Americium on:
[Wikipedia]
[Google]
[Amazon]
Americium is a synthetic
 Although americium was likely produced in previous nuclear experiments, it was first intentionally synthesized, isolated and identified in late autumn 1944, at the
Although americium was likely produced in previous nuclear experiments, it was first intentionally synthesized, isolated and identified in late autumn 1944, at the
 The longest-lived and most common isotopes of americium, 241Am and 243Am, have half-lives of 432.2 and 7,370 years, respectively. Therefore, any
The longest-lived and most common isotopes of americium, 241Am and 243Am, have half-lives of 432.2 and 7,370 years, respectively. Therefore, any
, Los Alamos National Laboratory, Retrieved 28 November 2010 Americium is produced mostly artificially in small quantities, for research purposes. A tonne of spent nuclear fuel contains about 100 grams of various americium isotopes, mostly 241Am and 243Am. Their prolonged radioactivity is undesirable for the disposal, and therefore americium, together with other long-lived actinides, must be neutralized. The associated procedure may involve several steps, where americium is first separated and then converted by neutron bombardment in special reactors to short-lived nuclides. This procedure is well known as nuclear transmutation, but it is still being developed for americium. The transuranic elements from americium to fermium occurred naturally in the
 Americium has been produced in small quantities in
Americium has been produced in small quantities in
, World Nuclear Association, January 2009, Retrieved 28 November 2010 The heavier isotope 243Am is produced in much smaller amounts; it is thus more difficult to separate, resulting in a higher cost of the order .Hammond C. R. "The elements" in Americium is not synthesized directly from uranium – the most common reactor material – but from the plutonium isotope 239Pu. The latter needs to be produced first, according to the following nuclear process: :^_U -> ce^_U -> beta^-23.5 \ \ce] ^_Np -> beta^-2.3565 \ \ce] ^_Pu
The capture of two neutrons by 239Pu (a so-called (n,γ) reaction), followed by a β-decay, results in 241Am:
: ^_Pu -> ce^_Pu -> beta^-14.35 \ \ce] ^_Am
The plutonium present in spent nuclear fuel contains about 12% of 241Pu. Because it beta-decays to 241Am, 241Pu can be extracted and may be used to generate further 241Am. However, this process is rather slow: half of the original amount of 241Pu decays to 241Am after about 15 years, and the 241Am amount reaches a maximum after 70 years.
The obtained 241Am can be used for generating heavier americium isotopes by further neutron capture inside a nuclear reactor. In a light water reactor (LWR), 79% of 241Am converts to 242Am and 10% to its nuclear isomer 242mAm:The "metastable" state is marked by the letter m.
:
Americium-242 has a half-life of only 16 hours, which makes its further conversion to 243Am extremely inefficient. The latter isotope is produced instead in a process where 239Pu captures four neutrons under high neutron flux:
: ^_Pu -> ce\ ^_Pu -> beta^-4.956 \ \ce] ^_Am
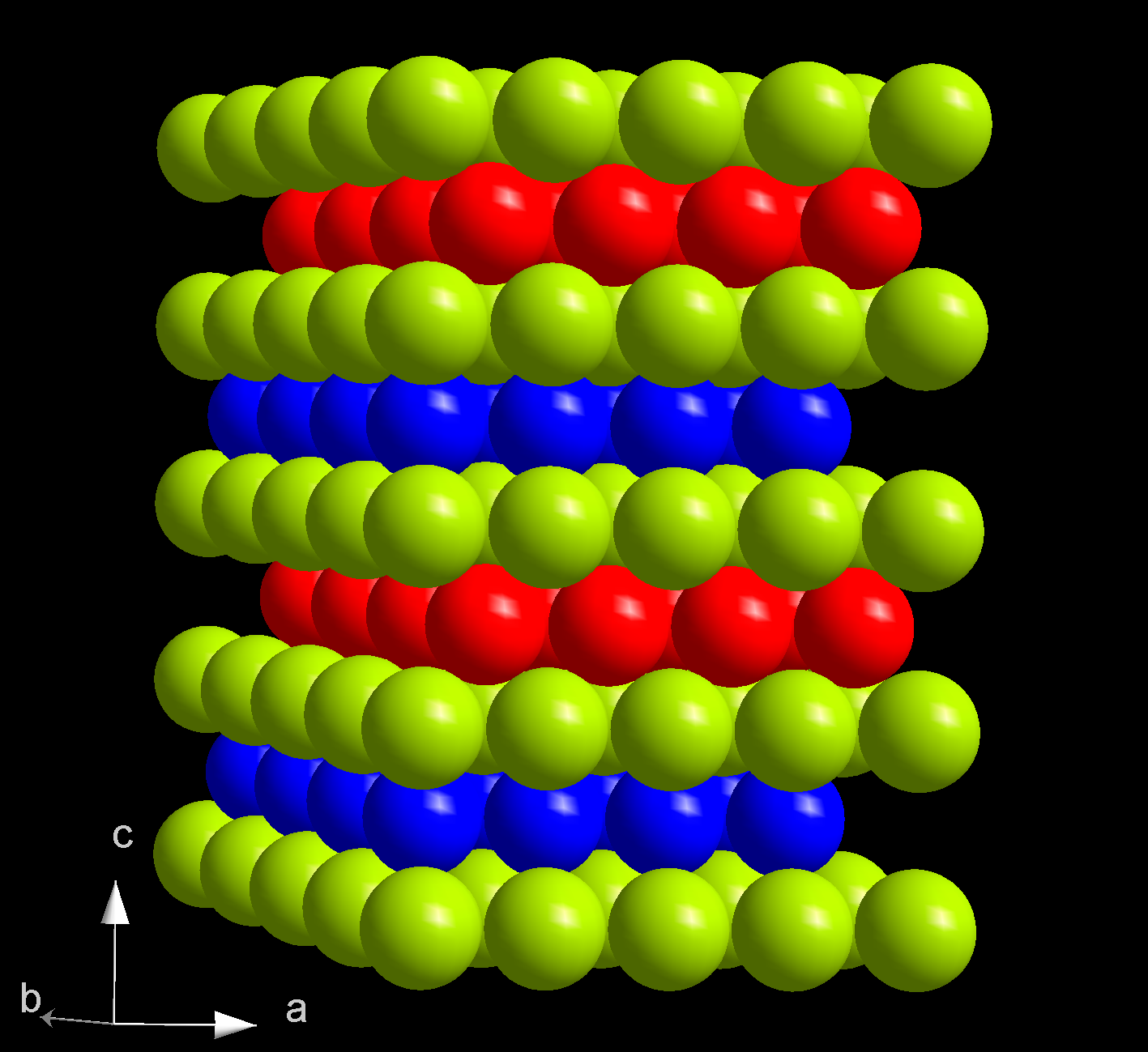 In the periodic table, americium is located to the right of plutonium, to the left of curium, and below the lanthanide europium, with which it shares many physical and chemical properties. Americium is a highly radioactive element. When freshly prepared, it has a silvery-white metallic lustre, but then slowly tarnishes in air. With a density of 12 g/cm3, americium is less dense than both curium (13.52 g/cm3) and plutonium (19.8 g/cm3); but has a higher density than europium (5.264 g/cm3)—mostly because of its higher atomic mass. Americium is relatively soft and easily deformable and has a significantly lower
In the periodic table, americium is located to the right of plutonium, to the left of curium, and below the lanthanide europium, with which it shares many physical and chemical properties. Americium is a highly radioactive element. When freshly prepared, it has a silvery-white metallic lustre, but then slowly tarnishes in air. With a density of 12 g/cm3, americium is less dense than both curium (13.52 g/cm3) and plutonium (19.8 g/cm3); but has a higher density than europium (5.264 g/cm3)—mostly because of its higher atomic mass. Americium is relatively soft and easily deformable and has a significantly lower
+ \underset -> \atop 400 - 500 ^\circ \ce C +
Americium(III) fluoride (AmF3) is poorly soluble and precipitates upon reaction of Am3+ and fluoride ions in weak acidic solutions:
: Am^3+ + 3F^- -> AmF3(v)
The tetravalent americium(IV) fluoride (AmF4) is obtained by reacting solid americium(III) fluoride with molecular fluorine:Greenwood, p. 1271
: 2AmF3 + F2 -> 2AmF4
Another known form of solid tetravalent americium fluoride is KAmF5.Penneman, p. 6 Tetravalent americium has also been observed in the aqueous phase. For this purpose, black Am(OH)4 was dissolved in 15- M NH4F with the americium concentration of 0.01 M. The resulting reddish solution had a characteristic optical absorption spectrum which is similar to that of AmF4 but differed from other oxidation states of americium. Heating the Am(IV) solution to 90 °C did not result in its disproportionation or reduction, however a slow reduction was observed to Am(III) and assigned to self-irradiation of americium by alpha particles.
