camptothecin on:
[Wikipedia]
[Google]
[Amazon]
Camptothecin (CPT) is a topoisomerase inhibitor. It was discovered in 1966 by M. E. Wall and M. C. Wani in systematic screening of

 Studies have shown that
Studies have shown that

CPT is linked to a
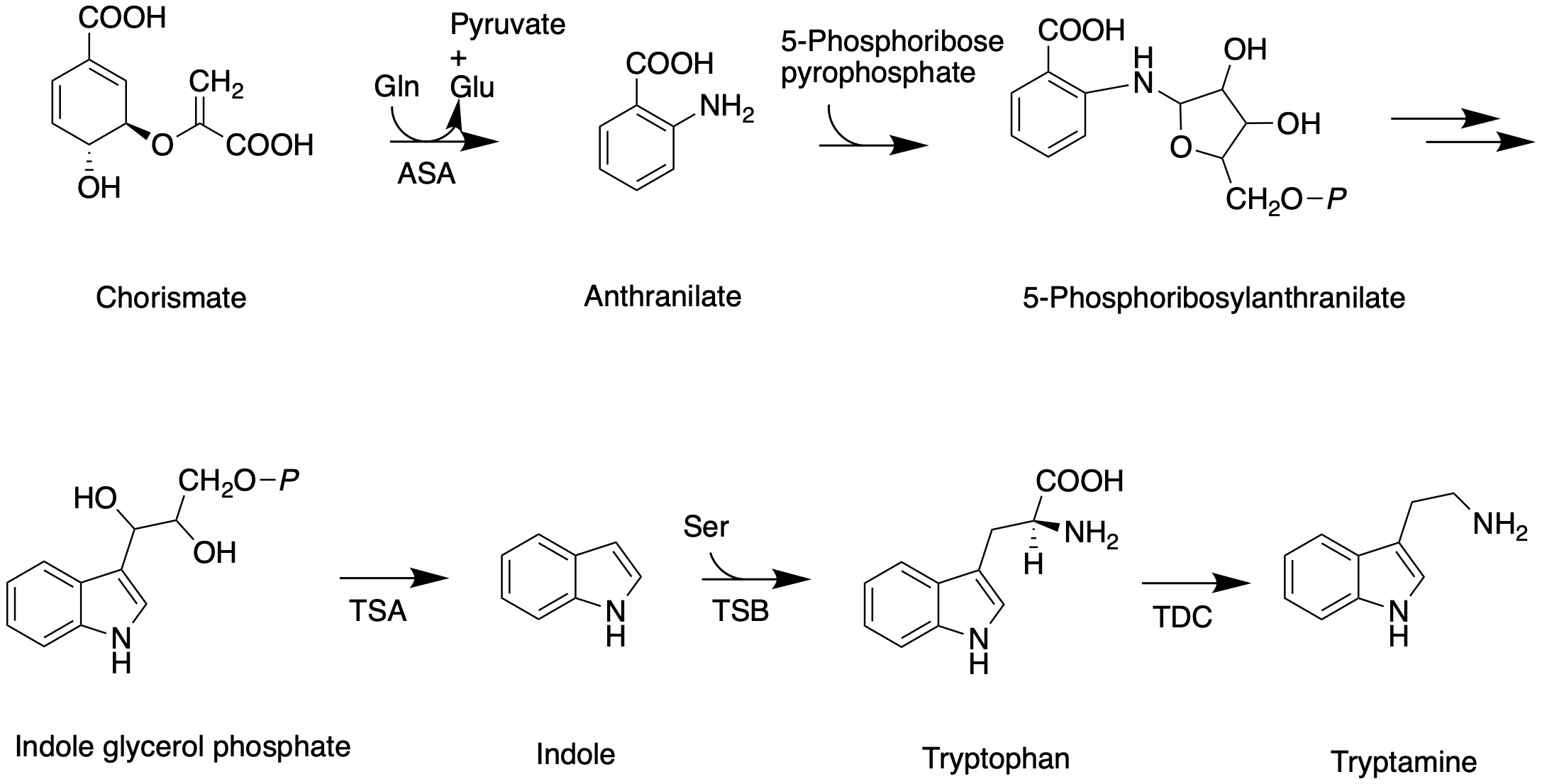

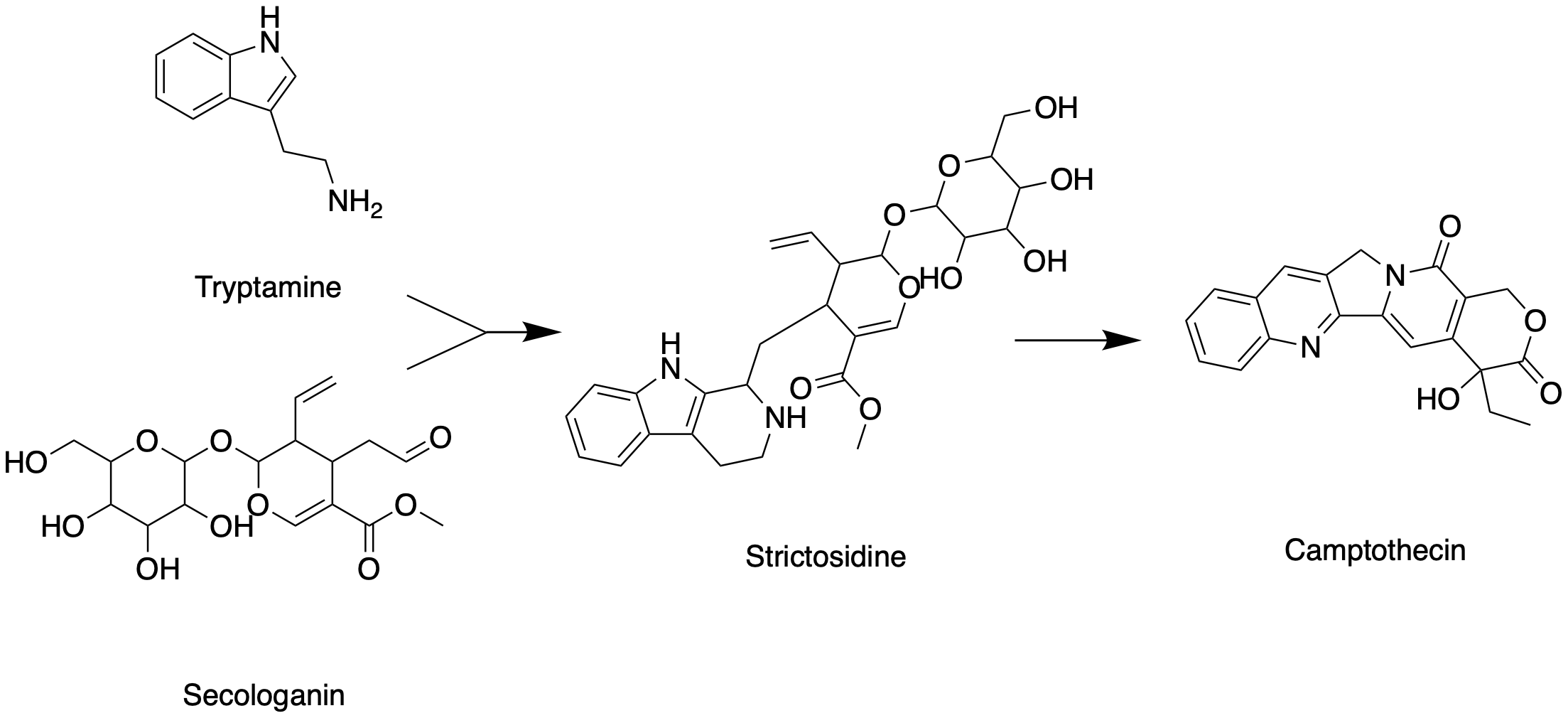 Like all other monoterpenoid indole-alkaloids, biosynthesis of camptothecin requires production of the strictosidine. Strictosidine is synthesized through condensation reaction between trypamine from shikimate pathway and secologanin from either mevalonate (MVA) pathway or non-mevalonate pathway (MEP). Strictosidine then undergoes intermolecular cyclization to produce strictosamide, which is converted to camptothecin through a series of oxidation reactions by enzymes that still needs to be resolved.
