
Amino acids are
organic compounds that contain both
amino
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such ...
and
carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise
proteins. Only 22 alpha amino acids appear in the
genetic code
The genetic code is the set of rules used by living cells to translate information encoded within genetic material ( DNA or RNA sequences of nucleotide triplets, or codons) into proteins. Translation is accomplished by the ribosome, which links ...
.
Amino acids can be classified according to the locations of the core structural functional groups, as Alpha and beta carbon, alpha-
(α-), beta-
(β-), gamma-
(γ-) or delta-
(δ-) amino acids; other categories relate to Chemical polarity, polarity, ionization, and side chain group type (aliphatic, Open-chain compound, acyclic, aromatic, containing hydroxyl or
sulfur, etc.). In the form of proteins, amino acid ''
residues'' form the second-largest component (
water being the largest) of human
muscles and other
tissues. Beyond their role as residues in proteins, amino acids participate in a number of processes such as
neurotransmitter transport and
biosynthesis
Biosynthesis is a multi-step, enzyme- catalyzed process where substrates are converted into more complex products in living organisms. In biosynthesis, simple compounds are modified, converted into other compounds, or joined to form macromolecule ...
. It is thought that they played a key role in
enabling life on Earth and its emergence.
Amino acids are formally named by the IUPAC-IUBMB Joint Commission on Biochemical Nomenclature
in terms of the fictitious "neutral" structure shown in the illustration. For example, the systematic name of alanine is 2-aminopropanoic acid, based on the formula . The Commission justified this approach as follows:
The systematic names and formulas given refer to hypothetical forms in which amino groups are unprotonated and carboxyl groups are undissociated. This convention is useful to avoid various nomenclatural problems but should not be taken to imply that these structures represent an appreciable fraction of the amino-acid molecules.
History
The first few amino acids were discovered in the early 1800s. In 1806, French chemists
Louis-Nicolas Vauquelin
Prof. Louis Nicolas Vauquelin FRS(For) HFRSE (16 May 1763 – 14 November 1829) was a French pharmacist and chemist. He was the discoverer of both chromium and beryllium.
Early life
Vauquelin was born at Saint-André-d'Hébertot in Normandy, Fr ...
and
Pierre Jean Robiquet
Pierre Jean Robiquet (13 January 1780 – 29 April 1840) was a French chemist. He laid founding work in identifying amino acids, the fundamental building blocks of proteins. He did this through recognizing the first of them, asparagine, in 180 ...
isolated a compound from
asparagus that was subsequently named
asparagine
Asparagine (symbol Asn or N) is an α-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated −NH form under biological conditions), an α-carboxylic acid group (which is in the depro ...
, the first amino acid to be discovered.
Cystine
Cystine is the oxidized derivative of the amino acid cysteine and has the formula (SCH2CH(NH2)CO2H)2. It is a white solid that is poorly soluble in water. As a residue in proteins, cystine serves two functions: a site of redox reactions and a mec ...
was discovered in 1810, although its monomer,
cysteine, remained undiscovered until 1884.
[ ]Glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid ( carbamic acid is unstable), with the chemical formula NH2‐ CH2‐ COOH. Glycine is one of the proteinogen ...
and leucine
Leucine (symbol Leu or L) is an essential amino acid that is used in the biosynthesis of proteins. Leucine is an α-amino acid, meaning it contains an α-amino group (which is in the protonated −NH3+ form under biological conditions), an α- c ...
were discovered in 1820. The last of the 20 common amino acids to be discovered was threonine in 1935 by William Cumming Rose
William Cumming Rose (April 4, 1887 – September 25, 1985) was an American biochemist and nutritionist. He discovered the amino acid threonine, and his research determined the necessity for essential amino acids in diet and the minimum daily re ...
, who also determined the essential amino acid
An essential amino acid, or indispensable amino acid, is an amino acid that cannot be synthesized from scratch by the organism fast enough to supply its demand, and must therefore come from the diet. Of the 21 amino acids common to all life form ...
s and established the minimum daily requirements of all amino acids for optimal growth.