Most americium(III) halides form hexagonal crystals with slight variation of the color and exact structure between the halogens. So, chloride (AmCl3) is reddish and has a structure isotypic to
 Analogous to uranocene, americium forms the organometallic compound amerocene with two cyclooctatetraene ligands, with the chemical formula (η8-C8H8)2Am. A
Analogous to uranocene, americium forms the organometallic compound amerocene with two cyclooctatetraene ligands, with the chemical formula (η8-C8H8)2Am. A
"Evaluation of nuclear criticality safety data and limits for actinides in transport"
p. 16. Scarcity and high price yet hinder application of americium as a nuclear fuel in
Performance of Home Smoke Alarms Analysis of the Response of Several Available Technologies in Residential Fire Settings
, NIST Technical Note 1455-1
, G.L. Kulcinski, NEEP 602 Course Notes (Spring 2000), Nuclear Power in Space, University of Wisconsin Fusion Technology Institute (see last page) Although americium produces less heat and electricity – the power yield is 114.7 mW/g for 241Am and 6.31 mW/g for 243Am (cf. 390 mW/g for 238Pu) – and its radiation poses more threat to humans owing to neutron emission, the
^_Am -> ^_Np + ^_He + \gamma
: ^_Be + ^_He -> ^_C + ^_n + \gamma
The most widespread use of 241AmBe neutron sources is a neutron probe – a device used to measure the quantity of water present in soil, as well as moisture/density for quality control in highway construction. 241Am neutron sources are also used in well logging applications, as well as in neutron radiography, tomography and other radiochemical investigations.
^_Am -> ce^_Am -> beta^-10.1 \ \ce] ^_Cm
Irradiation of 241Am by 12C or 22Ne ions yields the isotopes 247Es ( einsteinium) or 260Db ( dubnium), respectively. Furthermore, the element
The Radioactive Boy Scout: When a teenager attempts to build a breeder reactor
'' Harper's Magazine'', November 1998 There have been a few cases of exposure to americium, the worst case being that of chemical operations technician Harold McCluskey, who at the age of 64 was exposed to 500 times the occupational standard for americium-241 as a result of an explosion in his lab. McCluskey died at the age of 75 of unrelated pre-existing disease.
The radiochemistry of americium and curium
University of California, Los Alamos, California, 1960 *
at '' The Periodic Table of Videos'' (University of Nottingham)
ATSDR – Public Health Statement: Americium
{{Authority control Chemical elements Chemical elements with double hexagonal close-packed structure Actinides Carcinogens Synthetic elements
radioactive
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is consi ...
chemical element
A chemical element is a species of atoms that have a given number of protons in their atomic nucleus, nuclei, including the pure Chemical substance, substance consisting only of that species. Unlike chemical compounds, chemical elements canno ...
with the symbol Am and atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of an atomic nucleus. For ordinary nuclei, this is equal to the proton number (''n''p) or the number of protons found in the nucleus of ever ...
95. It is a transuranic member of the actinide
The actinide () or actinoid () series encompasses the 15 metallic chemical elements with atomic numbers from 89 to 103, actinium through lawrencium. The actinide series derives its name from the first element in the series, actinium. The info ...
series, in the periodic table located under the lanthanide element europium, and thus by analogy was named after the Americas
The Americas, which are sometimes collectively called America, are a landmass comprising the totality of North America, North and South America. The Americas make up most of the land in Earth's Western Hemisphere and comprise the New World. ...
.
Americium was first produced in 1944 by the group of Glenn T. Seaborg from Berkeley, California
Berkeley ( ) is a city on the eastern shore of San Francisco Bay in northern Alameda County, California, United States. It is named after the 18th-century Irish bishop and philosopher George Berkeley. It borders the cities of Oakland and E ...
, at the Metallurgical Laboratory of the University of Chicago
The University of Chicago (UChicago, Chicago, U of C, or UChi) is a private research university in Chicago, Illinois. Its main campus is located in Chicago's Hyde Park neighborhood. The University of Chicago is consistently ranked among the b ...
, as part of the Manhattan Project
The Manhattan Project was a research and development undertaking during World War II that produced the first nuclear weapons. It was led by the United States with the support of the United Kingdom and Canada. From 1942 to 1946, the project w ...
. Although it is the third element in the transuranic series, it was discovered fourth, after the heavier curium. The discovery was kept secret and only released to the public in November 1945. Most americium is produced by uranium
Uranium is a chemical element with the symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium is weak ...
or plutonium
Plutonium is a radioactive chemical element with the symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, and forms a dull coating when oxidized. The element normally exh ...
being bombarded with neutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the atomic nucleus, nuclei of atoms. Since protons and ...
s in nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
s – one tonne
The tonne ( or ; symbol: t) is a unit of mass equal to 1000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton to distinguish it from the non-metric units of the short ton ( United State ...
of spent nuclear fuel contains about 100 grams of americium. It is widely used in commercial ionization chamber smoke detector
A smoke detector is a device that senses smoke, typically as an indicator of fire. Smoke detectors are usually housed in plastic enclosures, typically shaped like a disk about in diameter and thick, but shape and size vary. Smoke can be detecte ...
s, as well as in neutron sources and industrial gauges. Several unusual applications, such as nuclear batteries or fuel for space ships with nuclear propulsion, have been proposed for the isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers ( mass num ...
242mAm, but they are as yet hindered by the scarcity and high price of this nuclear isomer.