The shikimate pathway leading to biosynthesis of tryptamine is mostly understood. First, chorismate is converted to anthranilate by the alpha-subunit of anthranilate synthase (ASA). Anthranilate reacts with 5-phosphoribose pyrrophosphate to produce 5-phosphoribosylanthranilate. Then this intermediate is converted to indole glycerol phosphate, which interacts with the alpha-subunit of tryptophan (TSA) synthase to yield indole. The beta-subunit of tryptophan synthase (TSB) catalyzes condensation of indole with serine, leading to tryptophan. In the next step, tryptoamine is produced as the result of decarboxylation by tryptophan decarboxylase (TDC).
Secologanin synthesis begins with condensation reaction between pyruvate and D-Glyceraldehyde-3-phosphate catalyzed by 1-deoxy-D-xylulose-5-phosphate synthase (DXS) to produce 1-deoxy-D-xylulose-5-phosphate (DXP). The conversion of DXP to isopentenyl diphosphate (IPP), which is the common terpenoid biosynthesis precursor involves 1-deoxy-D-xylulose-5-phosphate reductoisomerase (DXR) and 1-hydroxy-2-methyl-2(E)-butenyl-4-diphosphate reductase (HDR).
The formation of IPP can be achieved by both MVA and MEP pathways. Condensation of IPP and dimethylallyl diphosphate (DMAPP) yields geranyl diphosphate (GPP). The geraniol synthase (GS) then converts GPP to geraniol. The conversion of geraniol to secologanin occurs through various enzymatic reactions. Based on studies with radioactive labelling and pathway specific inhibitors, MEP pathway is the primary source for secologanin.
Tryptamine from shikimate pathway and secologanin from MVA or MEP pathway are converted to strictosidine through a condensation reaction catalyzed by strictosidine synthase. Although it is not fully resolved, it has been postulated that camptothecin is produced from strictosidine via strictosamide, 3 (S)-pumiloside and 3 (S)-deoxypumiloside.
Like all other monoterpenoid indole-alkaloids, biosynthesis of camptothecin requires production of the strictosidine. Strictosidine is synthesized through condensation reaction between trypamine from shikimate pathway and secologanin from either mevalonate (MVA) pathway or non-mevalonate pathway (MEP). Strictosidine then undergoes intermolecular cyclization to produce strictosamide, which is converted to camptothecin through a series of oxidation reactions by enzymes that still needs to be resolved.
The shikimate pathway leading to biosynthesis of tryptamine is mostly understood. First, chorismate is converted to anthranilate by the alpha-subunit of anthranilate synthase (ASA). Anthranilate reacts with 5-phosphoribose pyrrophosphate to produce 5-phosphoribosylanthranilate. Then this intermediate is converted to indole glycerol phosphate, which interacts with the alpha-subunit of tryptophan (TSA) synthase to yield indole. The beta-subunit of tryptophan synthase (TSB) catalyzes condensation of indole with serine, leading to tryptophan. In the next step, tryptoamine is produced as the result of decarboxylation by tryptophan decarboxylase (TDC).
Secologanin synthesis begins with condensation reaction between pyruvate and D-Glyceraldehyde-3-phosphate catalyzed by 1-deoxy-D-xylulose-5-phosphate synthase (DXS) to produce 1-deoxy-D-xylulose-5-phosphate (DXP). The conversion of DXP to isopentenyl diphosphate (IPP), which is the common terpenoid biosynthesis precursor involves 1-deoxy-D-xylulose-5-phosphate reductoisomerase (DXR) and 1-hydroxy-2-methyl-2(E)-butenyl-4-diphosphate reductase (HDR).
The formation of IPP can be achieved by both MVA and MEP pathways. Condensation of IPP and dimethylallyl diphosphate (DMAPP) yields geranyl diphosphate (GPP). The geraniol synthase (GS) then converts GPP to geraniol. The conversion of geraniol to secologanin occurs through various enzymatic reactions. Based on studies with radioactive labelling and pathway specific inhibitors, MEP pathway is the primary source for secologanin.
Tryptamine from shikimate pathway and secologanin from MVA or MEP pathway are converted to strictosidine through a condensation reaction catalyzed by strictosidine synthase. Although it is not fully resolved, it has been postulated that camptothecin is produced from strictosidine via strictosamide, 3 (S)-pumiloside and 3 (S)-deoxypumiloside.
natural products
A natural product is a natural compound or substance produced by a living organism—that is, found in nature. In the broadest sense, natural products include any substance produced by life. Natural products can also be prepared by chemical syn ...
for anticancer drugs
A drug is any chemical substance that causes a change in an organism's physiology or psychology when consumed. Drugs are typically distinguished from food and substances that provide nutritional support. Consumption of drugs can be via inhalati ...
