics of the process. If the narrowly directed beam happens to be pointed toward the Earth, it shines at gamma ray frequencies with such intensity, that it can be detected even at distances of up to 10 billion light years, which is close to the edge of the visible universe.Properties
Penetration of matter
alpha particle
Alpha particles, also called alpha rays or alpha radiation, consist of two protons and two neutrons bound together into a particle identical to a helium-4 nucleus. They are generally produced in the process of alpha decay, but may also be prod ...
s, which can be stopped by paper or skin, and beta particles, which can be shielded by thin aluminium. Gamma rays are best absorbed by materials with high atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of ever ...
s (''Z'') and high density, which contribute to the total stopping power. Because of this, a lead (high ''Z'') shield is 20–30% better as a gamma shield than an equal mass of another low-''Z'' shielding material, such as aluminium, concrete, water, or soil; lead's major advantage is not in lower weight, but rather its compactness due to its higher density. Protective clothing, goggles and respirators can protect from internal contact with or ingestion of alpha or beta emitting particles, but provide no protection from gamma radiation from external sources.
The higher the energy of the gamma rays, the thicker the shielding made from the same shielding material is required. Materials for shielding gamma rays are typically measured by the thickness required to reduce the intensity of the gamma rays by one half (the half value layer or HVL). For example, gamma rays that require 1 cm (0.4 inch) of lead
Lead is a chemical element with the symbol Pb (from the Latin ) and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cut, ...
to reduce their intensity by 50% will also have their intensity reduced in half by of granite
Granite () is a coarse-grained ( phaneritic) intrusive igneous rock composed mostly of quartz, alkali feldspar, and plagioclase. It forms from magma with a high content of silica and alkali metal oxides that slowly cools and solidifies un ...
rock, 6 cm (2.5 inches) of concrete
Concrete is a composite material composed of fine and coarse aggregate bonded together with a fluid cement (cement paste) that hardens (cures) over time. Concrete is the second-most-used substance in the world after water, and is the most wid ...
, or 9 cm (3.5 inches) of packed soil
Soil, also commonly referred to as earth or dirt, is a mixture of organic matter, minerals, gases, liquids, and organisms that together support life. Some scientific definitions distinguish ''dirt'' from ''soil'' by restricting the former ...
. However, the mass of this much concrete or soil is only 20–30% greater than that of lead with the same absorption capability. Depleted uranium is used for shielding in portable gamma ray sources, but here the savings in weight over lead are larger, In a nuclear power plant, shielding can be provided by steel and concrete in the pressure and particle containment vessel, while water provides a radiation shielding of fuel rods during storage or transport into the reactor core. The loss of water or removal of a "hot" fuel assembly into the air would result in much higher radiation levels than when kept under water.
Matter interaction
atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of ever ...
of the absorbing material, which is why very dense materials like lead are only modestly better shields, on a ''per weight'' basis, than are less dense materials.
* Pair production: This becomes possible with gamma energies exceeding 1.02 MeV, and becomes important as an absorption mechanism at energies over 5 MeV (see illustration at right, for lead). By interaction with the electric field of a nucleus, the energy of the incident photon is converted into the mass of an electron-positron pair. Any gamma energy in excess of the equivalent rest mass of the two particles (totaling at least 1.02 MeV) appears as the kinetic energy of the pair and in the recoil of the emitting nucleus. At the end of the positron's range, it combines with a free electron, and the two annihilate, and the entire mass of these two is then converted into two gamma photons of at least 0.51 MeV energy each (or higher according to the kinetic energy of the annihilated particles).
The secondary electrons (and/or positrons) produced in any of these three processes frequently have enough energy to produce much ionization themselves.
Additionally, gamma rays, particularly high energy ones, can interact with atomic nuclei resulting in ejection of particles in photodisintegration, or in some cases, even nuclear fission (photofission
Photofission is a process in which a nucleus, after absorbing a gamma ray, undergoes nuclear fission and splits into two or more fragments.
The reaction was discovered in 1940 by a small team of engineers and scientists operating the Westingho ...
).
Light interaction
High-energy (from 80 GeV to ~10TeV
TEV may refer to:
* Transient Earth Voltage: a term for voltages appearing on the metal work of switchgear due to internal partial discharges
* TeV, or teraelectronvolt or trillion electron volt, a measure of energy
* Total Enterprise Value, a ...