Americium is a relatively soft radioactive
Radioactive decay (also known as nuclear decay, radioactivity, radioactive disintegration, or nuclear disintegration) is the process by which an unstable atomic nucleus loses energy by radiation. A material containing unstable nuclei is consi ...
metal with silvery appearance. Its most common isotopes
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers ( mass numbers ...
are 241Am and 243Am. In chemical compounds, americium usually assumes the oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
+3, especially in solutions. Several other oxidation states are known, ranging from +2 to +7, and can be identified by their characteristic optical absorption
In physics, absorption of electromagnetic radiation is how matter (typically electrons bound in atoms) takes up a photon's energy — and so transforms electromagnetic energy into internal energy of the absorber (for example, thermal energy). A ...
spectra. The crystal lattice of solid americium and its compounds contain small intrinsic radiogenic defects, due to metamictization
Metamictisation (sometimes called metamictization or metamiction) is a natural process resulting in the gradual and ultimately complete destruction of a mineral's crystal structure, leaving the mineral amorphous. The affected material is therefore ...
induced by self-irradiation with alpha particles, which accumulates with time; this can cause a drift of some material properties over time, more noticeable in older samples.
History
 Although americium was likely produced in previous nuclear experiments, it was first intentionally synthesized, isolated and identified in late autumn 1944, at the
Although americium was likely produced in previous nuclear experiments, it was first intentionally synthesized, isolated and identified in late autumn 1944, at the University of California, Berkeley
The University of California, Berkeley (UC Berkeley, Berkeley, Cal, or California) is a public land-grant research university in Berkeley, California. Established in 1868 as the University of California, it is the state's first land-grant un ...
, by Glenn T. Seaborg, Leon O. Morgan, Ralph A. James
Ralph Arthur James (23 September 1920 in Salt Lake City, Utah – 24 February 1973 in Alamo, California) was an American chemist at the University of Chicago who co-discovered the elements curium (1944) and americium (1944–1945). Later h ...
, and Albert Ghiorso. They used a 60-inch cyclotron at the University of California, Berkeley. The element was chemically identified at the Metallurgical Laboratory (now Argonne National Laboratory) of the University of Chicago
The University of Chicago (UChicago, Chicago, U of C, or UChi) is a private research university in Chicago, Illinois. Its main campus is located in Chicago's Hyde Park neighborhood. The University of Chicago is consistently ranked among the b ...
. Following the lighter neptunium, plutonium
Plutonium is a radioactive chemical element with the symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, and forms a dull coating when oxidized. The element normally exh ...
, and heavier curium, americium was the fourth transuranium element to be discovered. At the time, the periodic table had been restructured by Seaborg to its present layout, containing the actinide row below the lanthanide one. This led to americium being located right below its twin lanthanide element europium; it was thus by analogy named after the Americas
The Americas, which are sometimes collectively called America, are a landmass comprising the totality of North America, North and South America. The Americas make up most of the land in Earth's Western Hemisphere and comprise the New World. ...
: "The name americium (after the Americas) and the symbol Am are suggested for the element on the basis of its position as the sixth member of the actinide rare-earth series, analogous to europium, Eu, of the lanthanide series."Greenwood, p. 1252
The new element was isolated from its oxides in a complex, multi-step process. First plutonium
Plutonium is a radioactive chemical element with the symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, and forms a dull coating when oxidized. The element normally exh ...
-239 nitrate (239PuNO3) solution was coated on a platinum
Platinum is a chemical element with the symbol Pt and atomic number 78. It is a dense, malleable, ductile, highly unreactive, precious, silverish-white transition metal. Its name originates from Spanish , a diminutive of "silver".
Pla ...
foil of about 0.5 cm2 area, the solution was evaporated and the residue was converted into plutonium dioxide (PuO2) by calcining. After cyclotron irradiation, the coating was dissolved with nitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available ni ...
, and then precipitated as the hydroxide using concentrated aqueous ammonia solution. The residue was dissolved in perchloric acid
Perchloric acid is a mineral acid with the formula H Cl O4. Usually found as an aqueous solution, this colorless compound is a stronger acid than sulfuric acid, nitric acid and hydrochloric acid. It is a powerful oxidizer when hot, but aqueo ...
. Further separation was carried out by ion exchange, yielding a certain isotope of curium. The separation of curium and americium was so painstaking that those elements were initially called by the Berkeley group as '' pandemonium'' (from Greek for ''all demons'' or ''hell'') and '' delirium'' (from Latin for ''madness'').
Initial experiments yielded four americium isotopes: 241Am, 242Am, 239Am and 238Am. Americium-241 was directly obtained from plutonium upon absorption of two neutrons. It decays by emission of a α-particle to 237Np; the half-life
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable ...
of this decay was first determined as years but then corrected to 432.2 years.
:
: The times are half-lives
Half-life (symbol ) is the time required for a quantity (of substance) to reduce to half of its initial value. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable ato ...
The second isotope 242Am was produced upon neutron bombardment of the already-created 241Am. Upon rapid β-decay, 242Am converts into the isotope of curium 242Cm (which had been discovered previously). The half-life of this decay was initially determined at 17 hours, which was close to the presently accepted value of 16.02 h.
:
The discovery of americium and curium in 1944 was closely related to the Manhattan Project
The Manhattan Project was a research and development undertaking during World War II that produced the first nuclear weapons. It was led by the United States with the support of the United Kingdom and Canada. From 1942 to 1946, the project w ...
; the results were confidential and declassified only in 1945. Seaborg leaked the synthesis of the elements 95 and 96 on the U.S. radio show for children '' Quiz Kids'' five days before the official presentation at an American Chemical Society
The American Chemical Society (ACS) is a scientific society based in the United States that supports scientific inquiry in the field of chemistry. Founded in 1876 at New York University, the ACS currently has more than 155,000 members at all ...
meeting on 11 November 1945, when one of the listeners asked whether any new transuranium element besides plutonium and neptunium had been discovered during the war. After the discovery of americium isotopes 241Am and 242Am, their production and compounds were patented listing only Seaborg as the inventor. The initial americium samples weighed a few micrograms; they were barely visible and were identified by their radioactivity. The first substantial amounts of metallic americium weighing 40–200 micrograms were not prepared until 1951 by reduction of americium(III) fluoride with barium metal in high vacuum at 1100 °C.
Occurrence
 The longest-lived and most common isotopes of americium, 241Am and 243Am, have half-lives of 432.2 and 7,370 years, respectively. Therefore, any
The longest-lived and most common isotopes of americium, 241Am and 243Am, have half-lives of 432.2 and 7,370 years, respectively. Therefore, any primordial
Primordial may refer to:
* Primordial era, an era after the Big Bang. See Chronology of the universe
* Primordial sea (a.k.a. primordial ocean, ooze or soup). See Abiogenesis
* Primordial nuclide, nuclides, a few radioactive, that formed before t ...
americium (americium that was present on Earth during its formation) should have decayed by now. Trace amounts of americium probably occur naturally in uranium minerals as a result of nuclear reactions, though this has not been confirmed.
Existing americium is concentrated in the areas used for the atmospheric nuclear weapons tests conducted between 1945 and 1980, as well as at the sites of nuclear incidents, such as the Chernobyl disaster. For example, the analysis of the debris at the testing site of the first U.S. hydrogen bomb, Ivy Mike
Ivy Mike was the codename given to the first full-scale test of a thermonuclear device, in which part of the explosive yield comes from nuclear fusion.
Ivy Mike was detonated on November 1, 1952, by the United States on the island of Elugelab ...
, (1 November 1952, Enewetak Atoll), revealed high concentrations of various actinides including americium; but due to military secrecy, this result was not published until later, in 1956. Trinitite, the glassy residue left on the desert floor near Alamogordo
Alamogordo () is the seat of Otero County, New Mexico, United States. A city in the Tularosa Basin of the Chihuahuan Desert, it is bordered on the east by the Sacramento Mountains and to the west by Holloman Air Force Base. The population was ...
