Control rod on:
[Wikipedia]
[Google]
[Amazon]
 Control rods are used in
Control rods are used in
 Control rods are inserted into the core of a nuclear reactor and adjusted in order to
Control rods are inserted into the core of a nuclear reactor and adjusted in order to
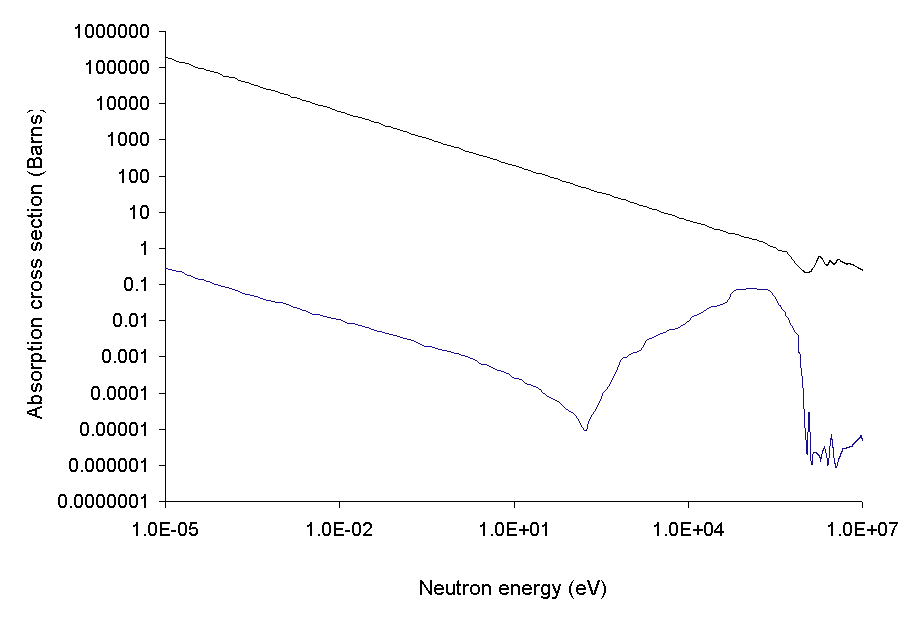 Chemical elements with usefully high neutron capture cross-sections include
Chemical elements with usefully high neutron capture cross-sections include
 Control rods are used in
Control rods are used in nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
s to control the rate of fission of the nuclear fuel – uranium
Uranium is a chemical element with the symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium is weak ...
or plutonium
Plutonium is a radioactive chemical element with the symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, and forms a dull coating when oxidized. The element normally exh ...
. Their compositions include chemical element
A chemical element is a species of atoms that have a given number of protons in their atomic nucleus, nuclei, including the pure Chemical substance, substance consisting only of that species. Unlike chemical compounds, chemical elements canno ...
s such as boron
Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the '' boron group'' it has t ...
, cadmium
Cadmium is a chemical element with the Symbol (chemistry), symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12 element, group 12, zinc and mercury (element), mercury. Li ...
, silver
Silver is a chemical element with the symbol Ag (from the Latin ', derived from the Proto-Indo-European ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, white, lustrous transition metal, it exhibits the highest electrical ...
, hafnium
Hafnium is a chemical element with the symbol Hf and atomic number 72. A lustrous, silvery gray, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirconium minerals. Its existence was predicted by Dmitri M ...
, or indium, that are capable of absorbing many neutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the atomic nucleus, nuclei of atoms. Since protons and ...
s without themselves decaying. These elements have different neutron capture
Neutron capture is a nuclear reaction in which an atomic nucleus and one or more neutrons collide and merge to form a heavier nucleus. Since neutrons have no electric charge, they can enter a nucleus more easily than positively charged protons ...
cross sections for neutrons of various energies. Boiling water reactors (BWR), pressurized water reactor
A pressurized water reactor (PWR) is a type of light-water nuclear reactor. PWRs constitute the large majority of the world's nuclear power plants (with notable exceptions being the UK, Japan and Canada). In a PWR, the primary coolant (water) i ...
s (PWR), and heavy-water reactor
A pressurized heavy-water reactor (PHWR) is a nuclear reactor that uses heavy water ( deuterium oxide D2O) as its coolant and neutron moderator. PHWRs frequently use natural uranium as fuel, but sometimes also use very low enriched uranium. ...
s (HWR) operate with thermal neutrons, while breeder reactors operate with fast neutrons. Each reactor design can use different control rod materials based on the energy spectrum of its neutrons. Control rods have been used in nuclear aircraft engines like Project Pluto as a method of control.