. It was isolated from the bark and stem
Stem or STEM may refer to:
Plant structures
* Plant stem, a plant's aboveground axis, made of vascular tissue, off which leaves and flowers hang
* Stipe (botany), a stalk to support some other structure
* Stipe (mycology), the stem of a mushro ...
of ''Camptotheca acuminata
''Camptotheca'' (happy tree, cancer tree, or tree of life) is a genus of medium-sized deciduous trees growing to tall, native to southern China and Tibet. The genus is usually included in the tupelo family Nyssaceae, but sometimes included (wit ...
'' (Camptotheca, Happy tree), a tree
In botany, a tree is a perennial plant with an elongated stem, or trunk, usually supporting branches and leaves. In some usages, the definition of a tree may be narrower, including only woody plants with secondary growth, plants that ar ...
native to China
China, officially the People's Republic of China (PRC), is a country in East Asia. It is the world's List of countries and dependencies by population, most populous country, with a Population of China, population exceeding 1.4 billion, slig ...
used in traditional Chinese medicine
Traditional Chinese medicine (TCM) is an alternative medical practice drawn from traditional medicine in China. It has been described as "fraught with pseudoscience", with the majority of its treatments having no logical mechanism of actio ...
. It has been used clinically more recently in China for the treatment of gastrointestinal tumors. CPT showed anticancer activity in preliminary clinical trials
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical or behavioral interventions, including new treatments (such as novel vaccines, drugs, dietar ...
, especially against breast, ovarian, colon, lung, and stomach cancers. However, it has low solubility
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution.
The extent of the solubil ...
and adverse effects have been reported when used therapeutically, so synthetic and medicinal chemists have developed numerous syntheses of camptothecin and various derivatives to increase the benefits of the chemical
A chemical substance is a form of matter having constant chemical composition and characteristic properties. Some references add that chemical substance cannot be separated into its constituent elements by physical separation methods, i.e., wit ...
, with good results. Four CPT analogues have been approved and are used in cancer
Cancer is a group of diseases involving abnormal cell growth with the potential to invade or spread to other parts of the body. These contrast with benign tumors, which do not spread. Possible signs and symptoms include a lump, abnormal bl ...
chemotherapy
Chemotherapy (often abbreviated to chemo and sometimes CTX or CTx) is a type of cancer treatment that uses one or more anti-cancer drugs ( chemotherapeutic agents or alkylating agents) as part of a standardized chemotherapy regimen. Chemother ...
today, topotecan
Topotecan, sold under the brand name Hycamtin among others, is a chemotherapeutic agent medication that is a topoisomerase inhibitor. It is a synthetic, water-soluble analog of the natural chemical compound camptothecin. It is used in the form ...
, irinotecan
Irinotecan, sold under the brand name Camptosar among others, is a medication used to treat colon cancer, and small cell lung cancer. For colon cancer it is used either alone or with fluorouracil. For small cell lung cancer it is used with cisp ...
, belotecan, and trastuzumab deruxtecan. Camptothecin has also been found in other plants including '' Chonemorpha fragrans''.
Structures
CPT has a planar pentacyclic ringstructure
A structure is an arrangement and organization of interrelated elements in a material object or system, or the object or system so organized. Material structures include man-made objects such as buildings and machines and natural objects such ...
, that includes a pyrrolo ,4-βquinoline moiety (rings A, B and C), conjugated pyridone moiety (ring D) and one chiral
Chirality is a property of asymmetry important in several branches of science. The word ''chirality'' is derived from the Greek (''kheir''), "hand", a familiar chiral object.
An object or a system is ''chiral'' if it is distinguishable from ...
center at position 20 within the alpha- hydroxy lactone
Lactones are cyclic carboxylic esters, containing a 1-oxacycloalkan-2-one structure (), or analogues having unsaturation or heteroatoms replacing one or more carbon atoms of the ring.
Lactones are formed by intramolecular esterification of the c ...
ring with (S) configuration (the E-ring). Its planar structure is thought to be one of the most important factors in topoisomerase inhibition.