, New Mexico
)
, population_demonym = New Mexican ( es, Neomexicano, Neomejicano, Nuevo Mexicano)
, seat = Santa Fe, New Mexico, Santa Fe
, LargestCity = Albuquerque, New Mexico, Albuquerque
, LargestMetro = Albuquerque metropolitan area, Tiguex
, Offi ...
, after the plutonium
Plutonium is a radioactive chemical element with the symbol Pu and atomic number 94. It is an actinide metal of silvery-gray appearance that tarnishes when exposed to air, and forms a dull coating when oxidized. The element normally exh ...
-based Trinity
The Christian doctrine of the Trinity (, from 'threefold') is the central dogma concerning the nature of God in most Christian churches, which defines one God existing in three coequal, coeternal, consubstantial divine persons: God th ...
nuclear bomb test on 16 July 1945, contains traces of americium-241. Elevated levels of americium were also detected at the crash site of a US Boeing B-52
The Boeing B-52 Stratofortress is an American long-range, subsonic, jet-powered strategic bomber. The B-52 was designed and built by Boeing, which has continued to provide support and upgrades. It has been operated by the United States Air ...
bomber aircraft, which carried four hydrogen bombs, in 1968 in Greenland
Greenland ( kl, Kalaallit Nunaat, ; da, Grønland, ) is an island country in North America that is part of the Kingdom of Denmark. It is located between the Arctic and Atlantic oceans, east of the Canadian Arctic Archipelago. Greenland ...
.
In other regions, the average radioactivity of surface soil due to residual americium is only about 0.01 picocuries/g (0.37 mBq/g). Atmospheric americium compounds are poorly soluble in common solvents and mostly adhere to soil particles. Soil analysis revealed about 1,900 times higher concentration of americium inside sandy soil particles than in the water present in the soil pores; an even higher ratio was measured in loam soils.Human Health Fact Sheet on Americium, Los Alamos National Laboratory, Retrieved 28 November 2010 Americium is produced mostly artificially in small quantities, for research purposes. A tonne of spent nuclear fuel contains about 100 grams of various americium isotopes, mostly 241Am and 243Am. Their prolonged radioactivity is undesirable for the disposal, and therefore americium, together with other long-lived actinides, must be neutralized. The associated procedure may involve several steps, where americium is first separated and then converted by neutron bombardment in special reactors to short-lived nuclides. This procedure is well known as nuclear transmutation, but it is still being developed for americium. The transuranic elements from americium to fermium occurred naturally in the
natural nuclear fission reactor
A natural nuclear fission reactor is a uranium deposit where self-sustaining nuclear chain reactions occur. The conditions under which a natural nuclear reactor could exist had been predicted in 1956 by Japanese American chemist Paul Kuroda. ...
at Oklo
Oklo is a region near the town of Franceville, in the Haut-Ogooué province of the Central African country of Gabon. Several natural nuclear fission reactors were discovered in the uranium mines in the region in 1972.
History
Gabon was a Frenc ...
, but no longer do so.
Americium is also one of the elements that have been detected in Przybylski's Star.
Synthesis and extraction
Isotope nucleosynthesis
 Americium has been produced in small quantities in
Americium has been produced in small quantities in nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
s for decades, and kilograms of its 241Am and 243Am isotopes have been accumulated by now.Greenwood, p. 1262 Nevertheless, since it was first offered for sale in 1962, its price, about of 241Am, remains almost unchanged owing to the very complex separation procedure.Smoke detectors and americium, World Nuclear Association, January 2009, Retrieved 28 November 2010 The heavier isotope 243Am is produced in much smaller amounts; it is thus more difficult to separate, resulting in a higher cost of the order .Hammond C. R. "The elements" in Americium is not synthesized directly from uranium – the most common reactor material – but from the plutonium isotope 239Pu. The latter needs to be produced first, according to the following nuclear process: :
Metal generation
Most synthesis routines yield a mixture of different actinide isotopes in oxide forms, from which isotopes of americium can be separated. In a typical procedure, the spent reactor fuel (e.g. MOX fuel) is dissolved innitric acid
Nitric acid is the inorganic compound with the formula . It is a highly corrosive mineral acid. The compound is colorless, but older samples tend to be yellow cast due to decomposition into oxides of nitrogen. Most commercially available ni ...
, and the bulk of uranium and plutonium is removed using a PUREX
PUREX (plutonium uranium reduction extraction) is a chemical method used to purify fuel for nuclear reactors or nuclear weapons. PUREX is the '' de facto'' standard aqueous nuclear reprocessing method for the recovery of uranium and pluto ...
-type extraction (Plutonium–URanium EXtraction) with tributyl phosphate in a hydrocarbon
In organic chemistry, a hydrocarbon is an organic compound consisting entirely of hydrogen and carbon. Hydrocarbons are examples of group 14 hydrides. Hydrocarbons are generally colourless and hydrophobic, and their odors are usually weak or ...
. The lanthanides and remaining actinides are then separated from the aqueous residue ( raffinate) by a diamide-based extraction, to give, after stripping, a mixture of trivalent actinides and lanthanides. Americium compounds are then selectively extracted using multi-step chromatographic
In chemical analysis, chromatography is a laboratory technique for the separation of a mixture into its components. The mixture is dissolved in a fluid solvent (gas or liquid) called the ''mobile phase'', which carries it through a system (a ...
and centrifugation techniques with an appropriate reagent. A large amount of work has been done on the solvent extraction of americium. For example, a 2003 EU-funded project codenamed "EUROPART" studied triazines and other compounds as potential extraction agents. A ''bis''-triazinyl bipyridine complex was proposed in 2009 as such a reagent is highly selective to americium (and curium). Separation of americium from the highly similar curium can be achieved by treating a slurry of their hydroxides in aqueous sodium bicarbonate with ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the l ...
, at elevated temperatures. Both Am and Cm are mostly present in solutions in the +3 valence state; whereas curium remains unchanged, americium oxidizes to soluble Am(IV) complexes which can be washed away.
Metallic americium is obtained by reduction from its compounds. Americium(III) fluoride was first used for this purpose. The reaction was conducted using elemental barium as reducing agent in a water- and oxygen-free environment inside an apparatus made of tantalum and tungsten
Tungsten, or wolfram, is a chemical element with the symbol W and atomic number 74. Tungsten is a rare metal found naturally on Earth almost exclusively as compounds with other elements. It was identified as a new element in 1781 and first isol ...
.''Gmelin Handbook of Inorganic Chemistry'', System No. 71, transuranics, Part B 1, pp. 57–67.Penneman, p. 3
:
An alternative is the reduction of americium dioxide by metallic lanthanum
Lanthanum is a chemical element with the symbol La and atomic number 57. It is a soft, ductile, silvery-white metal that tarnishes slowly when exposed to air. It is the eponym of the lanthanide series, a group of 15 similar elements between l ...
or thorium:
:
Physical properties
 In the periodic table, americium is located to the right of plutonium, to the left of curium, and below the lanthanide europium, with which it shares many physical and chemical properties. Americium is a highly radioactive element. When freshly prepared, it has a silvery-white metallic lustre, but then slowly tarnishes in air. With a density of 12 g/cm3, americium is less dense than both curium (13.52 g/cm3) and plutonium (19.8 g/cm3); but has a higher density than europium (5.264 g/cm3)—mostly because of its higher atomic mass. Americium is relatively soft and easily deformable and has a significantly lower
In the periodic table, americium is located to the right of plutonium, to the left of curium, and below the lanthanide europium, with which it shares many physical and chemical properties. Americium is a highly radioactive element. When freshly prepared, it has a silvery-white metallic lustre, but then slowly tarnishes in air. With a density of 12 g/cm3, americium is less dense than both curium (13.52 g/cm3) and plutonium (19.8 g/cm3); but has a higher density than europium (5.264 g/cm3)—mostly because of its higher atomic mass. Americium is relatively soft and easily deformable and has a significantly lower bulk modulus
The bulk modulus (K or B) of a substance is a measure of how resistant to compression the substance is. It is defined as the ratio of the infinitesimal pressure increase to the resulting ''relative'' decrease of the volume.