The unity of the chemical category was recognized by Wurtz in 1865, but he gave no particular name to it. The first use of the term "amino acid" in the English language dates from 1898, while the German term, , was used earlier. Proteins were found to yield amino acids after enzymatic digestion or acid hydrolysis. In 1902, Emil Fischer
Hermann Emil Louis Fischer (; 9 October 1852 – 15 July 1919) was a German chemist and 1902 recipient of the Nobel Prize in Chemistry. He discovered the Fischer esterification. He also developed the Fischer projection, a symbolic way of dra ...
and Franz Hofmeister
Franz Hofmeister (30 August 1850, in Prague – 26 July 1922, in Würzburg) was an early protein scientist, and is famous for his studies of salts that influence the solubility and conformational stability of proteins. In 1902, Hofmeister became ...
independently proposed that proteins are formed from many amino acids, whereby bonds are formed between the amino group of one amino acid with the carboxyl group of another, resulting in a linear structure that Fischer termed " peptide".
General structure
 In the structure shown at the top of the page, R represents a side chain specific to each amino acid. The carbon atom next to the
In the structure shown at the top of the page, R represents a side chain specific to each amino acid. The carbon atom next to the carboxyl group
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is or , with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic ...
is called the α–carbon. Amino acids containing an amino group
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such ...
bonded directly to the α-carbon are referred to as ''α-amino acids''. These include proline and hydroxyproline, which are secondary amine
In chemistry, amines (, ) are compounds and functional groups that contain a basic nitrogen atom with a lone pair. Amines are formally derivatives of ammonia (), wherein one or more hydrogen atoms have been replaced by a substituent such ...
s. In the past they were often called ''imino acids'', a misnomer because they do not contain an imine grouping .[Retrieved 2 April 2012] The obsolete term remains frequent.
Isomerism
The common natural forms of amino acids have the structure ( in the case of proline) and functional groups attached to the same C atom, and are thus α-amino acids. With the exception of achiral glycine, natural amino acids have the L configuration,glyceraldehyde
Glyceraldehyde (glyceral) is a triose monosaccharide with chemical formula C3 H6 O3. It is the simplest of all common aldoses. It is a sweet, colorless, crystalline solid that is an intermediate compound in carbohydrate metabolism. The word comes ...
from which that amino acid can, in theory, be synthesized (D-glyceraldehyde is dextrorotatory; L-glyceraldehyde is levorotatory).
An alternative convention is to use the (''S'') and (''R'') designators to specify the ''absolute configuration''.chiral
Chirality is a property of asymmetry important in several branches of science. The word ''chirality'' is derived from the Greek (''kheir''), "hand", a familiar chiral object.
An object or a system is ''chiral'' if it is distinguishable from i ...
. Cysteine has its side chain in the same geometric location as the other amino acids, but the ''R''/''S'' terminology is reversed because sulfur has higher atomic number compared to the carboxyl oxygen which gives the side chain a higher priority by the Cahn-Ingold-Prelog sequence rules, whereas the atoms in most other side chains give them lower priority compared to the carboxyl group.
Side chains
Amino acids are designated as α- when the amino nitrogen atom is attached to the α-carbon, the carbon atom adjacent to the carboxylate group.
In all cases below in this section the values (if any) refer to the ionization of the groups as amino acid residues in proteins. They are not values for the free amino acids (which are of little biochemical importance).
Aliphatic side-chains
 Seven (of the 21 proteinogenic) amino acids have side-chains that contain only H and C. These, therefore, do not ionize. They are as follows (with three- and one-letter symbols in parentheses):
*
Seven (of the 21 proteinogenic) amino acids have side-chains that contain only H and C. These, therefore, do not ionize. They are as follows (with three- and one-letter symbols in parentheses):
*Glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid ( carbamic acid is unstable), with the chemical formula NH2‐ CH2‐ COOH. Glycine is one of the proteinogen ...
(Gly, G):
*Alanine
Alanine (symbol Ala or A), or α-alanine, is an α-amino acid that is used in the biosynthesis of proteins. It contains an amine group and a carboxylic acid group, both attached to the central carbon atom which also carries a methyl group side ...
(Ala, A):
* Valine (Val, V):
* Isoleucine (Ile, I):
*Leucine
Leucine (symbol Leu or L) is an essential amino acid that is used in the biosynthesis of proteins. Leucine is an α-amino acid, meaning it contains an α-amino group (which is in the protonated −NH3+ form under biological conditions), an α- c ...
(Leu, L):
* Phenylalanine (Phe, F):
* Proline (Pro, P): cyclized onto the amine
Polar neutral side-chains
Two amino acids contain alcohol side chains. These do not ionize in normal conditions, though one, serine, becomes deprotonated during the catalysis by serine proteases: this is an example of severe perturbation, and is not characteristic of serine residues in general.
