dihydrofolate reductase on:
[Wikipedia]
[Google]
[Amazon]
Dihydrofolate reductase, or DHFR, is an

 The mechanism of this enzyme is stepwise and steady-state random. Specifically, the catalytic reaction begins with the NADPH and the substrate attaching to the binding site of the enzyme, followed by the protonation and the hydride transfer from the cofactor NADPH to the substrate. However, two latter steps do not take place simultaneously in a same transition state. In a study using computational and experimental approaches, Liu ''et al'' conclude that the protonation step precedes the hydride transfer.
The mechanism of this enzyme is stepwise and steady-state random. Specifically, the catalytic reaction begins with the NADPH and the substrate attaching to the binding site of the enzyme, followed by the protonation and the hydride transfer from the cofactor NADPH to the substrate. However, two latter steps do not take place simultaneously in a same transition state. In a study using computational and experimental approaches, Liu ''et al'' conclude that the protonation step precedes the hydride transfer.
 DHFR's enzymatic mechanism is shown to be pH dependent, particularly the hydride transfer step, since pH changes are shown to have remarkable influence on the electrostatics of the active site and the ionization state of its residues. The acidity of the targeted nitrogen on the substrate is important in the binding of the substrate to the enzyme's binding site which is proved to be hydrophobic even though it has direct contact to water. Asp27 is the only charged hydrophilic residue in the binding site, and neutralization of the charge on Asp27 may alter the pKa of the enzyme. Asp27 plays a critical role in the catalytic mechanism by helping with protonation of the substrate and restraining the substrate in the conformation favorable for the hydride transfer. The protonation step is shown to be associated with enol tautomerization even though this conversion is not considered favorable for the proton donation. A water molecule is proved to be involved in the protonation step. Entry of the water molecule to the active site of the enzyme is facilitated by the Met20 loop.
DHFR's enzymatic mechanism is shown to be pH dependent, particularly the hydride transfer step, since pH changes are shown to have remarkable influence on the electrostatics of the active site and the ionization state of its residues. The acidity of the targeted nitrogen on the substrate is important in the binding of the substrate to the enzyme's binding site which is proved to be hydrophobic even though it has direct contact to water. Asp27 is the only charged hydrophilic residue in the binding site, and neutralization of the charge on Asp27 may alter the pKa of the enzyme. Asp27 plays a critical role in the catalytic mechanism by helping with protonation of the substrate and restraining the substrate in the conformation favorable for the hydride transfer. The protonation step is shown to be associated with enol tautomerization even though this conversion is not considered favorable for the proton donation. A water molecule is proved to be involved in the protonation step. Entry of the water molecule to the active site of the enzyme is facilitated by the Met20 loop.


 Dihydrofolate reductase from Bacillus anthracis (BaDHFR) a validated drug target in the treatment of the infectious disease, anthrax. BaDHFR is less sensitive to trimethoprim analogs than is dihydrofolate reductase from other species such as ''
Dihydrofolate reductase from Bacillus anthracis (BaDHFR) a validated drug target in the treatment of the infectious disease, anthrax. BaDHFR is less sensitive to trimethoprim analogs than is dihydrofolate reductase from other species such as ''
1988 Nobel lecture in Medicine
Proteopedia: ''Dihydrofolate reductase''
* {{DEFAULTSORT:Dihydrofolate Reductase Protein domains EC 1.5.1 Enzymes of known structure
enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products ...
that reduces dihydrofolic acid to tetrahydrofolic acid, using NADPH as an electron donor, which can be converted to the kinds of tetrahydrofolate cofactors used in 1-carbon transfer chemistry. In humans, the DHFR enzyme is encoded by the ''DHFR'' gene
In biology, the word gene (from , ; "...Wilhelm Johannsen coined the word gene to describe the Mendelian units of heredity..." meaning ''generation'' or ''birth'' or ''gender'') can have several different meanings. The Mendelian gene is a b ...
.