Operating principle
 Control rods are inserted into the core of a nuclear reactor and adjusted in order to
Control rods are inserted into the core of a nuclear reactor and adjusted in order to control
Control may refer to:
Basic meanings Economics and business
* Control (management), an element of management
* Control, an element of management accounting
* Comptroller (or controller), a senior financial officer in an organization
* Controlli ...
the rate of the nuclear chain reaction and, thereby, the thermal power output of the reactor, the rate of steam
Steam is a substance containing water in the gas phase, and sometimes also an aerosol of liquid water droplets, or air. This may occur due to evaporation or due to boiling, where heat is applied until water reaches the enthalpy of vaporizatio ...
production, and the electrical power
Electric power is the rate at which electrical energy is transferred by an electric circuit. The SI unit of power is the watt, one joule per second. Standard prefixes apply to watts as with other SI units: thousands, millions and billion ...
output of the power station.
The number of control rods inserted, and the distance to which they are inserted, strongly influence the ''reactivity'' of the reactor. When reactivity (as effective neutron multiplication factor) is above 1, the rate of the nuclear chain reaction increases exponentially over time. When reactivity is below 1, the rate of the reaction decreases exponentially over time. When all control rods are fully inserted, they keep reactivity barely above 0, which quickly slows a running reactor to a stop and keeps it stopped (in shutdown
Shutdown or shut down may refer to:
* Government shutdowns in the United States
* Shutdown (computing)
* Shutdown (economics)
* Shutdown (nuclear reactor)
Arts and entertainment Music
* "Shut Down" (The Beach Boys song), 1963
* ''Shut Down Volu ...
). If all control rods are fully removed, reactivity is significantly above 1, and the reactor quickly runs hotter and hotter, until some other factor (such as temperature reactivity feedback) slows the reaction rate. Maintaining a constant power output requires keeping the long-term average neutron multiplication factor close to 1.
A new reactor is assembled with its control rods fully inserted. Control rods are partially removed from the core to allow the nuclear chain reaction to start up and increase to the desired power level. Neutron flux can be measured, and is roughly proportional to reaction rate and power level. To increase power output, some control rods are pulled out a small distance for a while. To decrease power output, some control rods are pushed in a small distance for a while. Several other factors affect the reactivity; to compensate for them, an automatic control system adjusts the control rods small amounts in or out, as-needed in some reactors. Each control rod influences some part of the reactor more than others; calculated adjustments to fuel distribution can be made to maintain similar reaction rates and temperatures in different parts of the core.
Typical shutdown
Shutdown or shut down may refer to:
* Government shutdowns in the United States
* Shutdown (computing)
* Shutdown (economics)
* Shutdown (nuclear reactor)
Arts and entertainment Music
* "Shut Down" (The Beach Boys song), 1963
* ''Shut Down Volu ...
time for modern reactors such as the European Pressurized Reactor
The EPR is a third generation pressurised water reactor design. It has been designed and developed mainly by Framatome (part of Areva between 2001 and 2017) and Électricité de France (EDF) in France, and Siemens in Germany. In Europe this ...
or Advanced CANDU reactor is 2 seconds for 90% reduction, limited by decay heat.
Control rods are usually used in control rod assemblies (typically 20 rods for a commercial PWR assembly) and inserted into guide tubes within the fuel elements. Control rods often stand vertically within the core. In PWRs they are inserted from above, with the control rod drive mechanisms mounted on the reactor pressure vessel
A pressure vessel is a container designed to hold gases or liquids at a pressure substantially different from the ambient pressure.
Construction methods and materials may be chosen to suit the pressure application, and will depend on the size o ...
head. In BWRs, due to the necessity of a steam dryer above the core, this design requires insertion of the control rods from beneath.