* Serine (Ser, S, no when not severely perturbed):
* Threonine (Thr, T, no ):
Threonine has two chiral centers, not only the L (2''S'') chiral center at the α-carbon shared by all amino acids apart from achiral glycine, but also (3''R'') at the β-carbon. The full stereochemical specification is L-threonine (2''S'',3''R'').
Amide side-chains
Two amino acids have amide side-chains, as follows:
*Asparagine
Asparagine (symbol Asn or N) is an α-amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated −NH form under biological conditions), an α-carboxylic acid group (which is in the depro ...
(Asn, N):
* Glutamine (Gln, Q):
These side-chains do not ionize in the normal range of pH.
Sulfur-containing side-chains
Two side-chains contain sulfur atoms, of which one ionizes in the normal range (with indicated) and the other does not:
* Cysteine (Cys, C, ):
* Methionine (Met, M, no ):
Aromatic side-chains
Three amino acids have aromatic ring structures as side-chains, as illustrated. Of these, tyrosine ionizes in the normal range; the other two do not).
* Phenylalanine (Phe, F, no ): left in the illustration
* Tyrosine (Tyr, Y, ): middle in the illustration
*Tryptophan
Tryptophan (symbol Trp or W)
is an α-amino acid that is used in the biosynthesis of proteins. Tryptophan contains an α-amino group, an α-carboxylic acid group, and a side chain indole, making it a polar molecule with a non-polar aromatic ...
(Trp, W, no ): right in the illustration
Anionic side-chains
Two amino acids have side-chains that are anions at ordinary pH. These amino acids are often referred to as if carboxylic acids but are more correctly called carboxylates, as they are deprotonated at most relevant pH values. The anionic carboxylate groups behave as Brønsted bases in all circumstances except for enzymes like pepsin
Pepsin is an endopeptidase that breaks down proteins into smaller peptides. It is produced in the gastric chief cells of the stomach lining and is one of the main digestive enzymes in the digestive systems of humans and many other animals, ...
that act in environments of very low pH like the mammalian stomach.
*Aspartate
Aspartic acid (symbol Asp or D; the ionic form is known as aspartate), is an α-amino acid that is used in the biosynthesis of proteins. Like all other amino acids, it contains an amino group and a carboxylic acid. Its α-amino group is in the pro ...
("aspartic acid", Asp, D, ):
* Glutamate ("glutamic acid", Glu, E, ):
Cationic side-chains
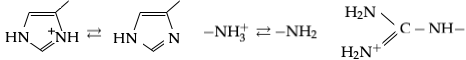 There are three amino acids with side-chains that are cations at neutral pH (though in one, histidine, cationic and neutral forms both exist). They are commonly called ''basic amino acids'', but this term is misleading: histidine can act both as a Brønsted acid and as a Brønsted base at neutral pH, lysine acts as a Brønsted acid, and arginine has a fixed positive charge and does not ionize in neutral conditions. The names ''histidinium, lysinium'' and ''argininium'' would be more accurate names for the structures, but have essentially no currency.
*
There are three amino acids with side-chains that are cations at neutral pH (though in one, histidine, cationic and neutral forms both exist). They are commonly called ''basic amino acids'', but this term is misleading: histidine can act both as a Brønsted acid and as a Brønsted base at neutral pH, lysine acts as a Brønsted acid, and arginine has a fixed positive charge and does not ionize in neutral conditions. The names ''histidinium, lysinium'' and ''argininium'' would be more accurate names for the structures, but have essentially no currency.
*Histidine
Histidine (symbol His or H) is an essential amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated –NH3+ form under biological conditions), a carboxylic acid group (which is in the d ...
(His, H, ): Protonated and deprotonated forms in equilibrium are shown at the left of the image
*Lysine
Lysine (symbol Lys or K) is an α-amino acid that is a precursor to many proteins. It contains an α-amino group (which is in the protonated form under biological conditions), an α-carboxylic acid group (which is in the deprotonated −CO ...
(Lys, K, ): Shown in the middle of the image
*Arginine
Arginine is the amino acid with the formula (H2N)(HN)CN(H)(CH2)3CH(NH2)CO2H. The molecule features a guanidino group appended to a standard amino acid framework. At physiological pH, the carboxylic acid is deprotonated (−CO2−) and both the a ...
(Arg, R, ): Shown at the right of the image
β- and γ-amino acids
Amino acids with the structure , such as β-alanine, a component of carnosine
Carnosine (''beta''-alanyl-L-histidine) is a dipeptide molecule, made up of the amino acids beta-alanine and histidine. It is highly concentrated in muscle and brain tissues. Carnosine was discovered by Russian chemist Vladimir Gulevich.