) gamma rays arriving from far-distant quasars are used to estimate the extragalactic background light in the universe: The highest-energy rays interact more readily with the background light photons and thus the density of the background light may be estimated by analyzing the incoming gamma ray spectra.
Gamma spectroscopy
Gamma spectroscopy is the study of the energetic transitions in atomic nuclei, which are generally associated with the absorption or emission of gamma rays. As in opticalspectroscopy
Spectroscopy is the field of study that measures and interprets the electromagnetic spectra that result from the interaction between electromagnetic radiation and matter as a function of the wavelength or frequency of the radiation. Matter ...
(see Franck–Condon effect) the absorption of gamma rays by a nucleus is especially likely (i.e., peaks in a "resonance") when the energy of the gamma ray is the same as that of an energy transition in the nucleus. In the case of gamma rays, such a resonance is seen in the technique of Mössbauer spectroscopy. In the Mössbauer effect
The Mössbauer effect, or recoilless nuclear resonance fluorescence, is a physical phenomenon discovered by Rudolf Mössbauer in 1958. It involves the resonant and recoil-free emission and absorption of gamma radiation by atomic nuclei bound in a ...
the narrow resonance absorption for nuclear gamma absorption can be successfully attained by physically immobilizing atomic nuclei in a crystal. The immobilization of nuclei at both ends of a gamma resonance interaction is required so that no gamma energy is lost to the kinetic energy of recoiling nuclei at either the emitting or absorbing end of a gamma transition. Such loss of energy causes gamma ray resonance absorption to fail. However, when emitted gamma rays carry essentially all of the energy of the atomic nuclear de-excitation that produces them, this energy is also sufficient to excite the same energy state in a second immobilized nucleus of the same type.
Applications
Container Security Initiative
The Container Security Initiative (CSI) a.k.a. the 24-Hour Rule was launched in 2002 by the U.S. Bureau of Customs and Border Protection (CBP), an agency of the Department of Homeland Security. Its purpose was to increase security for container ...
(CSI). These machines are advertised to be able to scan 30 containers per hour.
Gamma radiation is often used to kill living organisms, in a process called irradiation. Applications of this include the sterilization of medical equipment (as an alternative to autoclaves or chemical means), the removal of decay-causing bacteria
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were am ...
from many foods and the prevention of the sprouting of fruit and vegetables to maintain freshness and flavor.
Despite their cancer-causing properties, gamma rays are also used to treat some types of cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal b ...
, since the rays also kill cancer cells. In the procedure called gamma-knife surgery, multiple concentrated beams of gamma rays are directed to the growth in order to kill the cancerous cells. The beams are aimed from different angles to concentrate the radiation on the growth while minimizing damage to surrounding tissues.
Gamma rays are also used for diagnostic purposes in nuclear medicine
Nuclear medicine or nucleology is a medical specialty involving the application of radioactive substances in the diagnosis and treatment of disease. Nuclear imaging, in a sense, is " radiology done inside out" because it records radiation emi ...
in imaging techniques. A number of different gamma-emitting radioisotopes are used. For example, in a PET scan
Positron emission tomography (PET) is a functional imaging technique that uses radioactive substances known as radiotracers to visualize and measure changes in metabolic processes, and in other physiological activities including blood flo ...
a radiolabeled sugar called fluorodeoxyglucose emits positrons that are annihilated by electrons, producing pairs of gamma rays that highlight cancer as the cancer often has a higher metabolic rate than the surrounding tissues. The most common gamma emitter used in medical applications is the nuclear isomer technetium-99m
Technetium-99m (99mTc) is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medica ...
which emits gamma rays in the same energy range as diagnostic X-rays. When this radionuclide tracer is administered to a patient, a gamma camera
A gamma camera (γ-camera), also called a scintillation camera or Anger camera, is a device used to image gamma radiation emitting radioisotopes, a technique known as scintigraphy. The applications of scintigraphy include early drug development ...
can be used to form an image of the radioisotope's distribution by detecting the gamma radiation emitted (see also SPECT
Single-photon emission computed tomography (SPECT, or less commonly, SPET) is a nuclear medicine tomographic imaging technique using gamma rays. It is very similar to conventional nuclear medicine planar imaging using a gamma camera (that is ...
). Depending on which molecule has been labeled with the tracer, such techniques can be employed to diagnose a wide range of conditions (for example, the spread of cancer to the bones via bone scan
A bone scan or bone scintigraphy is a nuclear medicine imaging technique of the bone. It can help diagnose a number of bone conditions, including cancer of the bone or metastasis, location of bone inflammation and fractures (that may not be ...