Materials
 Chemical elements with usefully high neutron capture cross-sections include
Chemical elements with usefully high neutron capture cross-sections include silver
Silver is a chemical element with the symbol Ag (from the Latin ', derived from the Proto-Indo-European ''h₂erǵ'': "shiny" or "white") and atomic number 47. A soft, white, lustrous transition metal, it exhibits the highest electrical ...
, indium, and cadmium
Cadmium is a chemical element with the Symbol (chemistry), symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12 element, group 12, zinc and mercury (element), mercury. Li ...
. Other candidate elements include boron
Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the '' boron group'' it has t ...
, cobalt
Cobalt is a chemical element with the symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. The free element, p ...
, hafnium
Hafnium is a chemical element with the symbol Hf and atomic number 72. A lustrous, silvery gray, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirconium minerals. Its existence was predicted by Dmitri M ...
, samarium, europium
Europium is a chemical element with the symbol Eu and atomic number 63. Europium is the most reactive lanthanide by far, having to be stored under an inert fluid to protect it from atmospheric oxygen or moisture. Europium is also the softest lan ...
, gadolinium, terbium, dysprosium
Dysprosium is the chemical element with the symbol Dy and atomic number 66. It is a rare-earth element in the lanthanide series with a metallic silver luster. Dysprosium is never found in nature as a free element, though, like other lanthanide ...
, holmium, erbium
Erbium is a chemical element with the symbol Er and atomic number 68. A silvery-white solid metal when artificially isolated, natural erbium is always found in chemical combination with other elements. It is a lanthanide, a rare-earth element, ...
, thulium, ytterbium, and lutetium. Alloys or compounds may also be used, such as high- boron steel, silver-indium-cadmium alloy, boron carbide
Boron carbide (chemical formula approximately B4C) is an extremely hard boron– carbon ceramic, a covalent material used in tank armor, bulletproof vests, engine sabotage powders,
as well as numerous industrial applications. With a Vickers ...
, zirconium diboride, titanium diboride, hafnium diboride, gadolinium nitrate, gadolinium titanate, dysprosium titanate
Dysprosium titanate ( Dy2 Ti2 O7) is an inorganic compound, a ceramic of the titanate family, with pyrochlore structure.
Dysprosium titanate, like holmium titanate and holmium stannate, is a spin ice material. In 2009, quasiparticles resembli ...
, and boron carbide–europium hexaboride composite.
The material choice is influenced by the neutron energy in the reactor, their resistance to neutron-induced swelling, and the required mechanical and lifespan properties. The rods may have the form of tubes filled with neutron-absorbing pellets or powder. The tubes can be made of stainless steel or other "neutron window" materials such as zirconium, chromium, silicon carbide, or cubic (cubic boron nitride).
The burnup of "burnable poison
In applications such as nuclear reactors, a neutron poison (also called a neutron absorber or a nuclear poison) is a substance with a large neutron absorption cross-section. In such applications, absorbing neutrons is normally an undesirable eff ...
" isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers ( mass num ...
s also limits lifespan of a control rod. They may be reduced by using an element such as hafnium, a "non-burnable poison" which captures multiple neutrons before losing effectiveness, or by not using neutron absorbers for trimming. For example, in pebble bed reactors or in possible new type lithium-7-moderated and -cooled reactors that use fuel and absorber pebbles.
Some rare-earth elements are excellent neutron absorbers and are more common than silver (reserves of about 500,000t). For example, ytterbium (reserves about 1 M tons) and yttrium, 400 times more common, with middle capturing values, can be found and used together without separation inside minerals like xenotime
Xenotime is a rare-earth phosphate mineral, the major component of which is yttrium orthophosphate ( Y P O4). It forms a solid solution series with chernovite-(Y) ( Y As O4) and therefore may contain trace impurities of arsenic, as well as sili ...