Binding
CPT binds to the topoisomerase I and DNA complex (the covalent complex) resulting in a ternary complex, and thereby stabilizing it. This prevents DNA re-ligation and therefore causes DNA damage which results inapoptosis
Apoptosis (from grc, ἀπόπτωσις, apóptōsis, 'falling off') is a form of programmed cell death that occurs in multicellular organisms. Biochemical events lead to characteristic cell changes ( morphology) and death. These changes inclu ...
.
CPT binds both to the enzyme and DNA with hydrogen bonds
In chemistry, a hydrogen bond (or H-bond) is a primarily electrostatic force of attraction between a hydrogen (H) atom which is covalently bound to a more electronegative "donor" atom or group (Dn), and another electronegative atom bearing a ...
. The most important part of the structure is the E-ring which interacts from three different positions with the enzyme. The hydroxyl group in position 20 forms hydrogen bond to the side chain on aspartic acid
Aspartic acid (symbol Asp or D; the ionic form is known as aspartate), is an α-amino acid that is used in the biosynthesis of proteins. Like all other amino acids, it contains an amino group and a carboxylic acid. Its α-amino group is in the pro ...
number 533 (Asp533) in the enzyme. It is critical that the configuration
Configuration or configurations may refer to:
Computing
* Computer configuration or system configuration
* Configuration file, a software file used to configure the initial settings for a computer program
* Configurator, also known as choice board ...
of the chiral carbon
Carbon () is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—its atom making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table. Carbon makes u ...
is (S) because (R) is inactive. The lactone is bonded with two hydrogen bonds to the amino
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such ...
groups on arginine
Arginine is the amino acid with the formula (H2N)(HN)CN(H)(CH2)3CH(NH2)CO2H. The molecule features a guanidino group appended to a standard amino acid framework. At physiological pH, the carboxylic acid is deprotonated (−CO2−) and both the a ...
364 (Arg364).
The D-ring interacts with the +1 cytosine
Cytosine () (symbol C or Cyt) is one of the four nucleobases found in DNA and RNA, along with adenine, guanine, and thymine (uracil in RNA). It is a pyrimidine derivative, with a heterocyclic aromatic ring and two substituents attached (an a ...
on non-cleaved strand and stabilizes the topoisomerase I-DNA covalent complex by forming hydrogen bond. This hydrogen bond is between carbonyl
In organic chemistry, a carbonyl group is a functional group composed of a carbon atom double-bonded to an oxygen atom: C=O. It is common to several classes of organic compounds, as part of many larger functional groups. A compound containing a ...
group in position 17 on the D-ring and amino group on the pyrimidine
Pyrimidine (; ) is an aromatic, heterocyclic, organic compound similar to pyridine (). One of the three diazines (six-membered heterocyclics with two nitrogen atoms in the ring), it has nitrogen atoms at positions 1 and 3 in the ring. The othe ...
ring of +1 cytosine. CPT is selectively cytotoxic to the cells replicating DNA during S phase and its toxicity is primarily a result of conversion of single-strand breaks into double-strand breaks when the replication fork collides with the cleavage complexes formed by DNA and CPT.
Chemistry
The lactone ring in CPT is highly susceptible tohydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution, elimination, and solvation reactions in which water is the nucleophile.
Biological hydrolys ...
. The open ring form is inactive and it must therefore be closed to inhibit topoisomerase I. The closed form is favored in acidic condition, as it is in many cancer cells
Cell most often refers to:
* Cell (biology), the functional basic unit of life
Cell may also refer to:
Locations
* Monastic cell, a small room, hut, or cave in which a religious recluse lives, alternatively the small precursor of a monastery w ...
microenvironment.
CPT is transported into the cell by passive diffusion. Cellular uptake is favored by lipophilicity
Lipophilicity (from Greek λίπος "fat" and φίλος "friendly"), refers to the ability of a chemical compound to dissolve in fats, oils, lipids, and non-polar solvents such as hexane or toluene. Such non-polar solvents are themselves lipo ...
, which enhances intracellular
This glossary of biology terms is a list of definitions of fundamental terms and concepts used in biology, the study of life and of living organisms. It is intended as introductory material for novices; for more specific and technical definitions ...
accumulation.