Other moduli descri ...
than the actinides before it: Th, Pa, U, Np and Pu. Its melting point of 1173 °C is significantly higher than that of plutonium (639 °C) and europium (826 °C), but lower than for curium (1340 °C).
At ambient conditions, americium is present in its most stable α form which has a hexagonal crystal symmetry, and a space group P63/mmc with cell parameters ''a'' = 346.8 pm and ''c'' = 1124 pm, and four atoms per unit cell. The crystal consists of a double- hexagonal close packing with the layer sequence ABAC and so is isotypic with α-lanthanum and several actinides such as α-curium. The crystal structure of americium changes with pressure and temperature. When compressed at room temperature to 5 GPa, α-Am transforms to the β modification, which has a face-centered cubic (''fcc'') symmetry, space group Fmm and lattice constant ''a'' = 489 pm. This ''fcc'' structure is equivalent to the closest packing with the sequence ABC. Upon further compression to 23 GPa, americium transforms to an orthorhombic γ-Am structure similar to that of α-uranium. There are no further transitions observed up to 52 GPa, except for an appearance of a monoclinic phase at pressures between 10 and 15 GPa. There is no consistency on the status of this phase in the literature, which also sometimes lists the α, β and γ phases as I, II and III. The β-γ transition is accompanied by a 6% decrease in the crystal volume; although theory also predicts a significant volume change for the α-β transition, it is not observed experimentally. The pressure of the α-β transition decreases with increasing temperature, and when α-americium is heated at ambient pressure, at 770 °C it changes into an ''fcc'' phase which is different from β-Am, and at 1075 °C it converts to a body-centered cubic structure. The pressure-temperature phase diagram of americium is thus rather similar to those of lanthanum, praseodymium and neodymium
Neodymium is a chemical element with the symbol Nd and atomic number 60. It is the fourth member of the lanthanide series and is considered to be one of the rare-earth metals. It is a hard, slightly malleable, silvery metal that quickly tarn ...
.
As with many other actinides, self-damage of the crystal structure due to alpha-particle irradiation is intrinsic to americium. It is especially noticeable at low temperatures, where the mobility of the produced structure defects is relatively low, by broadening of X-ray diffraction peaks. This effect makes somewhat uncertain the temperature of americium and some of its properties, such as electrical resistivity. So for americium-241, the resistivity at 4.2 K increases with time from about 2 µOhm·cm to 10 µOhm·cm after 40 hours, and saturates at about 16 µOhm·cm after 140 hours. This effect is less pronounced at room temperature, due to annihilation of radiation defects; also heating to room temperature the sample which was kept for hours at low temperatures restores its resistivity. In fresh samples, the resistivity gradually increases with temperature from about 2 µOhm·cm at liquid helium to 69 µOhm·cm at room temperature; this behavior is similar to that of neptunium, uranium, thorium and protactinium, but is different from plutonium and curium which show a rapid rise up to 60 K followed by saturation. The room temperature value for americium is lower than that of neptunium, plutonium and curium, but higher than for uranium, thorium and protactinium.
Americium is paramagnetic in a wide temperature range, from that of liquid helium, to room temperature and above. This behavior is markedly different from that of its neighbor curium which exhibits antiferromagnetic transition at 52 K. The thermal expansion
Thermal expansion is the tendency of matter to change its shape, area, volume, and density in response to a change in temperature, usually not including phase transitions.
Temperature is a monotonic function of the average molecular kin ...
coefficient of americium is slightly anisotropic and amounts to along the shorter ''a'' axis and for the longer ''c'' hexagonal axis. The enthalpy of dissolution of americium metal in hydrochloric acid
Hydrochloric acid, also known as muriatic acid, is an aqueous solution of hydrogen chloride. It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid. It is a component of the gastric acid in the dige ...
at standard conditions is , from which the standard enthalpy change of formation (Δf''H''°) of aqueous Am3+ ion is . The standard potential Am3+/Am0 is .
Chemical properties
Americium metal readily reacts with oxygen and dissolves in aqueous acids. The most stableoxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical charge of an atom if all of its bonds to different atoms were fully ionic. It describes the degree of oxidation (loss of electrons) of an atom in a chemical compound. C ...
for americium is +3,.Penneman, p. 4 The chemistry of americium(III) has many similarities to the chemistry of lanthanide(III) compounds. For example, trivalent americium forms insoluble fluoride, oxalate, iodate, hydroxide
Hydroxide is a diatomic anion with chemical formula OH−. It consists of an oxygen and hydrogen atom held together by a single covalent bond, and carries a negative electric charge. It is an important but usually minor constituent of water. ...
, phosphate
In chemistry, a phosphate is an anion, salt, functional group or ester derived from a phosphoric acid. It most commonly means orthophosphate, a derivative of orthophosphoric acid .
The phosphate or orthophosphate ion is derived from phosph ...
and other salts. Compounds of americium in oxidation states 2, 4, 5, 6 and 7 have also been studied. This is the widest range that has been observed with actinide elements. The color of americium compounds in aqueous solution is as follows: Am3+ (yellow-reddish), Am4+ (yellow-reddish), ; (yellow), (brown) and (dark green).Greenwood, p. 1265 The absorption spectra have sharp peaks, due to ''f''-''f'' transitions' in the visible and near-infrared regions. Typically, Am(III) has absorption maxima at ca. 504 and 811 nm, Am(V) at ca. 514 and 715 nm, and Am(VI) at ca. 666 and 992 nm.
Americium compounds with oxidation state +4 and higher are strong oxidizing agents, comparable in strength to the permanganate
A permanganate () is a chemical compound containing the manganate(VII) ion, , the conjugate base of permanganic acid. Because the manganese atom is in the +7 oxidation state, the permanganate(VII) ion is a strong oxidizing agent. The ion is a ...
ion () in acidic solutions.Wiberg, p. 1956 Whereas the Am4+ ions are unstable in solutions and readily convert to Am3+, compounds such as americium dioxide (AmO2) and americium(IV) fluoride (AmF4) are stable in the solid state.
The pentavalent oxidation state of americium was first observed in 1951. In acidic aqueous solution the ion is unstable with respect to disproportionation
In chemistry, disproportionation, sometimes called dismutation, is a redox reaction in which one compound of intermediate oxidation state converts to two compounds, one of higher and one of lower oxidation states. More generally, the term can ...