It is found in the q11→q22 region of chromosome 5. Bacterial species
In biology, a species is the basic unit of classification and a taxonomic rank of an organism, as well as a unit of biodiversity. A species is often defined as the largest group of organisms in which any two individuals of the appropriat ...
possess distinct DHFR enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrates, and the enzyme converts the substrates into different molecules known as products ...
s (based on their pattern of binding diaminoheterocyclic molecules), but mammalia
Mammals () are a group of vertebrate animals constituting the class Mammalia (), characterized by the presence of mammary glands which in females produce milk for feeding (nursing) their young, a neocortex (a region of the brain), fur ...
n DHFRs are highly similar.
Structure
A central eight-strandedbeta-pleated sheet
The beta sheet, (β-sheet) (also β-pleated sheet) is a common motif of the regular protein secondary structure. Beta sheets consist of beta strands (β-strands) connected laterally by at least two or three backbone hydrogen bonds, forming a gen ...
makes up the main feature of the polypeptide backbone folding of DHFR. Seven of these strands are parallel and the eighth runs antiparallel. Four alpha helices connect successive beta strands. Residues 9 – 24 are termed "Met20" or "loop 1" and, along with other loops, are part of the major subdomain that surround the active site. The active site is situated in the N-terminal half of the sequence, which includes a conserved Pro
Pro is an abbreviation meaning " professional".
Pro, PRO or variants thereof may also refer to:
People
* Miguel Pro (1891–1927), Mexican priest
* Pro Hart (1928–2006), Australian painter
* Mlungisi Mdluli (born 1980), South African retire ...
- Trp dipeptide; the tryptophan has been shown to be involved in the binding of substrate by the enzyme.
Function
Dihydrofolate reductase convertsdihydrofolate
Dihydrofolic acid (conjugate base dihydrofolate) (DHF) is a folic acid ( vitamin B9) derivative which is converted to tetrahydrofolic acid by dihydrofolate reductase. Since tetrahydrofolate is needed to make both purines and pyrimidines, which a ...
into tetrahydrofolate, a proton shuttle required for the de novo synthesis of purines, thymidylic acid, and certain amino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha ...
s. While the functional dihydrofolate reductase gene has been mapped to chromosome 5, multiple intronless processed pseudogenes or dihydrofolate reductase-like genes have been identified on separate chromosomes.
Found in all organisms, DHFR has a critical role in regulating the amount of tetrahydrofolate in the cell. Tetrahydrofolate and its derivatives are essential for purine
Purine is a heterocyclic aromatic organic compound that consists of two rings ( pyrimidine and imidazole) fused together. It is water-soluble. Purine also gives its name to the wider class of molecules, purines, which include substituted purines ...
and thymidylate synthesis, which are important for cell proliferation and cell growth. DHFR plays a central role in the synthesis of nucleic acid precursors, and it has been shown that mutant cells that completely lack DHFR require glycine, a purine, and thymidine to grow. DHFR has also been demonstrated as an enzyme involved in the salvage of tetrahydrobiopterin from dihydrobiopterin
Mechanism

General mechanism
DHFR catalyzes the transfer of a hydride from NADPH todihydrofolate
Dihydrofolic acid (conjugate base dihydrofolate) (DHF) is a folic acid ( vitamin B9) derivative which is converted to tetrahydrofolic acid by dihydrofolate reductase. Since tetrahydrofolate is needed to make both purines and pyrimidines, which a ...
with an accompanying protonation to produce tetrahydrofolate. In the end, dihydrofolate is reduced to tetrahydrofolate and NADPH is oxidized to NADP+. The high flexibility of Met20 and other loops near the active site play a role in promoting the release of the product, tetrahydrofolate. In particular the Met20 loop helps stabilize the nicotinamide ring of the NADPH to promote the transfer of the hydride from NADPH to dihydrofolate.
 The mechanism of this enzyme is stepwise and steady-state random. Specifically, the catalytic reaction begins with the NADPH and the substrate attaching to the binding site of the enzyme, followed by the protonation and the hydride transfer from the cofactor NADPH to the substrate. However, two latter steps do not take place simultaneously in a same transition state. In a study using computational and experimental approaches, Liu ''et al'' conclude that the protonation step precedes the hydride transfer.