Lipophilicity makes compounds more stable because of improved lactone partitioning into red blood cells
Red blood cells (RBCs), also referred to as red cells, red blood corpuscles (in humans or other animals not having nucleus in red blood cells), haematids, erythroid cells or erythrocytes (from Greek ''erythros'' for "red" and ''kytos'' for "holl ...
and consequently less hydrolysis of the lactone.
CPT has affinity for human serum albumin (HSA), especially the carboxylate form of CPT. Because of that, the equilibrium between the lactone ring and the carboxylate form is driven toward the carboxylate. Reduced drug-HSA interactions
Interaction is action that occurs between two or more objects, with broad use in philosophy and the sciences. It may refer to:
Science
* Interaction hypothesis, a theory of second language acquisition
* Interaction (statistics)
* Interactions o ...
could result in improved activity.
Structure-activity relationship
substitution
Substitution may refer to:
Arts and media
* Chord substitution, in music, swapping one chord for a related one within a chord progression
* Substitution (poetry), a variation in poetic scansion
* "Substitution" (song), a 2009 song by Silversun P ...
at position 7, 9, 10 and 11 can have positive effect on CPT activity and physical properties
A physical property is any property that is measurable, whose value describes a state of a physical system. The changes in the physical properties of a system can be used to describe its changes between momentary states. Physical properties are o ...
, e.g. potency and metabolic stability. Enlargement of the lactone ring by one unit also enhances its abilities, as in homocamptothecin. Substitution at position 12 and 14 leads to inactive derivative.
A- and B-ring modification
Alkyl substitution
Alkyl
In organic chemistry, an alkyl group is an alkane missing one hydrogen.
The term ''alkyl'' is intentionally unspecific to include many possible substitutions.
An acyclic alkyl has the general formula of . A cycloalkyl is derived from a cycloa ...
substitution at position 7 has shown increased cytotoxicity, such as ethyl (C2H5) or chloromethyl (CH2Cl). These groups are able to react with the DNA in the presence of topoisomerase I which leads to more tumor
A neoplasm () is a type of abnormal and excessive growth of tissue. The process that occurs to form or produce a neoplasm is called neoplasia. The growth of a neoplasm is uncoordinated with that of the normal surrounding tissue, and persists ...
activity. It has also been shown that increasing the length of the carbon chain (in position 7) leads to increased lipophilicity and consequently greater potency and stability in human plasma.
Other 7-modified CPT analogues are silatecans and karenitecins. They are potent inhibitors on topoisomerase I and both have alkylsilyl groups in position 7 which make them lipophilic and more stable. Silatecans or 7-silylcampthothecins have shown reduced drug-HSA interactions which contributes to its blood stability and they can also cross the blood brain barrier
Blood is a body fluid in the circulatory system of humans and other vertebrates that delivers necessary substances such as nutrients and oxygen to the cells, and transports metabolic waste products away from those same cells. Blood in the ...
. DB-67 is a 10-hydroxy derivative and is among the most active silatecans. BNP1350 which belongs to the series of karenitecins exhibits cytotoxic activity and ability to overcome drug resistance. Still another route to make CPT’s lipophilic is to introduce lipophilic substituents, such as iminomethyl or oxyiminomethyl moieties. One of the most potent compounds is the oxyiminomethyl derivative ST1481 that has the advantage to overcome drug resistance caused by transport systems.
Basic nitrogen
Nitrogen is the chemical element with the symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at sevent ...
in a carbon chain at position 7 makes the compound more hydrophilic
A hydrophile is a molecule or other molecular entity that is attracted to water molecules and tends to be dissolved by water.Liddell, H.G. & Scott, R. (1940). ''A Greek-English Lexicon'' Oxford: Clarendon Press.
In contrast, hydrophobes are no ...
and hence more water-soluble. For example, is a derivate called CKD-602, which is a potent topoisomerase I inhibitor and successfully overcomes the poor water solubility and toxicity seen with CPT.
Considerably greater activity can be achieved by putting electron-withdrawing groups like amino, nitro, bromo or chloro at position 9 and 10 and hydroxyl group at position 10 or 11. But these compounds are relatively insoluble in aqueous solutions, which causes difficulty in administrations. Methoxy group at both position 10 and 11 simultaneously leads to inactivity.