).
Health effects
Gamma rays cause damage at a cellular level and are penetrating, causing diffuse damage throughout the body. However, they are less ionising than alpha or beta particles, which are less penetrating. Low levels of gamma rays cause astochastic
Stochastic (, ) refers to the property of being well described by a random probability distribution. Although stochasticity and randomness are distinct in that the former refers to a modeling approach and the latter refers to phenomena themselv ...
health risk, which for radiation dose assessment is defined as the ''probability'' of cancer induction and genetic damage. High doses produce deterministic
Determinism is a philosophical view, where all events are determined completely by previously existing causes. Deterministic theories throughout the history of philosophy have developed from diverse and sometimes overlapping motives and cons ...
effects, which is the ''severity'' of acute tissue damage that is certain to happen. These effects are compared to the physical quantity absorbed dose measured by the unit gray
Grey (more common in British English) or gray (more common in American English) is an intermediate color between black and white. It is a neutral or achromatic color, meaning literally that it is "without color", because it can be compose ...
(Gy).
Body response
When gamma radiation breaks DNA molecules, a cell may be able to repair the damaged genetic material, within limits. However, a study of Rothkamm and Lobrich has shown that this repair process works well after high-dose exposure but is much slower in the case of a low-dose exposure.Risk assessment
The natural outdoor exposure in the United Kingdom ranges from 0.1 to 0.5 µSv/h with significant increase around known nuclear and contaminated sites. Natural exposure to gamma rays is about 1 to 2 mSv per year, and the average total amount of radiation received in one year per inhabitant in the USA is 3.6 mSv. There is a small increase in the dose, due to naturally occurring gamma radiation, around small particles of high atomic number materials in the human body caused by the photoelectric effect. By comparison, the radiation dose from chestradiography
Radiography is an imaging technique using X-rays, gamma rays, or similar ionizing radiation and non-ionizing radiation to view the internal form of an object. Applications of radiography include medical radiography ("diagnostic" and "therapeu ...
(about 0.06 mSv) is a fraction of the annual naturally occurring background radiation dose. A chest CT delivers 5 to 8 mSv. A whole-body PET/CT scan can deliver 14 to 32 mSv depending on the protocol. The dose from fluoroscopy of the stomach is much higher, approximately 50 mSv (14 times the annual background).
An acute full-body equivalent single exposure dose of 1 Sv (1000 mSv) causes slight blood changes, but 2.0–3.5 Sv (2.0–3.5 Gy) causes very severe syndrome of nausea, hair loss, and hemorrhaging, and will cause death in a sizable number of cases—-about 10% to 35% without medical treatment. A dose of 5 Sv (5 Gy) is considered approximately the LD50 (lethal dose for 50% of exposed population) for an acute exposure to radiation even with standard medical treatment. A dose higher than 5 Sv (5 Gy) brings an increasing chance of death above 50%. Above 7.5–10 Sv (7.5–10 Gy) to the entire body, even extraordinary treatment, such as bone-marrow transplants, will not prevent the death of the individual exposed (see radiation poisoning). (Doses much larger than this may, however, be delivered to selected parts of the body in the course of radiation therapy
Radiation therapy or radiotherapy, often abbreviated RT, RTx, or XRT, is a therapy using ionizing radiation, generally provided as part of cancer treatment to control or kill malignant cells and normally delivered by a linear accelerator. Rad ...
.)
For low-dose exposure, for example among nuclear workers, who receive an average yearly radiation dose of 19 mSv, the risk of dying from cancer (excluding leukemia
Leukemia ( also spelled leukaemia and pronounced ) is a group of blood cancers that usually begin in the bone marrow and result in high numbers of abnormal blood cells. These blood cells are not fully developed and are called ''blasts'' or ...
) increases by 2 percent. For a dose of 100 mSv, the risk increase is 10 percent. By comparison, risk of dying from cancer was increased by 32 percent for the survivors of the atomic bombing of Hiroshima and Nagasaki
The United States detonated two atomic bombs over the Japanese cities of Hiroshima and Nagasaki on 6 and 9 August 1945, respectively. The two bombings killed between 129,000 and 226,000 people, most of whom were civilians, and remain the on ...
.