The mechanism of this enzyme is stepwise and steady-state random. Specifically, the catalytic reaction begins with the NADPH and the substrate attaching to the binding site of the enzyme, followed by the protonation and the hydride transfer from the cofactor NADPH to the substrate. However, two latter steps do not take place simultaneously in a same transition state. In a study using computational and experimental approaches, Liu ''et al'' conclude that the protonation step precedes the hydride transfer.
 DHFR's enzymatic mechanism is shown to be pH dependent, particularly the hydride transfer step, since pH changes are shown to have remarkable influence on the electrostatics of the active site and the ionization state of its residues. The acidity of the targeted nitrogen on the substrate is important in the binding of the substrate to the enzyme's binding site which is proved to be hydrophobic even though it has direct contact to water. Asp27 is the only charged hydrophilic residue in the binding site, and neutralization of the charge on Asp27 may alter the pKa of the enzyme. Asp27 plays a critical role in the catalytic mechanism by helping with protonation of the substrate and restraining the substrate in the conformation favorable for the hydride transfer. The protonation step is shown to be associated with enol tautomerization even though this conversion is not considered favorable for the proton donation. A water molecule is proved to be involved in the protonation step. Entry of the water molecule to the active site of the enzyme is facilitated by the Met20 loop.
DHFR's enzymatic mechanism is shown to be pH dependent, particularly the hydride transfer step, since pH changes are shown to have remarkable influence on the electrostatics of the active site and the ionization state of its residues. The acidity of the targeted nitrogen on the substrate is important in the binding of the substrate to the enzyme's binding site which is proved to be hydrophobic even though it has direct contact to water. Asp27 is the only charged hydrophilic residue in the binding site, and neutralization of the charge on Asp27 may alter the pKa of the enzyme. Asp27 plays a critical role in the catalytic mechanism by helping with protonation of the substrate and restraining the substrate in the conformation favorable for the hydride transfer. The protonation step is shown to be associated with enol tautomerization even though this conversion is not considered favorable for the proton donation. A water molecule is proved to be involved in the protonation step. Entry of the water molecule to the active site of the enzyme is facilitated by the Met20 loop.
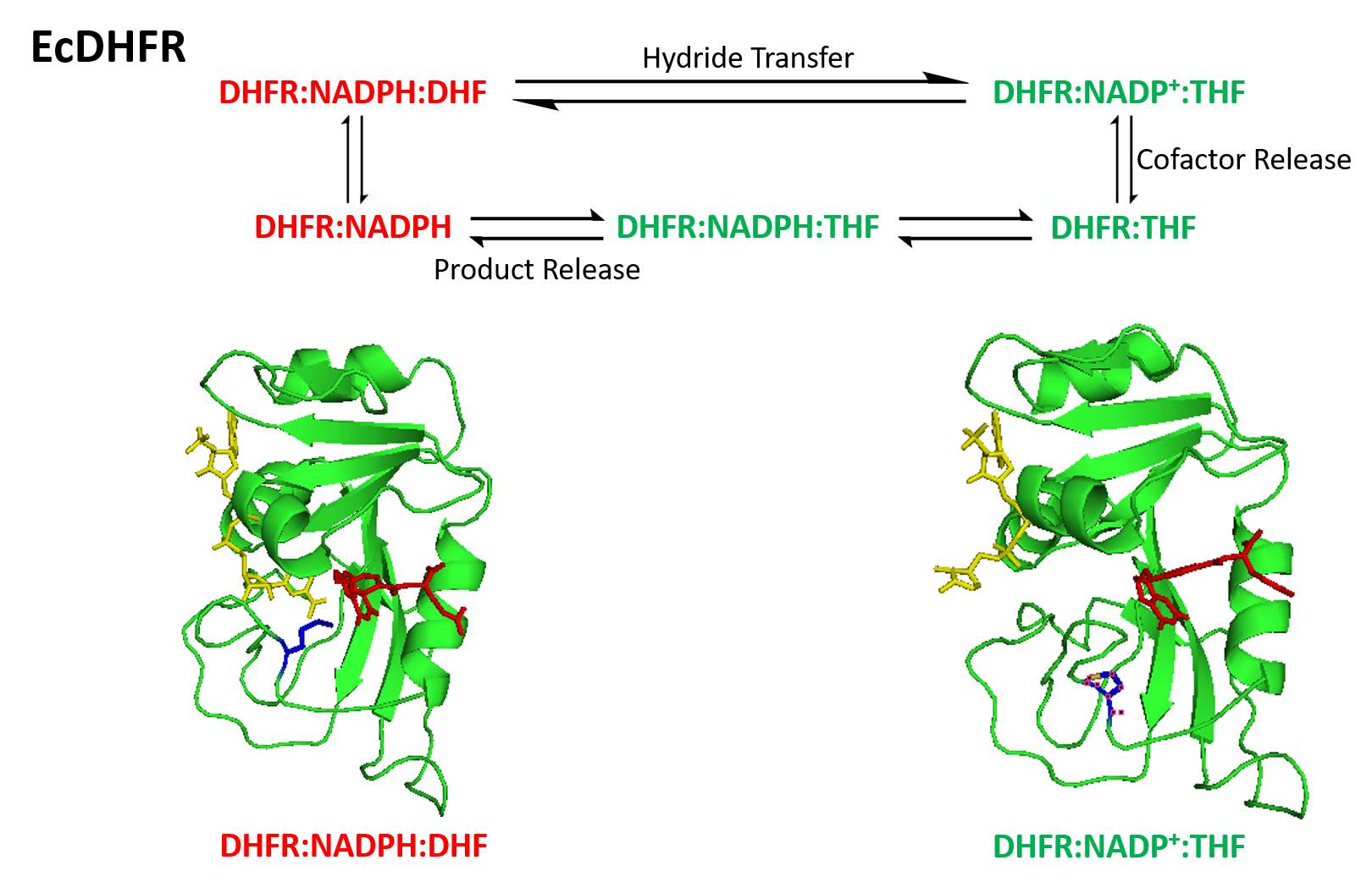
Conformational changes of DHFR