.Greenwood, p. 1275 The reaction
:
is typical. The chemistry of Am(V) and Am(VI) is comparable to the chemistry of uranium
Uranium is a chemical element with the symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium is weak ...
in those oxidation states. In particular, compounds like and are comparable to uranates and the ion is comparable to the uranyl ion, . Such compounds can be prepared by oxidation of Am(III) in dilute nitric acid with ammonium persulfate. Other oxidising agents that have been used include silver(I) oxide, ozone
Ozone (), or trioxygen, is an inorganic molecule with the chemical formula . It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope , breaking down in the l ...
and sodium persulfate
Sodium persulfate is the inorganic compound with the formula Na2 S2 O8. It is the sodium salt of peroxydisulfuric acid, H2S2O8, an oxidizing agent. It is a white solid that dissolves in water. It is almost non-hygroscopic and has good shelf-lif ...
.
Chemical compounds
Oxygen compounds
Three americium oxides are known, with the oxidation states +2 (AmO), +3 (Am2O3) and +4 (AmO2). Americium(II) oxide was prepared in minute amounts and has not been characterized in detail. Americium(III) oxide is a red-brown solid with a melting point of 2205 °C.Wiberg, p. 1972 Americium(IV) oxide is the main form of solid americium which is used in nearly all its applications. As most other actinide dioxides, it is a black solid with a cubic ( fluorite) crystal structure.Greenwood, p. 1267 The oxalate of americium(III), vacuum dried at room temperature, has the chemical formula Am2(C2O4)3·7H2O. Upon heating in vacuum, it loses water at 240 °C and starts decomposing into AmO2 at 300 °C, the decomposition completes at about 470 °C. The initial oxalate dissolves in nitric acid with the maximum solubility of 0.25 g/L.Penneman, p. 5Halides
Halides of americium are known for the oxidation states +2, +3 and +4,Wiberg, p. 1969 where the +3 is most stable, especially in solutions. Reduction of Am(III) compounds with sodiumamalgam
Amalgam most commonly refers to:
* Amalgam (chemistry), mercury alloy
* Amalgam (dentistry), material of silver tooth fillings
** Bonded amalgam, used in dentistry
Amalgam may also refer to:
* Amalgam Comics, a publisher
* Amalgam Digital, an in ...
yields Am(II) salts – the black halides AmCl2, AmBr2 and AmI2. They are very sensitive to oxygen and oxidize in water, releasing hydrogen and converting back to the Am(III) state. Specific lattice constants are:
* Orthorhombic AmCl2: ''a'' = , ''b'' = and ''c'' =
* Tetragonal AmBr2: ''a'' = and ''c'' = . They can also be prepared by reacting metallic americium with an appropriate mercury halide HgX2, where X = Cl, Br or I:Greenwood, p. 1272
: uranium(III) chloride
Uranium(III) chloride, UCl3, is a water soluble salt of uranium. UCl3 is used mostly to reprocess spent nuclear fuel. Uranium(III) chloride is synthesized in various ways from uranium(IV) chloride; however, UCl3 is less stable than UCl4.
Prepar ...
(space group P63/m) and the melting point of 715 °C. The fluoride is isotypic to LaF3 (space group P63/mmc) and the iodide to BiI3 (space group R). The bromide is an exception with the orthorhombic PuBr3-type structure and space group Cmcm. Crystals of americium hexahydrate (AmCl3·6H2O) can be prepared by dissolving americium dioxide in hydrochloric acid and evaporating the liquid. Those crystals are hygroscopic and have yellow-reddish color and a monoclinic
In crystallography, the monoclinic crystal system is one of the seven crystal systems. A crystal system is described by three vectors. In the monoclinic system, the crystal is described by vectors of unequal lengths, as in the orthorhombic ...
crystal structure.
Oxyhalides of americium in the form AmVIO2X2, AmVO2X, AmIVOX2 and AmIIIOX can be obtained by reacting the corresponding americium halide with oxygen or Sb2O3, and AmOCl can also be produced by vapor phase hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution, elimination, and solvation reactions in which water is the nucleophile.
Biological hydrolysi ...
:
: AmCl3 + H2O -> AmOCl + 2HCl
Chalcogenides and pnictides
The known chalcogenides of americium include thesulfide
Sulfide (British English also sulphide) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to chemical compounds la ...
AmS2, selenides AmSe2 and Am3Se4, and tellurides Am2Te3 and AmTe2. The pnictides
A pnictogen ( or ; from grc, πνῑ́γω "to choke" and -gen, "generator") is any of the chemical elements in group 15 of the periodic table. Group 15 is also known as the nitrogen group or nitrogen family. Group 15 consists of the ele ...
of americium (243Am) of the AmX type are known for the elements phosphorus
Phosphorus is a chemical element with the symbol P and atomic number 15. Elemental phosphorus exists in two major forms, white phosphorus and red phosphorus, but because it is highly reactive, phosphorus is never found as a free element on Ea ...
, arsenic
Arsenic is a chemical element with the symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a metalloid. It has various allotropes, b ...
, antimony
Antimony is a chemical element with the symbol Sb (from la, stibium) and atomic number 51. A lustrous gray metalloid, it is found in nature mainly as the sulfide mineral stibnite (Sb2S3). Antimony compounds have been known since ancient ti ...
and bismuth. They crystallize in the rock-salt
Halite (), commonly known as rock salt, is a type of salt, the mineral (natural) form of sodium chloride ( Na Cl). Halite forms isometric crystals. The mineral is typically colorless or white, but may also be light blue, dark blue, purple, p ...
lattice.
Silicides and borides
Americium monosilicide (AmSi) and "disilicide" (nominally AmSix with: 1.87 < x < 2.0) were obtained by reduction of americium(III) fluoride with elementarysilicon
Silicon is a chemical element with the symbol Si and atomic number 14. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. It is a member of group 14 in the periodic ...
in vacuum at 1050 °C (AmSi) and 1150−1200 °C (AmSix). AmSi is a black solid isomorphic with LaSi, it has an orthorhombic crystal symmetry. AmSix has a bright silvery lustre and a tetragonal crystal lattice (space group ''I''41/amd), it is isomorphic with PuSi2 and ThSi2. Borides of americium include AmB4 and AmB6. The tetraboride can be obtained by heating an oxide or halide of americium with magnesium diboride
Magnesium diboride is the inorganic compound with the formula MgB2. It is a dark gray, water-insoluble solid. The compound has attracted attention because it becomes superconducting at 39 K (−234 °C). In terms of its composition, Mg ...
in vacuum or inert atmosphere.
Organoamericium compounds
 Analogous to uranocene, americium forms the organometallic compound amerocene with two cyclooctatetraene ligands, with the chemical formula (η8-C8H8)2Am. A
Analogous to uranocene, americium forms the organometallic compound amerocene with two cyclooctatetraene ligands, with the chemical formula (η8-C8H8)2Am. A cyclopentadienyl complex
A cyclopentadienyl complex is a coordination complex of a metal and cyclopentadienyl groups (, abbreviated as Cp−). Cyclopentadienyl ligands almost invariably bind to metals as a pentahapto (''η''5-) bonding mode. The metal–cyclopentadien ...
is also known that is likely to be stoichiometrically AmCp3.
Formation of the complexes of the type Am(n-C3H7-BTP)3, where BTP stands for 2,6-di(1,2,4-triazin-3-yl)pyridine, in solutions containing n-C3H7-BTP and Am3+ ions has been confirmed by EXAFS. Some of these BTP-type complexes selectively interact with americium and therefore are useful in its selective separation from lanthanides and another actinides.