(Yb) (Yb0.40Y0.27Lu0.12Er0.12Dy0.05Tm0.04Ho0.01)PO4, or keiviite (Yb) (Yb1.43Lu0.23Er0.17Tm0.08Y0.05Dy0.03Ho0.02)2Si2O7, lowering the cost. Xenon
Xenon is a chemical element with the symbol Xe and atomic number 54. It is a dense, colorless, odorless noble gas found in Earth's atmosphere in trace amounts. Although generally unreactive, it can undergo a few chemical reactions such as the ...
is also a strong neutron absorber as a gas, and can be used for controlling and (emergency) stopping helium
Helium (from el, ἥλιος, helios, lit=sun) is a chemical element with the symbol He and atomic number 2. It is a colorless, odorless, tasteless, non-toxic, inert, monatomic gas and the first in the noble gas group in the periodic ta ...
-cooled reactors, but does not function in cases of pressure loss, or as a burning protection gas together with argon
Argon is a chemical element with the symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third-most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice a ...
around the vessel part especially in case of core catching reactors or if filled with sodium or lithium. Fission-produced xenon can be used after waiting for caesium
Caesium (IUPAC spelling) (or cesium in American English) is a chemical element with the symbol Cs and atomic number 55. It is a soft, silvery-golden alkali metal with a melting point of , which makes it one of only five elemental metals that a ...
to precipitate, when practically no radioactivity is left. Cobalt-59 is also used as an absorber for winning of cobalt-60 for X-ray
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30&nb ...
production. Control rods can also be constructed as thick turnable rods with a tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isol ...
reflector and absorber side turned to stop by a spring in less than 1 second.
Silver-indium-cadmium alloys, generally 80% Ag, 15% In, and 5% Cd, are a common control rod material for pressurized water reactor
A pressurized water reactor (PWR) is a type of light-water nuclear reactor. PWRs constitute the large majority of the world's nuclear power plants (with notable exceptions being the UK, Japan and Canada). In a PWR, the primary coolant (water) i ...
s. The somewhat different energy absorption regions of the materials make the alloy an excellent neutron absorber. It has good mechanical strength and can be easily fabricated. It must be encased in stainless steel to prevent corrosion in hot water. Although indium is less rare than silver, it is more expensive.
Boron is another common neutron absorber. Due to the different cross sections of 10B and 11B, materials containing boron enriched in 10B by isotopic separation are frequently used. The wide absorption spectrum of boron also makes it suitable as a neutron shield. The mechanical properties of boron in its elementary form are unsuitable, and therefore alloys or compounds have to be used instead. Common choices are high-boron steel
Steel is an alloy made up of iron with added carbon to improve its strength and fracture resistance compared to other forms of iron. Many other elements may be present or added. Stainless steels that are corrosion- and oxidation-resistan ...
and boron carbide
Boron carbide (chemical formula approximately B4C) is an extremely hard boron– carbon ceramic, a covalent material used in tank armor, bulletproof vests, engine sabotage powders,
as well as numerous industrial applications. With a Vickers ...
. The latter is used as a control rod material in both PWRs and BWRs. 10B/11B separation is done commercially with gas centrifuges over BF3, but can also be done over BH3 from borane production or directly with an energy optimized melting centrifuge, using the heat of freshly separated boron for preheating.
Hafnium
Hafnium is a chemical element with the symbol Hf and atomic number 72. A lustrous, silvery gray, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirconium minerals. Its existence was predicted by Dmitri M ...
has excellent properties for reactors using water for both moderation and cooling. It has good mechanical strength, can be easily fabricated, and is resistant to corrosion
Corrosion is a natural process that converts a refined metal into a more chemically stable oxide. It is the gradual deterioration of materials (usually a metal) by chemical or electrochemical reaction with their environment. Corrosion engi ...
in hot water. Hafnium can be alloyed with other elements, e.g. with tin and oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements ...
to increase tensile and creep strength, with iron
Iron () is a chemical element with symbol Fe (from la, ferrum) and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, right in ...
, chromium
Chromium is a chemical element with the symbol Cr and atomic number 24. It is the first element in group 6. It is a steely-grey, lustrous, hard, and brittle transition metal.
Chromium metal is valued for its high corrosion resistance and hard ...