The catalytic cycle of the reaction catalyzed by DHFR incorporates five important intermediate: holoenzyme (E:NADPH), Michaelis complex (E:NADPH:DHF), ternary product complex (E:NADP+:THF), tetrahydrofolate binary complex (E:THF), and THF‚NADPH complex (E:NADPH:THF). The product (THF) dissociation step from E:NADPH:THF to E:NADPH is the rate determining step during steady-state turnover. Conformational changes are critical in DHFR's catalytic mechanism. The Met20 loop of DHFR is able to open, close or occlude the active site. Correspondingly, three different conformations classified as the opened, closed and occluded states are assigned to Met20. In addition, an extra distorted conformation of Met20 was defined due to its indistinct characterization results. The Met20 loop is observed in its occluded conformation in the three product ligating intermediates, where the nicotinamide ring is occluded from the active site. This conformational feature accounts for the fact that the substitution of NADP+ by NADPH is prior to product dissociation. Thus, the next round of reaction can occur upon the binding of substrate.

R67 DHFR
Due to its unique structure and catalytic features, R67 DHFR is widely studied. R67 DHFR is a type II R-plasmid-encoded DHFR without genetically and structurally relation to the E. coli chromosomal DHFR. It is a homotetramer that possesses the 222 symmetry with a single active site pore that is exposed to solvent ull .This symmetry of active site results in the different binding mode of the enzyme: It can bind with two dihydrofolate (DHF) molecules with positive cooperativity or two NADPH molecules with negative cooperativity, or one substrate plus one, but only the latter one has the catalytical activity. Compare with E. coli chromosomal DHFR, it has higher Km in binding dihydrofolate (DHF) and NADPH. The much lower catalytical kinetics show that hydride transfer is the rate determine step rather than product (THF) release. In the R67 DHFR structure, the homotetramer forms an active site pore. In the catalytical process, DHF and NADPH enters into the pore from opposite position. The π-π stacking interaction between NADPH's nicotinamide ring and DHF's pteridine ring tightly connect two reactants in the active site. However, the flexibility of p-aminobenzoylglutamate tail of DHF was observed upon binding which can promote the formation of the transition state.Clinical significance
Dihydrofolate reductase deficiency has been linked to megaloblastic anemia. Treatment is with reduced forms of folic acid. Because tetrahydrofolate, the product of this reaction, is the active form of folate in humans, inhibition of DHFR can cause functionalfolate deficiency
Folate deficiency, also known as vitamin B9 deficiency, is a low level of folate and derivatives in the body. Signs of folate deficiency are often subtle. A low number of red blood cells (anemia) is a late finding in folate deficiency and folat ...