Biological aspects
Americium is an artificial element of recent origin, and thus does not have a biological requirement. It is harmful tolife
Life is a quality that distinguishes matter that has biological processes, such as signaling and self-sustaining processes, from that which does not, and is defined by the capacity for growth, reaction to stimuli, metabolism, energy ...
. It has been proposed to use bacteria for removal of americium and other heavy metals from rivers and streams. Thus, Enterobacteriaceae of the genus '' Citrobacter'' precipitate americium ions from aqueous solutions, binding them into a metal-phosphate complex at their cell walls. Several studies have been reported on the biosorption and bioaccumulation of americium by bacteria and fungi.
Fission
The isotope 242mAm (half-life 141 years) has the largest cross sections for absorption of thermal neutrons (5,700barns
A barn is an agricultural building usually on farms and used for various purposes. In North America, a barn refers to structures that house livestock, including cattle and horses, as well as equipment and fodder, and often grain.Allen G ...
),Pfennig, G.; Klewe-Nebenius, H and Seelmann Eggebert, W. (Eds.): Karlsruhe nuclide, 7 Edition 2006. that results in a small critical mass
In nuclear engineering, a critical mass is the smallest amount of fissile material needed for a sustained nuclear chain reaction. The critical mass of a fissionable material depends upon its nuclear properties (specifically, its nuclear fi ...
for a sustained nuclear chain reaction. The critical mass for a bare 242mAm sphere is about 9–14 kg (the uncertainty results from insufficient knowledge of its material properties). It can be lowered to 3–5 kg with a metal reflector and should become even smaller with a water reflector. Such small critical mass is favorable for portable nuclear weapon
A nuclear weapon is an explosive device that derives its destructive force from nuclear reactions, either fission (fission bomb) or a combination of fission and fusion reactions ( thermonuclear bomb), producing a nuclear explosion. Both bomb ...
s, but those based on 242mAm are not known yet, probably because of its scarcity and high price. The critical masses of two other readily available isotopes, 241Am and 243Am, are relatively high – 57.6 to 75.6 kg for 241Am and 209 kg for 243Am.Institut de Radioprotection et de Sûreté Nucléaire"Evaluation of nuclear criticality safety data and limits for actinides in transport"
p. 16. Scarcity and high price yet hinder application of americium as a nuclear fuel in
nuclear reactor
A nuclear reactor is a device used to initiate and control a fission nuclear chain reaction or nuclear fusion reactions. Nuclear reactors are used at nuclear power plants for electricity generation and in nuclear marine propulsion. Heat fr ...
s.
There are proposals of very compact 10-kW high-flux reactors using as little as 20 grams of 242mAm. Such low-power reactors would be relatively safe to use as neutron sources for radiation therapy
Radiation therapy or radiotherapy, often abbreviated RT, RTx, or XRT, is a therapy using ionizing radiation, generally provided as part of cancer treatment to control or kill malignant cells and normally delivered by a linear accelerator. Rad ...
in hospitals.
Isotopes
About 19isotope
Isotopes are two or more types of atoms that have the same atomic number (number of protons in their nuclei) and position in the periodic table (and hence belong to the same chemical element), and that differ in nucleon numbers ( mass num ...
s and 8 nuclear isomers are known for americium. There are two long-lived alpha-emitters; 243Am has a half-life of 7,370 years and is the most stable isotope, and 241Am has a half-life of 432.2 years. The most stable nuclear isomer is 242m1Am; it has a long half-life of 141 years. The half-lives of other isotopes and isomers range from 0.64 microseconds for 245m1Am to 50.8 hours for 240Am. As with most other actinides, the isotopes of americium with odd number of neutrons have relatively high rate of nuclear fission and low critical mass.
Americium-241 decays to 237Np emitting alpha particles of 5 different energies, mostly at 5.486 MeV (85.2%) and 5.443 MeV (12.8%). Because many of the resulting states are metastable, they also emit gamma ray
A gamma ray, also known as gamma radiation (symbol γ or \gamma), is a penetrating form of electromagnetic radiation arising from the radioactive decay of atomic nuclei. It consists of the shortest wavelength electromagnetic waves, typically ...
s with the discrete energies between 26.3 and 158.5 keV.
Americium-242 is a short-lived isotope with a half-life of 16.02 h. It mostly (82.7%) converts by β-decay to 242Cm, but also by electron capture
Electron capture (K-electron capture, also K-capture, or L-electron capture, L-capture) is a process in which the proton-rich nucleus of an electrically neutral atom absorbs an inner atomic electron, usually from the K or L electron shells. ...
to 242Pu (17.3%). Both 242Cm and 242Pu transform via nearly the same decay chain through 238Pu down to 234U.
Nearly all (99.541%) of 242m1Am decays by internal conversion to 242Am and the remaining 0.459% by α-decay to 238Np. The latter subsequently decays to 238Pu and then to 234U.
Americium-243 transforms by α-emission into 239Np, which converts by β-decay to 239Pu, and the 239Pu changes into 235U by emitting an α-particle.
Applications
Ionization-type smoke detector
Americium is used in the most common type of householdsmoke detector
A smoke detector is a device that senses smoke, typically as an indicator of fire. Smoke detectors are usually housed in plastic enclosures, typically shaped like a disk about in diameter and thick, but shape and size vary. Smoke can be detecte ...
, which uses 241Am in the form of americium dioxide as its source of ionizing radiation. This isotope is preferred over 226 Ra because it emits 5 times more alpha particles and relatively little harmful gamma radiation.
The amount of americium in a typical new smoke detector is 1 microcurie (37 kBq) or 0.29 microgram. This amount declines slowly as the americium decays into neptunium-237, a different transuranic element with a much longer half-life (about 2.14 million years). With its half-life of 432.2 years, the americium in a smoke detector includes about 3% neptunium after 19 years, and about 5% after 32 years. The radiation passes through an ionization chamber, an air-filled space between two electrode
An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit (e.g. a semiconductor, an electrolyte, a vacuum or air). Electrodes are essential parts of batteries that can consist of a variety of materials ...
s, and permits a small, constant current between the electrodes. Any smoke that enters the chamber absorbs the alpha particles, which reduces the ionization and affects this current, triggering the alarm. Compared to the alternative optical smoke detector, the ionization smoke detector is cheaper and can detect particles which are too small to produce significant light scattering; however, it is more prone to false alarms
A false alarm, also called a nuisance alarm, is the deceptive or erroneous report of an emergency, causing unnecessary panic and/or bringing resources (such as emergency services) to a place where they are not needed. False alarms may occur with ...
.Bukowski, R. W. ''et al''. (2007Performance of Home Smoke Alarms Analysis of the Response of Several Available Technologies in Residential Fire Settings
, NIST Technical Note 1455-1
Radionuclide
As 241Am has a roughly similar half-life to 238Pu (432.2 years vs. 87 years), it has been proposed as an active element ofradioisotope thermoelectric generator
A radioisotope thermoelectric generator (RTG, RITEG), sometimes referred to as a radioisotope power system (RPS), is a type of nuclear battery that uses an array of thermocouples to convert the heat released by the decay of a suitable radioa ...
s, for example in spacecraft.Basic elements of static RTGs, G.L. Kulcinski, NEEP 602 Course Notes (Spring 2000), Nuclear Power in Space, University of Wisconsin Fusion Technology Institute (see last page) Although americium produces less heat and electricity – the power yield is 114.7 mW/g for 241Am and 6.31 mW/g for 243Am (cf. 390 mW/g for 238Pu) – and its radiation poses more threat to humans owing to neutron emission, the
European Space Agency
, owners =
, headquarters = Paris, Île-de-France, France
, coordinates =
, spaceport = Guiana Space Centre
, seal = File:ESA emblem seal.png
, seal_size = 130px
, image = Views in the Main Control Room (120 ...
is considering using americium for its space probes.
