Cyanogen on:
[Wikipedia]
[Google]
[Amazon]
Cyanogen is the
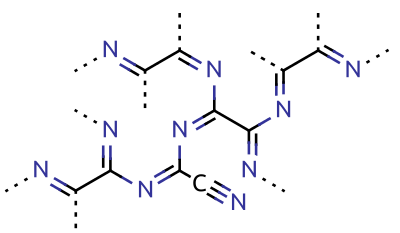 Paracyanogen is a polymer of cyanogen. It can be best prepared by heating mercuric cyanide. It can also be prepared by heating
Paracyanogen is a polymer of cyanogen. It can be best prepared by heating mercuric cyanide. It can also be prepared by heating
National Pollutant Inventory - Cyanide compounds fact sheet
{{Authority control Alkanedinitriles Pseudohalogens Blood agents Lachrymatory agents
chemical compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element ...
with the formula ( C N)2. It is a colorless and highly toxic
Toxicity is the degree to which a chemical substance or a particular mixture of substances can damage an organism. Toxicity can refer to the effect on a whole organism, such as an animal, bacterium, or plant, as well as the effect on a sub ...
gas with a pungent odor. The molecule is a pseudohalogen
Pseudohalogens are polyatomic analogues of halogens, whose chemistry, resembling that of the true halogens, allows them to substitute for halogens in several classes of chemical compounds. Pseudohalogens occur in pseudohalogen molecules, inorganic ...
. Cyanogen molecules consist of two CN groups – analogous to diatomic halogen molecules, such as Cl2, but far less oxidizing. The two cyano groups are bonded together at their carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon ma ...
atoms: N≡C‒ C≡N, although other isomers have been detected. The name is also used for the CN radical, and hence is used for compounds such as cyanogen bromide (NCBr) (but see also '' Cyano radical''.)
Cyanogen is the anhydride of oxamide
Oxamide is the organic compound with the formula . This white crystalline solid is soluble in ethanol, slightly soluble in water and insoluble in diethyl ether. Oxamide is the diamide derived from oxalic acid.
Preparation
Oxamide is produced fr ...
:
:H2NC(O)C(O)NH2 → NCCN + 2 H2O
although oxamide is manufactured from cyanogen by hydrolysis:
:NCCN + 2 H2O → H2NC(O)C(O)NH2
Preparation
Cyanogen is typically generated from cyanide compounds. One laboratory method entails thermal decomposition ofmercuric cyanide
Mercury(II) cyanide, also known as mercuric cyanide, is a compound of mercury. It is an odorless, toxic white powder. It is highly soluble in polar solvents such as water, alcohol, and ammonia; slightly soluble in ether; and insoluble in benzene ...
:
:2 Hg(CN)2 → (CN)2 + Hg2(CN)2
Alternatively, one can combine solutions of copper(II) salts (such as copper(II) sulfate
Copper(II) sulfate, also known as copper sulphate, is an inorganic compound with the chemical formula . It forms hydrates , where ''n'' can range from 1 to 7. The pentahydrate (''n'' = 5), a bright blue crystal, is the most commonly encountered h ...
) with cyanides; an unstable copper(II) cyanide is formed which rapidly decomposes into copper(I) cyanide and cyanogen.
:2 CuSO4 + 4 KCN → (CN)2 + 2 CuCN + 2 K2SO4
Industrially, it is created by the oxidation
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate change. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or ...
of hydrogen cyanide, usually using chlorine
Chlorine is a chemical element with the symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine i ...
over an activated silicon dioxide catalyst
Catalysis () is the process of increasing the rate of a chemical reaction by adding a substance known as a catalyst (). Catalysts are not consumed in the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recyc ...
or nitrogen dioxide over a copper
Copper is a chemical element with the symbol Cu (from la, cuprum) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pink ...
salt. It is also formed when nitrogen and acetylene are reacted by an electrical spark or discharge.