. DHFR is an attractive pharmaceutical target for inhibition due to its pivotal role in DNA precursor synthesis. Trimethoprim, an antibiotic
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention ...
, inhibits bacterial DHFR while methotrexate, a chemotherapy
Chemotherapy (often abbreviated to chemo and sometimes CTX or CTx) is a type of cancer treatment that uses one or more anti-cancer drugs (chemotherapeutic agents or alkylating agents) as part of a standardized chemotherapy regimen. Chemother ...
agent, inhibits mammalian DHFR. However, resistance has developed against some drugs, as a result of mutational changes in DHFR itself.
DHFR mutations cause a rare autosomal recessive inborn error of folate metabolism that results in megaloblastic anemia, pancytopenia
Pancytopenia is a medical condition in which there is significant reduction in the number of almost all blood cells ( red blood cells, white blood cells, platelets, monocytes, lymphocytes, etc.).
If only two parameters from the complete blood ...
and severe cerebral folate deficiency which can be corrected by folinic acid supplementation .
Therapeutic applications
Since folate is needed by rapidly dividing cells to makethymine
Thymine () ( symbol T or Thy) is one of the four nucleobases in the nucleic acid of DNA that are represented by the letters G–C–A–T. The others are adenine, guanine, and cytosine. Thymine is also known as 5-methyluracil, a pyrimidin ...
, this effect may be used to therapeutic advantage.
DHFR can be targeted in the treatment of cancer and as a potential target against bacterial infections. DHFR is responsible for the levels of tetrahydrofolate in a cell, and the inhibition of DHFR can limit the growth and proliferation of cells that are characteristic of cancer and bacterial infections. Methotrexate, a competitive inhibitor of DHFR, is one such anticancer drug that inhibits DHFR. Other drugs include trimethoprim and pyrimethamine. These three are widely used as antitumor and antimicrobial agents. Other classes of compounds that target DHFR in general, and bacterial DHFRs in particular, belong to the classes such as diaminopteridines, diaminotriazines, diaminopyrroloquinazolines, stilbenes, chalcones, deoxybenzoins, to name but a few.
Trimethoprim has shown to have activity against a variety of Gram-positive bacterial pathogens. However, resistance to trimethoprim and other drugs aimed at DHFR can arise due to a variety of mechanisms, limiting the success of their therapeutical uses. Resistance can arise from DHFR gene amplification, mutations in DHFR, decrease in the uptake of the drugs, among others. Regardless, trimethoprim and sulfamethoxazole in combination has been used as an antibacterial agent for decades.
Folate is necessary for growth, and the pathway of the metabolism of folate is a target in developing treatments for cancer. DHFR is one such target. A regimen of fluorouracil
Fluorouracil (5-FU), sold under the brand name Adrucil among others, is a cytotoxic chemotherapy medication used to treat cancer. By intravenous injection it is used for treatment of colorectal cancer, oesophageal cancer, stomach cancer, panc ...
, doxorubicin, and methotrexate was shown to prolong survival in patients with advanced gastric cancer. Further studies into inhibitors of DHFR can lead to more ways to treat cancer.
Bacteria also need DHFR to grow and multiply and hence inhibitors selective for bacterial DHFR have found application as antibacterial agents.
Classes of small-molecules employed as inhibitors of dihydrofolate reductase include diaminoquinazoline & diaminopyrroloquinazoline, diaminopyrimidine,
diaminopteridine and diaminotriazines.