, and niobium for corrosion resistance, and with molybdenum
Molybdenum is a chemical element with the symbol Mo and atomic number 42 which is located in period 5 and group 6. The name is from Neo-Latin ''molybdaenum'', which is based on Ancient Greek ', meaning lead, since its ores were confused with lead ...
for wear resistance, hardness, and machineability. Such alloys are designated as Hafaloy, Hafaloy-M, Hafaloy-N, and Hafaloy-NM. The high cost and low availability of hafnium limit its use in civilian reactors, although it is used in some US Navy
The United States Navy (USN) is the maritime service branch of the United States Armed Forces and one of the eight uniformed services of the United States. It is the largest and most powerful navy in the world, with the estimated tonnage ...
reactors. Hafnium carbide can also be used as an insoluble material with a high melting point of 3890 °C and density higher than that of uranium dioxide for sinking, unmelted, through corium.
Dysprosium titanate
Dysprosium titanate ( Dy2 Ti2 O7) is an inorganic compound, a ceramic of the titanate family, with pyrochlore structure.
Dysprosium titanate, like holmium titanate and holmium stannate, is a spin ice material. In 2009, quasiparticles resembli ...
was undergoing evaluation for pressurized water control rods. Dysprosium titanate is a promising replacement for Ag-In-Cd alloys because it has a much higher melting point, does not tend to react with cladding materials, is easy to produce, does not produce radioactive waste, does not swell and does not outgas
Outgassing (sometimes called offgassing, particularly when in reference to indoor air quality) is the release of a gas that was dissolved, trapped, frozen, or absorbed in some material. Outgassing can include sublimation and evaporation (which ...
. It was developed in Russia and is recommended by some for VVER
The water-water energetic reactor (WWER), or VVER (from russian: водо-водяной энергетический реактор; transliterates as ; ''water-water power reactor'') is a series of pressurized water reactor designs originally de ...
and RBMK reactors. A disadvantage is less titanium and oxide absorption, that other neutron absorbing elements do not react with the already high-melting point cladding materials and that just using the unseparated content with dysprosium inside of minerals like Keiviit Yb inside chromium, SiC or c11B15N tubes deliver superior price and absorption without swelling and outgassing.
Hafnium diboride is another such material. It can be used alone or in a sintered mixture of hafnium and boron carbide powders.
Many other compounds of rare-earth elements can be used, such as samarium with boron-like europium
Europium is a chemical element with the symbol Eu and atomic number 63. Europium is the most reactive lanthanide by far, having to be stored under an inert fluid to protect it from atmospheric oxygen or moisture. Europium is also the softest lan ...
and samarium boride, which is already used in the colour industry. Less absorptive compounds of boron similar to titanium, but inexpensive, such as molybdenum
Molybdenum is a chemical element with the symbol Mo and atomic number 42 which is located in period 5 and group 6. The name is from Neo-Latin ''molybdaenum'', which is based on Ancient Greek ', meaning lead, since its ores were confused with lead ...
as Mo2B5. Since they all swell with boron, in practice other compounds are better, such as carbides, etc., or compounds with two or more neutron-absorbing elements together. It is important that tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isol ...
, and probably also other elements like tantalum
Tantalum is a chemical element with the symbol Ta and atomic number 73. Previously known as ''tantalium'', it is named after Tantalus, a villain in Greek mythology. Tantalum is a very hard, ductile, lustrous, blue-gray transition metal that ...
, have much the same high capture qualities as hafnium
Hafnium is a chemical element with the symbol Hf and atomic number 72. A lustrous, silvery gray, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirconium minerals. Its existence was predicted by Dmitri M ...
, but with the opposite effect. This is not explainable by neutron reflection alone. An obvious explanation is resonance gamma rays increasing the fission and breeding ratio versus causing more capture of uranium, etc. over metastable conditions like for isotope 235mU, which has a half-life of about 26 min.