Another proposed space-related application of americium is a fuel for space ships with nuclear propulsion. It relies on the very high rate of nuclear fission of 242mAm, which can be maintained even in a micrometer-thick foil. Small thickness avoids the problem of self-absorption of emitted radiation. This problem is pertinent to uranium or plutonium rods, in which only surface layers provide alpha-particles. The fission products of 242mAm can either directly propel the spaceship or they can heat a thrusting gas. They can also transfer their energy to a fluid and generate electricity through a magnetohydrodynamic generator.
One more proposal which utilizes the high nuclear fission rate of 242mAm is a nuclear battery. Its design relies not on the energy of the emitted by americium alpha particles, but on their charge, that is the americium acts as the self-sustaining "cathode". A single 3.2 kg 242mAm charge of such battery could provide about 140 kW of power over a period of 80 days. Even with all the potential benefits, the current applications of 242mAm are as yet hindered by the scarcity and high price of this particular nuclear isomer.
In 2019, researchers at the UK National Nuclear Laboratory and the University of Leicester demonstrated the use of heat generated by americium to illuminate a small light bulb. This technology could lead to systems to power missions with durations up to 400 years into interstellar space, where solar panels do not function.
Neutron source
The oxide of 241Am pressed withberyllium
Beryllium is a chemical element with the symbol Be and atomic number 4. It is a steel-gray, strong, lightweight and brittle alkaline earth metal. It is a divalent element that occurs naturally only in combination with other elements to for ...
is an efficient neutron source. Here americium acts as the alpha source, and beryllium produces neutrons owing to its large cross-section for the (α,n) nuclear reaction:
: Production of other elements
Americium is a starting material for the production of other transuranic elements andtransactinide
Superheavy elements, also known as transactinide elements, transactinides, or super-heavy elements, are the chemical elements with atomic number greater than 103. The superheavy elements are those beyond the actinides in the periodic table; the l ...
s – for example, 82.7% of 242Am decays to 242Cm and 17.3% to 242Pu. In the nuclear reactor, 242Am is also up-converted by neutron capture to 243Am and 244Am, which transforms by β-decay to 244Cm:
: berkelium
Berkelium is a transuranic radioactive chemical element with the symbol Bk and atomic number 97. It is a member of the actinide and transuranium element series. It is named after the city of Berkeley, California, the location of the Lawrence B ...
(243Bk isotope) had been first intentionally produced and identified by bombarding 241Am with alpha particles, in 1949, by the same Berkeley group, using the same 60-inch cyclotron. Similarly, nobelium was produced at the Joint Institute for Nuclear Research
The Joint Institute for Nuclear Research (JINR, russian: Объединённый институт ядерных исследований, ОИЯИ), in Dubna, Moscow Oblast (110 km north of Moscow), Russia, is an international research c ...
, Dubna
Dubna ( rus, Дубна́, p=dʊbˈna) is a town in Moscow Oblast, Russia. It has a status of ''naukograd'' (i.e. town of science), being home to the Joint Institute for Nuclear Research, an international nuclear physics research center and one o ...
, Russia, in 1965 in several reactions, one of which included irradiation of 243Am with 15N ions. Besides, one of the synthesis reactions for lawrencium, discovered by scientists at Berkeley and Dubna, included bombardment of 243Am with 18O.
Spectrometer
Americium-241 has been used as a portable source of both gamma rays and alpha particles for a number of medical and industrial uses. The 59.5409 keV gamma ray emissions from 241Am in such sources can be used for indirect analysis of materials inradiography
Radiography is an imaging technique using X-rays, gamma rays, or similar ionizing radiation and non-ionizing radiation to view the internal form of an object. Applications of radiography include medical radiography ("diagnostic" and "therapeu ...
and X-ray fluorescence spectroscopy, as well as for quality control in fixed nuclear density gauges and nuclear densometers. For example, the element has been employed to gauge glass
Glass is a non- crystalline, often transparent, amorphous solid that has widespread practical, technological, and decorative use in, for example, window panes, tableware, and optics. Glass is most often formed by rapid cooling (quenchin ...
thickness to help create flat glass. Americium-241 is also suitable for calibration of gamma-ray spectrometers in the low-energy range, since its spectrum consists of nearly a single peak and negligible Compton continuum (at least three orders of magnitude lower intensity). Americium-241 gamma rays were also used to provide passive diagnosis of thyroid function. This medical application is however obsolete.
Health concerns
As a highly radioactive element, americium and its compounds must be handled only in an appropriate laboratory under special arrangements. Although most americium isotopes predominantly emit alpha particles which can be blocked by thin layers of common materials, many of the daughter products emit gamma-rays and neutrons which have a long penetration depth. If consumed, most of the americium is excreted within a few days, with only 0.05% absorbed in the blood, of which roughly 45% goes to theliver
The liver is a major organ only found in vertebrates which performs many essential biological functions such as detoxification of the organism, and the synthesis of proteins and biochemicals necessary for digestion and growth. In humans, it i ...
and 45% to the bones, and the remaining 10% is excreted. The uptake to the liver depends on the individual and increases with age. In the bones, americium is first deposited over cortical and trabecular surfaces and slowly redistributes over the bone with time. The biological half-life of 241Am is 50 years in the bones and 20 years in the liver, whereas in the gonads (testicles and ovaries) it remains permanently; in all these organs, americium promotes formation of cancer cells as a result of its radioactivity.
Americium often enters landfills from discarded smoke detector
A smoke detector is a device that senses smoke, typically as an indicator of fire. Smoke detectors are usually housed in plastic enclosures, typically shaped like a disk about in diameter and thick, but shape and size vary. Smoke can be detecte ...
s. The rules associated with the disposal of smoke detectors are relaxed in most jurisdictions. In 1994, 17-year-old David Hahn extracted the americium from about 100 smoke detectors in an attempt to build a breeder nuclear reactor.Ken Silverstein
Ken Silverstein is an American journalist who worked for the '' Los Angeles Times'' as an investigative reporter, for The Associated Press in Brazil, and has written for ''Mother Jones'', ''Washington Monthly'', '' The Nation'', ''Slate'', ...
The Radioactive Boy Scout: When a teenager attempts to build a breeder reactor
'' Harper's Magazine'', November 1998 There have been a few cases of exposure to americium, the worst case being that of chemical operations technician Harold McCluskey, who at the age of 64 was exposed to 500 times the occupational standard for americium-241 as a result of an explosion in his lab. McCluskey died at the age of 75 of unrelated pre-existing disease.
See also
* Actinides in the environment * :Americium compoundsNotes
References
Bibliography
* * Penneman, R. A. and Keenan T. KThe radiochemistry of americium and curium
University of California, Los Alamos, California, 1960 *
Further reading
* ''Nuclides and Isotopes – 14th Edition'', GE Nuclear Energy, 1989. * *External links
at '' The Periodic Table of Videos'' (University of Nottingham)
ATSDR – Public Health Statement: Americium
{{Authority control Chemical elements Chemical elements with double hexagonal close-packed structure Actinides Carcinogens Synthetic elements