Potential anthrax treatment
 Dihydrofolate reductase from Bacillus anthracis (BaDHFR) a validated drug target in the treatment of the infectious disease, anthrax. BaDHFR is less sensitive to trimethoprim analogs than is dihydrofolate reductase from other species such as ''
Dihydrofolate reductase from Bacillus anthracis (BaDHFR) a validated drug target in the treatment of the infectious disease, anthrax. BaDHFR is less sensitive to trimethoprim analogs than is dihydrofolate reductase from other species such as ''Escherichia coli
''Escherichia coli'' (),Wells, J. C. (2000) Longman Pronunciation Dictionary. Harlow ngland Pearson Education Ltd. also known as ''E. coli'' (), is a Gram-negative, facultative anaerobic, rod-shaped, coliform bacterium of the genus '' Esc ...
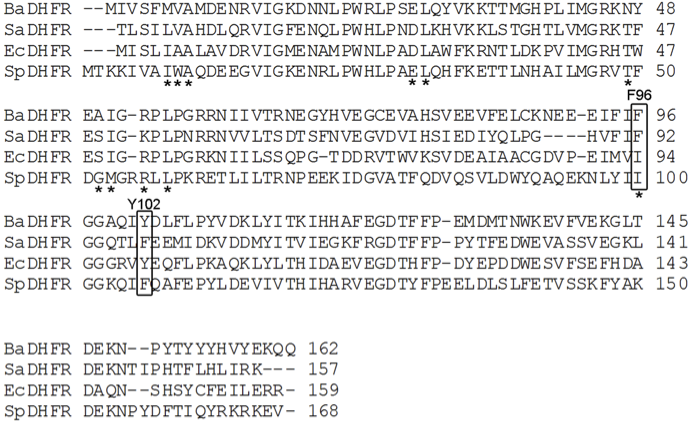
'', '' Staphylococcus aureus'', and '' Streptococcus pneumoniae''. A structural alignment of dihydrofolate reductase from all four species shows that only BaDHFR has the combination phenylalanine and tyrosine
-Tyrosine or tyrosine (symbol Tyr or Y) or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins. It is a non-essential amino acid with a polar side group. The word "tyrosine" is from the G ...
in positions 96 and 102, respectively.
BaDHFR's resistance to trimethoprim analogs is due to these two residues (F96 and Y102), which also confer improved kinetics and catalytic efficiency. Current research uses active site mutants in BaDHFR to guide lead optimization for new antifolate inhibitors.
As a research tool
DHFR has been used as a tool to detect protein–protein interactions in aprotein-fragment complementation assay Within the field of molecular biology, a protein-fragment complementation assay, or PCA, is a method for the identification and quantification of protein–protein interactions. In the PCA, the proteins of interest ("bait" and "prey") are each cov ...
(PCA).
CHO cells
DHFR lackingCHO cells
Chinese hamster ovary (CHO) cells are an epithelial cell line derived from the ovary of the Chinese hamster, often used in biological and medical research and commercially in the production of recombinant therapeutic proteins. They have found wi ...
are the most commonly used cell line for the production of recombinant proteins. These cells are transfected with a plasmid carrying the ''dhfr'' gene and the gene for the recombinant protein in a single expression system
Gene expression is the process by which information from a gene is used in the synthesis of a functional gene product that enables it to produce end products, protein or non-coding RNA, and ultimately affect a phenotype, as the final effect. The ...
, and then subjected to selective conditions in thymidine-lacking medium
Medium may refer to:
Science and technology
Aviation
* Medium bomber, a class of war plane
* Tecma Medium, a French hang glider design
Communication
* Media (communication), tools used to store and deliver information or data
* Medium ...
. Only the cells with the exogenous DHFR gene along with the gene of interest survive. Supplementation of this medium with methotrexate, a competitive inhibitor of DHFR, can further select for those cells expressing the highest levels of DHFR, and thus, select for the top recombinant protein producers.
Interactions
Dihydrofolate reductase has been shown to interact with GroEL and Mdm2.Interactive pathway map
References
Further reading
* * * * * * * * * * * * * * * * * * * * * *External links
1988 Nobel lecture in Medicine
Proteopedia: ''Dihydrofolate reductase''
* {{DEFAULTSORT:Dihydrofolate Reductase Protein domains EC 1.5.1 Enzymes of known structure