Units of measurement and exposure
The following table shows radiation quantities in SI and non-SI units: The measure of the ionizing effect of gamma and X-rays in dry air is called the exposure, for which a legacy unit, the röntgen was used from 1928. This has been replaced bykerma
Kerma was the capital city of the Kerma culture, which was located in present-day Sudan at least 5,500 years ago. Kerma is one of the largest archaeological sites in ancient Nubia. It has produced decades of extensive excavations and research, ...
, now mainly used for instrument calibration purposes but not for received dose effect. The effect of gamma and other ionizing radiation on living tissue is more closely related to the amount of energy
In physics, energy (from Ancient Greek: ἐνέργεια, ''enérgeia'', “activity”) is the quantitative property that is transferred to a body or to a physical system, recognizable in the performance of work and in the form of ...
deposited in tissue rather than the ionisation of air, and replacement radiometric units and quantities for radiation protection
Radiation protection, also known as radiological protection, is defined by the International Atomic Energy Agency (IAEA) as "The protection of people from harmful effects of exposure to ionizing radiation, and the means for achieving this". Expos ...
have been defined and developed from 1953 onwards. These are:
*The gray
Grey (more common in British English) or gray (more common in American English) is an intermediate color between black and white. It is a neutral or achromatic color, meaning literally that it is "without color", because it can be compose ...
(Gy), is the SI unit of absorbed dose, which is the amount of radiation energy deposited in the irradiated material. For gamma radiation this is numerically equivalent to equivalent dose measured by the sievert, which indicates the stochastic biological effect of low levels of radiation on human tissue. The radiation weighting conversion factor from absorbed dose to equivalent dose is 1 for gamma, whereas alpha particles have a factor of 20, reflecting their greater ionising effect on tissue.
*The rad is the deprecated CGS unit for absorbed dose and the rem
Rem or REM may refer to:
Music
* R.E.M., an American rock band
* ''R.E.M.'' (EP), by Green
* "R.E.M." (song), by Ariana Grande
Organizations
* La République En Marche!, a French centrist political party
* Reichserziehungsministerium, in Nazi ...
is the deprecated CGS unit of equivalent dose, used mainly in the USA.
Distinction from X-rays
wavelength
In physics, the wavelength is the spatial period of a periodic wave—the distance over which the wave's shape repeats.
It is the distance between consecutive corresponding points of the same phase on the wave, such as two adjacent crests, tr ...
than the radiation (gamma rays) emitted by radioactive
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is consi ...
nuclei. Older literature distinguished between X- and gamma radiation on the basis of wavelength, with radiation shorter than some arbitrary wavelength, such as 10−11 m, defined as gamma rays. Since the energy of photons is proportional to their frequency and inversely proportional to wavelength, this past distinction between X-rays and gamma rays can also be thought of in terms of its energy, with gamma rays considered to be higher energy electromagnetic radiation than are X-rays.
However, since current artificial sources are now able to duplicate any electromagnetic radiation that originates in the nucleus, as well as far higher energies, the wavelengths characteristic of radioactive gamma ray sources vs. other types now completely overlap. Thus, gamma rays are now usually distinguished by their origin: X-rays are emitted by definition by electrons outside the nucleus, while gamma rays are emitted by the nucleus. Exceptions to this convention occur in astronomy, where gamma decay is seen in the afterglow of certain supernovas, but radiation from high energy processes known to involve other radiation sources than radioactive decay is still classed as gamma radiation.
 For example, modern high-energy X-rays produced by
For example, modern high-energy X-rays produced by linear accelerators
A linear particle accelerator (often shortened to linac) is a type of particle accelerator that accelerates charged subatomic particles or ions to a high speed by subjecting them to a series of oscillating electric potentials along a linear b ...
for megavoltage
Megavoltage X-rays are produced by linear accelerators ("linacs") operating at voltages in excess of 1000 kV (1 MV) range, and therefore have an energy in the MeV range. The voltage in this case refers to the voltage used to accelerat ...
treatment in cancer often have higher energy (4 to 25 MeV) than do most classical gamma rays produced by nuclear gamma decay. One of the most common gamma ray emitting isotopes used in diagnostic nuclear medicine
Nuclear medicine or nucleology is a medical specialty involving the application of radioactive substances in the diagnosis and treatment of disease. Nuclear imaging, in a sense, is " radiology done inside out" because it records radiation emi ...