Isomers
Cyanogen is NCCN. There are less stable isomers in which the order of the atoms differs. Isocyanogen (or cyanoisocyanogen) is NCNC, diisocyanogen is CNNC, and diazodicarbon is CCNN.Paracyanogen
 Paracyanogen is a polymer of cyanogen. It can be best prepared by heating mercuric cyanide. It can also be prepared by heating
Paracyanogen is a polymer of cyanogen. It can be best prepared by heating mercuric cyanide. It can also be prepared by heating silver cyanide
Silver cyanide is the chemical compound with the formula AgCN. It is a white solid that precipitated upon treatment of solutions containing Ag+ with cyanide, which is used in some schemes to recover silver from solution. Silver cyanide is used in ...
, silver cyanate, cyanogen iodide or cyanuric iodide. It can also be prepared by the polymerization of cyanogen at in the presence of trace impurities. Paracyanogen can also be converted back to cyanogen by heating to . Based on experimental evidence, the structure of this polymeric material is thought to be rather irregular, with most of the carbon atoms being of sp2 type and localized domains of π conjugation.
History
Cyanogen was first synthesized in 1815 by Joseph Louis Gay-Lussac, who determined its empirical formula and named it. Gay-Lussac coined the word "cyanogène" from the Greek words κυανός (kyanos, blue) and γεννάω (gennao, I create), because cyanide was first isolated by the Swedish chemistCarl Wilhelm Scheele
Carl Wilhelm Scheele (, ; 9 December 1742 – 21 May 1786) was a Swedish German pharmaceutical chemist.
Scheele discovered oxygen (although Joseph Priestley published his findings first), and identified molybdenum, tungsten, barium, hydr ...
from the pigment " Prussian blue".
By the 1850s, cyanogen soap
Soap is a salt of a fatty acid used in a variety of cleansing and lubricating products. In a domestic setting, soaps are surfactants usually used for washing, bathing, and other types of housekeeping. In industrial settings, soaps are us ...
was used by photographers to remove silver
Silver is a chemical element with the symbol Ag (from the Latin ', derived from the Proto-Indo-European ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, white, lustrous transition metal, it exhibits the highest electrical ...
stains from their hands.
It attained importance with the growth of the fertilizer industry in the late 19th century and remains an important intermediate in the production of many fertilizers. It is also used as a stabilizer in the production of nitrocellulose.
In 1910 a spectroscopic analysis of Halley's Comet found cyanogen in the comet's tail, which led to public fear that the Earth would be poisoned as it passed through the tail. Because of the extremely diffuse nature of the tail, there was no effect when the planet passed through it.
Safety
Like othercyanide
Cyanide is a naturally occurring, rapidly acting, toxic chemical that can exist in many different forms.
In chemistry, a cyanide () is a chemical compound that contains a functional group. This group, known as the cyano group, consists of ...
s, cyanogen is very toxic, as it readily undergoes reduction to cyanide, which poisons the cytochrome c oxidase complex, thus interrupting the mitochondrial electron transfer chain. Cyanogen gas is an irritant to the eyes and respiratory system. Inhalation can lead to headache, dizziness, rapid pulse, nausea, vomiting, loss of consciousness, convulsions, and death, depending on exposure.
Lethal dose through inhalation typically ranges from .
Cyanogen produces the second-hottest-known natural flame (after carbon subnitride
Dicyanoacetylene, also called carbon subnitride or but-2-ynedinitrile (IUPAC), is a compound of carbon and nitrogen with chemical formula . It has a linear molecular structure, (often abbreviated as ), with alternating triple and single covalen ...
) with a temperature of over when it burns in oxygen.
In popular culture
In the '' Doctor Who'' serial " The Brain of Morbius" (the 5th serial of season 13), the Doctor synthesizes cyanogen using hydrogen cyanide as a starting material and vents it through a pipe to stop Solon from performing surgery on the brain of Morbius's body, however he completes it but shortly after dies of cyanogen poisoning. In '' Dragnet'' (1987) Friday (Dan Aykroyd) and Streebek (Tom Hanks) are tracking down the villain who stole "the pseudohalogenic compound cyanogen".See also
*Pseudohalogen
Pseudohalogens are polyatomic analogues of halogens, whose chemistry, resembling that of the true halogens, allows them to substitute for halogens in several classes of chemical compounds. Pseudohalogens occur in pseudohalogen molecules, inorganic ...
References
External links
National Pollutant Inventory - Cyanide compounds fact sheet
{{Authority control Alkanedinitriles Pseudohalogens Blood agents Lachrymatory agents