Ca ...
and a few other peptides, are β-amino acids. Ones with the structure are γ-amino acids, and so on, where X and Y are two substituents (one of which is normally H).
Zwitterions
 In aqueous solution at pH close to neutrality, amino acids exist as zwitterions, i.e. as dipolar ions with both and in charged states, so the overall structure is . At physiological pH the so-called "neutral forms" are not present to any measurable degree. Although the two charges in the zwitterion structure add up to zero it is misleading to call a species with a net charge of zero "uncharged".
In strongly acidic conditions (pH below 3), the carboxylate group becomes protonated and the structure becomes an ammonio carboxylic acid, . This is relevant for enzymes like pepsin that are active in acidic environments such as the mammalian stomach and
In aqueous solution at pH close to neutrality, amino acids exist as zwitterions, i.e. as dipolar ions with both and in charged states, so the overall structure is . At physiological pH the so-called "neutral forms" are not present to any measurable degree. Although the two charges in the zwitterion structure add up to zero it is misleading to call a species with a net charge of zero "uncharged".
In strongly acidic conditions (pH below 3), the carboxylate group becomes protonated and the structure becomes an ammonio carboxylic acid, . This is relevant for enzymes like pepsin that are active in acidic environments such as the mammalian stomach and lysosomes
A lysosome () is a membrane-bound organelle found in many animal cells. They are spherical vesicles that contain hydrolytic enzymes that can break down many kinds of biomolecules. A lysosome has a specific composition, of both its membrane pro ...
, but does not significantly apply to intracellular enzymes. In highly basic conditions (pH greater than 10, not normally seen in physiological conditions), the ammonio group is deprotonated to give .
Although various definitions of acids and bases are used in chemistry, the only one that is useful for chemistry in aqueous solution is that of Brønsted: an acid is a species that can donate a proton to another species, and a base is one that can accept a proton. This criterion is used to label the groups in the above illustration. Notice that aspartate and glutamate are the principal groups that act as Brønsted bases, and the common references to these as ''acidic amino acids'' (together with the C terminal) is completely wrong and misleading. Likewise the so-called ''basic amino acids'' include one (histidine) that acts as both a Brønsted acid and a base, one (lysine) that acts primarily as a Brønsted acid, and one (arginine) that is normally irrelevant to acid-base behavior as it has a fixed positive charge. In addition, tyrosine and cysteine, which act primarily as acids at neutral pH, are usually forgotten in the usual classification.
Isoelectric point
 For amino acids with uncharged side-chains the zwitterion predominates at pH values between the two p''K''a values, but coexists in equilibrium with small amounts of net negative and net positive ions. At the midpoint between the two p''K''a values, the trace amount of net negative and trace of net positive ions balance, so that average net charge of all forms present is zero. This pH is known as the
For amino acids with uncharged side-chains the zwitterion predominates at pH values between the two p''K''a values, but coexists in equilibrium with small amounts of net negative and net positive ions. At the midpoint between the two p''K''a values, the trace amount of net negative and trace of net positive ions balance, so that average net charge of all forms present is zero. This pH is known as the isoelectric point
The isoelectric point (pI, pH(I), IEP), is the pH at which a molecule carries no net electrical charge or is electrically neutral in the statistical mean. The standard nomenclature to represent the isoelectric point is pH(I). However, pI is also ...
p''I'', so p''I'' = (p''K''a1 + p''K''a2).
For amino acids with charged side chains, the p''K''a of the side chain is involved. Thus for aspartate or glutamate with negative side chains, the terminal amino group is essentially entirely in the charged form , but this positive charge needs to be balanced by the state with just one C-terminal carboxylate group is negatively charged. This occurs halfway between the two carboxylate p''K''a values: p''I'' = (p''K''a1 + p''K''a(R)), where p''K''a(R) is the side chain p''K''a.
Similar considerations apply to other amino acids with ionizable side-chains, including not only glutamate (similar to aspartate), but also
cysteine, histidine, lysine, tyrosine and arginine with positive side chains
Amino acids have zero mobility in electrophoresis at their isoelectric point, although this behaviour is more usually exploited for peptides and proteins than single amino acids. Zwitterions have minimum solubility at their isoelectric point, and some amino acids (in particular, with nonpolar side chains) can be isolated by precipitation from water by adjusting the pH to the required isoelectric point.