Hexacyclic CPT analogues
Hexacyclic CPT analogues have shown great potency. For example,methylenedioxy
Methylenedioxy is the term used in the field of chemistry, particularly in organic chemistry, for a functional group with the structural formula R-O-CH2-O-R' which is connected to the rest of a molecule by two chemical bonds. The methylenedio ...
or ethylenedioxy group connected between 10 and 11 form a 5 or 6 membered ring which leads to more water-soluble derivates and increased potency. Researches have shown that ethylenedioxy analogues are less potent than methylenedioxy. The reason is the unfavorable steric interactions of ethylenedioxy analogues with the enzyme.
Adding amino or chloro group at 9th position or chloromethyl group at 7th position to these 10, 11-methylenedioxy or ethylenedioxy analogues results in compounds with even greater cytotoxicity but weaker solubility in water. To yield 10, 11-methylenedioxy or ethylenedioxy analogues with good water solubility a good way is to introduce a water solubilising substituent at position 7. Lurtotecan meets those requirements; it’s a 10, 11-ethylenedioxy analogue with a 4-methylpiperazino-methylene at position 7 and has shown a great potency in clinical researches.
A ring can also be formed between position 7 and 9, like position 10 and 11. That gives new opportunities to make water-soluble derivatives These hexacyclic CPT become more active when electron-withdrawing groups are put in position 11 and methyl or amino groups at 10. Exatecan is an example of hexacyclic CPT that has a 6 membered ring over position 7 and 9, and is 10-methyl, 11-fluoro substituted It is water-soluble and more potent than topotecan.
C- and D-ring modification
The C- and D-rings have an essential role in the antitumor activity. Replacement in any position results in much less potent compound than parent compound in other cytotoxicity assay.E-ring modifications
The E-ring doesn’t allow many structural changes without losing CPT activity because it is necessary for binding to the active site of TOP I. One possible replacement is changing the hydroxyl group to Cl, F or Br because theirpolarizability
Polarizability usually refers to the tendency of matter, when subjected to an electric field, to acquire an electric dipole moment in proportion to that applied field. It is a property of all matter, considering that matter is made up of elementa ...
is sufficient to stabilize the enzyme-complex.
Another possible modification is to insert a methylene between hydroxyl and lactone on the E-ring yielding a seven membered β-hydroxylactone group, so-called homocamptothecin (hCPT). The hCPT’s hydroxyl has less inductive effect
In chemistry, the inductive effect in a molecule is a local change in the electron density due to electron-withdrawing or electron-donating groups elsewhere in the molecule, resulting in a permanent dipole in a bond.
It is present in a σ (si ...
on the carboxyl group which makes the lactone very reactive. This enhances the interaction of the free hydroxyl group optimally with topoisomerase I and the covalent complex that forms in its presence are more stable. The E-ring of hCPT opens more slowly and the opening is irreversible. hCPTs exhibit enhanced human plasma stability because of decreased protein binding and more affinity for red blood cells than CPT.
CPT analogues
Since the discovery of CPT many analogues have been synthesized. Below is a schematic view of the CPT analogues that have been mentioned in the text above.cyclodextrin
Cyclodextrins are a family of cyclic oligosaccharides, consisting of a macrocyclic ring of glucose subunits joined by α-1,4 glycosidic bonds. Cyclodextrins are produced from starch by enzymatic conversion. They are used in food, pharmaceutical, ...
-based polymer to form the investigational anti-cancer drug CRLX101.
Biosynthesis


 Like all other monoterpenoid indole-alkaloids, biosynthesis of camptothecin requires production of the strictosidine. Strictosidine is synthesized through condensation reaction between trypamine from shikimate pathway and secologanin from either mevalonate (MVA) pathway or non-mevalonate pathway (MEP). Strictosidine then undergoes intermolecular cyclization to produce strictosamide, which is converted to camptothecin through a series of oxidation reactions by enzymes that still needs to be resolved.
The shikimate pathway leading to biosynthesis of tryptamine is mostly understood. First, chorismate is converted to anthranilate by the alpha-subunit of anthranilate synthase (ASA). Anthranilate reacts with 5-phosphoribose pyrrophosphate to produce 5-phosphoribosylanthranilate. Then this intermediate is converted to indole glycerol phosphate, which interacts with the alpha-subunit of tryptophan (TSA) synthase to yield indole. The beta-subunit of tryptophan synthase (TSB) catalyzes condensation of indole with serine, leading to tryptophan. In the next step, tryptoamine is produced as the result of decarboxylation by tryptophan decarboxylase (TDC).