, technetium-99m
Technetium-99m (99mTc) is a metastable nuclear isomer of technetium-99 (itself an isotope of technetium), symbolized as 99mTc, that is used in tens of millions of medical diagnostic procedures annually, making it the most commonly used medica ...
, produces gamma radiation of the same energy (140 keV) as that produced by diagnostic X-ray machines, but of significantly lower energy than therapeutic photons from linear particle accelerators. In the medical community today, the convention that radiation produced by nuclear decay is the only type referred to as "gamma" radiation is still respected.
Due to this broad overlap in energy ranges, in physics the two types of electromagnetic radiation are now often defined by their origin: X-rays are emitted by electrons (either in orbitals outside of the nucleus, or while being accelerated to produce bremsstrahlung
''Bremsstrahlung'' (), from "to brake" and "radiation"; i.e., "braking radiation" or "deceleration radiation", is electromagnetic radiation produced by the deceleration of a charged particle when deflected by another charged particle, typical ...
-type radiation), while gamma rays are emitted by the nucleus or by means of other particle decays or annihilation events. There is no lower limit to the energy of photons produced by nuclear reactions, and thus ultraviolet
Ultraviolet (UV) is a form of electromagnetic radiation with wavelength from 10 nm (with a corresponding frequency around 30 PHz) to 400 nm (750 THz), shorter than that of visible light, but longer than X-rays. UV radiation ...
or lower energy photons produced by these processes would also be defined as "gamma rays". The only naming-convention that is still universally respected is the rule that electromagnetic radiation that is known to be of atomic nuclear origin is ''always'' referred to as "gamma rays", and never as X-rays. However, in physics and astronomy, the converse convention (that all gamma rays are considered to be of nuclear origin) is frequently violated.
In astronomy, higher energy gamma and X-rays are defined by energy, since the processes that produce them may be uncertain and photon energy, not origin, determines the required astronomical detectors needed. High-energy photons occur in nature that are known to be produced by processes other than nuclear decay but are still referred to as gamma radiation. An example is "gamma rays" from lightning discharges at 10 to 20 MeV, and known to be produced by the bremsstrahlung mechanism.
Another example is gamma-ray bursts, now known to be produced from processes too powerful to involve simple collections of atoms undergoing radioactive decay. This is part and parcel of the general realization that many gamma rays produced in astronomical processes result not from radioactive decay or particle annihilation, but rather in non-radioactive processes similar to X-rays. Although the gamma rays of astronomy often come from non-radioactive events, a few gamma rays in astronomy are specifically known to originate from gamma decay of nuclei (as demonstrated by their spectra and emission half life). A classic example is that of supernova SN 1987A
SN 1987A was a type II supernova in the Large Magellanic Cloud, a dwarf satellite galaxy of the Milky Way. It occurred approximately from Earth and was the closest observed supernova since Kepler's Supernova. 1987A's light reached Earth on ...
, which emits an "afterglow" of gamma-ray photons from the decay of newly made radioactive nickel-56
Naturally occurring nickel (28Ni) is composed of five stable isotopes; , , , and , with being the most abundant (68.077% natural abundance). 26 radioisotopes have been characterised with the most stable being with a half-life of 76,000 year ...
and cobalt-56
Naturally occurring cobalt (Co) consists of a single stable isotope, Co. Twenty-eight radioisotopes have been characterized; the most stable are Co with a half-life of 5.2714 years, Co (271.8 days), Co (77.27 days), and Co (70.86 days). All other ...
. Most gamma rays in astronomy, however, arise by other mechanisms.
See also
*Annihilation
In particle physics, annihilation is the process that occurs when a subatomic particle collides with its respective antiparticle to produce other particles, such as an electron colliding with a positron to produce two photons. The total ener ...
* Galactic Center GeV excess
* Gaseous ionization detectors
* Very-high-energy gamma ray
* Ultra-high-energy gamma ray
Ultra-high-energy gamma rays are gamma rays with photon energies higher than 100 TeV (0.1 PeV). They have a frequency higher than 2.42 × 1028 Hz and a wavelength shorter than 1.24 × 10−20 m. The existence of these rays was confirmed in 20 ...
Notes
References
External links
Basic reference on several types of radiation
Radiation Q & A
– Contains information on gamma-ray energies from isotopes.
with filter on gamma-ray energy
Health Physics Society Public Education Website
{{Authority control Electromagnetic spectrum IARC Group 1 carcinogens Nuclear physics Radiation Radioactivity Articles containing video clips