Additional means of reactivity regulation
Other means of controlling reactivity include (for PWR) a soluble neutron absorber ( boric acid) added to the reactor coolant, allowing the complete extraction of the control rods during stationary power operation, ensuring an even power and flux distribution over the entire core. This chemical shim, along with the use of burnable neutron poisons within the fuel pellets, is used to assist regulation of the core's long term reactivity, while the control rods are used for rapid reactor power changes (e.g. shutdown and start up). Operators of BWRs use the coolant flow through the core to control reactivity by varying the speed of the reactor recirculation pumps (an increase in coolant flow through the core improves the removal of steam bubbles, thus increasing the density of the coolant/ moderator, increasing power).Safety
In most reactor designs, as a safety measure, control rods are attached to the lifting machinery byelectromagnet
An electromagnet is a type of magnet in which the magnetic field is produced by an electric current. Electromagnets usually consist of wire wound into a coil. A current through the wire creates a magnetic field which is concentrated in ...
s, rather than direct mechanical linkage. This means that in the event of power failure, or if manually invoked due to failure of the lifting machinery, the control rods fall automatically, under gravity, all the way into the pile to stop the reaction. A notable exception to this fail-safe mode of operation is the BWR, which requires hydraulic insertion in the event of an emergency shut-down, using water from a special tank under high pressure. Quickly shutting down a reactor in this way is called scram
A scram or SCRAM is an emergency shutdown of a nuclear reactor effected by immediately terminating the fission reaction. It is also the name that is given to the manually operated kill switch that initiates the shutdown. In commercial reacto ...
ming.
Criticality accident prevention
Mismanagement or control rod failure have often been blamed for nuclear accidents, including the SL-1 explosion and theChernobyl disaster
The Chernobyl disaster was a nuclear accident that occurred on 26 April 1986 at the No. 4 reactor in the Chernobyl Nuclear Power Plant, near the city of Pripyat in the north of the Ukrainian SSR in the Soviet Union. It is one of only two n ...
.
''Homogeneous'' neutron absorbers have often been used to manage criticality accidents which involve aqueous solutions of fissile metal
A metal (from ancient Greek, Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electrical resistivity and conductivity, e ...
s. In several such accidents, either borax
Borax is a salt (ionic compound), a hydrated borate of sodium, with chemical formula often written . It is a colorless crystalline solid, that dissolves in water to make a basic solution. It is commonly available in powder or granular form ...
(sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable ...
borate) or a cadmium compound has been added to the system. The cadmium can be added as a metal to nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available ni ...
solutions of fissile material; the corrosion of the cadmium in the acid will then generate cadmium nitrate
Nitrate is a polyatomic ion with the chemical formula . Salts containing this ion are called nitrates. Nitrates are common components of fertilizers and explosives. Almost all inorganic nitrates are soluble in water. An example of an insolu ...
''in situ''.
In carbon dioxide
Carbon dioxide ( chemical formula ) is a chemical compound made up of molecules that each have one carbon atom covalently double bonded to two oxygen atoms. It is found in the gas state at room temperature. In the air, carbon dioxide is t ...
-cooled reactors such as the AGR, if the solid control rods fail to arrest the nuclear reaction, nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at se ...
gas can be injected into the primary coolant cycle. This is because nitrogen has a larger absorption cross-section for neutrons than carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon ma ...
or oxygen
Oxygen is the chemical element with the symbol O and atomic number 8. It is a member of the chalcogen group in the periodic table, a highly reactive nonmetal, and an oxidizing agent that readily forms oxides with most elements ...
; hence, the core then becomes less reactive.
As the neutron energy increases, the neutron cross section of most isotopes decreases. The boron
Boron is a chemical element with the symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the '' boron group'' it has t ...
isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers ( mass num ...
10B is responsible for the majority of the neutron absorption. Boron-containing materials can also be used as neutron shielding, to reduce the activation
Activation, in chemistry and biology, is the process whereby something is prepared or excited for a subsequent reaction.
Chemistry
In chemistry, "activation" refers to the reversible transition of a molecule into a nearly identical chemical o ...
of material close to a reactor core.
See also
*Nuclear power
Nuclear power is the use of nuclear reactions to produce electricity. Nuclear power can be obtained from nuclear fission, nuclear decay and nuclear fusion reactions. Presently, the vast majority of electricity from nuclear power is produced b ...
*Nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
* Nuclear safety
*Wigner effect
The Wigner effect (named for its discoverer, Eugene Wigner), also known as the discomposition effect or Wigner's disease, is the displacement of atoms in a solid caused by neutron radiation.
Any solid can display the Wigner effect. The effect ...
References
External links
Further reading
* * * {{Authority control Alloys Nuclear power plant components Nuclear reactor safety Pressurized water reactors