Secologanin synthesis begins with condensation reaction between pyruvate and D-Glyceraldehyde-3-phosphate catalyzed by 1-deoxy-D-xylulose-5-phosphate synthase (DXS) to produce 1-deoxy-D-xylulose-5-phosphate (DXP). The conversion of DXP to isopentenyl diphosphate (IPP), which is the common terpenoid biosynthesis precursor involves 1-deoxy-D-xylulose-5-phosphate reductoisomerase (DXR) and 1-hydroxy-2-methyl-2(E)-butenyl-4-diphosphate reductase (HDR).
The formation of IPP can be achieved by both MVA and MEP pathways. Condensation of IPP and dimethylallyl diphosphate (DMAPP) yields geranyl diphosphate (GPP). The geraniol synthase (GS) then converts GPP to geraniol. The conversion of geraniol to secologanin occurs through various enzymatic reactions. Based on studies with radioactive labelling and pathway specific inhibitors, MEP pathway is the primary source for secologanin.
Tryptamine from shikimate pathway and secologanin from MVA or MEP pathway are converted to strictosidine through a condensation reaction catalyzed by strictosidine synthase. Although it is not fully resolved, it has been postulated that camptothecin is produced from strictosidine via strictosamide, 3 (S)-pumiloside and 3 (S)-deoxypumiloside.
Like all other monoterpenoid indole-alkaloids, biosynthesis of camptothecin requires production of the strictosidine. Strictosidine is synthesized through condensation reaction between trypamine from shikimate pathway and secologanin from either mevalonate (MVA) pathway or non-mevalonate pathway (MEP). Strictosidine then undergoes intermolecular cyclization to produce strictosamide, which is converted to camptothecin through a series of oxidation reactions by enzymes that still needs to be resolved.
The shikimate pathway leading to biosynthesis of tryptamine is mostly understood. First, chorismate is converted to anthranilate by the alpha-subunit of anthranilate synthase (ASA). Anthranilate reacts with 5-phosphoribose pyrrophosphate to produce 5-phosphoribosylanthranilate. Then this intermediate is converted to indole glycerol phosphate, which interacts with the alpha-subunit of tryptophan (TSA) synthase to yield indole. The beta-subunit of tryptophan synthase (TSB) catalyzes condensation of indole with serine, leading to tryptophan. In the next step, tryptoamine is produced as the result of decarboxylation by tryptophan decarboxylase (TDC).
Secologanin synthesis begins with condensation reaction between pyruvate and D-Glyceraldehyde-3-phosphate catalyzed by 1-deoxy-D-xylulose-5-phosphate synthase (DXS) to produce 1-deoxy-D-xylulose-5-phosphate (DXP). The conversion of DXP to isopentenyl diphosphate (IPP), which is the common terpenoid biosynthesis precursor involves 1-deoxy-D-xylulose-5-phosphate reductoisomerase (DXR) and 1-hydroxy-2-methyl-2(E)-butenyl-4-diphosphate reductase (HDR).
The formation of IPP can be achieved by both MVA and MEP pathways. Condensation of IPP and dimethylallyl diphosphate (DMAPP) yields geranyl diphosphate (GPP). The geraniol synthase (GS) then converts GPP to geraniol. The conversion of geraniol to secologanin occurs through various enzymatic reactions. Based on studies with radioactive labelling and pathway specific inhibitors, MEP pathway is the primary source for secologanin.
Tryptamine from shikimate pathway and secologanin from MVA or MEP pathway are converted to strictosidine through a condensation reaction catalyzed by strictosidine synthase. Although it is not fully resolved, it has been postulated that camptothecin is produced from strictosidine via strictosamide, 3 (S)-pumiloside and 3 (S)-deoxypumiloside.
References
{{Chemotherapeutic agents Topoisomerase inhibitors Quinoline alkaloids Tertiary alcohols Delta-lactones Delta-lactams Pyranoindolizinoquinolines Enones Conjugated dienes Plant toxins