Physicochemical properties of amino acids
The ca. 20 canonical amino acids can be classified according to their properties. Important factors are charge, hydrophilicity or hydrophobicity
In chemistry, hydrophobicity is the physical property of a molecule that is seemingly repelled from a mass of water (known as a hydrophobe). In contrast, hydrophiles are attracted to water.
Hydrophobic molecules tend to be nonpolar and, t ...
, size, and functional groups.protein structure
Protein structure is the three-dimensional arrangement of atoms in an amino acid-chain molecule. Proteins are polymers specifically polypeptides formed from sequences of amino acids, the monomers of the polymer. A single amino acid monomer ma ...
and protein–protein interactions. The water-soluble proteins tend to have their hydrophobic residues (Leu
Leu may refer to:
Businesses and organisations
* LEU, NYSE American stock symbol for Centrus Energy Corp.
* London Ecology Unit, a former body (1986-2000) which advised London boroughs on environmental matters
* Free and Equal (''LeU - Liberi e ...
, Ile, Val
Val may refer to: Val-a
Film
* ''Val'' (film), an American documentary about Val Kilmer, directed by Leo Scott and Ting Poo
Military equipment
* Aichi D3A, a Japanese World War II dive bomber codenamed "Val" by the Allies
* AS Val, a Sov ...
, Phe, and Trp) buried in the middle of the protein, whereas hydrophilic side chains are exposed to the aqueous solvent. (Note that in biochemistry
Biochemistry or biological chemistry is the study of chemical processes within and relating to living organisms. A sub-discipline of both chemistry and biology, biochemistry may be divided into three fields: structural biology, enzymology and ...
, a residue refers to a specific monomer within the polymeric chain of a polysaccharide, protein or nucleic acid.) The integral membrane protein
An integral, or intrinsic, membrane protein (IMP) is a type of membrane protein that is permanently attached to the biological membrane. All ''transmembrane proteins'' are IMPs, but not all IMPs are transmembrane proteins. IMPs comprise a signif ...
s tend to have outer rings of exposed hydrophobic
In chemistry, hydrophobicity is the physical property of a molecule that is seemingly repelled from a mass of water (known as a hydrophobe). In contrast, hydrophiles are attracted to water.
Hydrophobic molecules tend to be nonpolar and, t ...
amino acids that anchor them into the lipid bilayer. Some peripheral membrane proteins have a patch of hydrophobic amino acids on their surface that locks onto the membrane. In similar fashion, proteins that have to bind to positively charged molecules have surfaces rich with negatively charged amino acids like glutamate and aspartate
Aspartic acid (symbol Asp or D; the ionic form is known as aspartate), is an α-amino acid that is used in the biosynthesis of proteins. Like all other amino acids, it contains an amino group and a carboxylic acid. Its α-amino group is in the pro ...
, while proteins binding to negatively charged molecules have surfaces rich with positively charged chains like lysine
Lysine (symbol Lys or K) is an α-amino acid that is a precursor to many proteins. It contains an α-amino group (which is in the protonated form under biological conditions), an α-carboxylic acid group (which is in the deprotonated −CO ...
and arginine
Arginine is the amino acid with the formula (H2N)(HN)CN(H)(CH2)3CH(NH2)CO2H. The molecule features a guanidino group appended to a standard amino acid framework. At physiological pH, the carboxylic acid is deprotonated (−CO2−) and both the a ...
. For example, lysine and arginine are highly enriched in low-complexity regions of nucleic-acid binding proteins.hydrophobicity scale Hydrophobicity scales are values that define the relative hydrophobicity or hydrophilicity of amino acid residues. The more positive the value, the more hydrophobic are the amino acids located in that region of the protein. These scales are commonly ...
s of amino acid residues.
Some amino acids have special properties such as cysteine, that can form covalent disulfide bond
In biochemistry, a disulfide (or disulphide in British English) refers to a functional group with the structure . The linkage is also called an SS-bond or sometimes a disulfide bridge and is usually derived by the coupling of two thiol groups. In ...
s to other cysteine residues, proline that forms a cycle to the polypeptide backbone, and glycine that is more flexible than other amino acids.
Furthermore, glycine and proline are highly enriched within low complexity regions of eukaryotic and prokaryotic proteins, whereas the opposite (under-represented) has been observed for highly reactive, or complex, or hydrophobic amino acids, such as cysteine, phenylalanine, tryptophan, methionine, valine, leucine, isoleucine.
Table of standard amino acid abbreviations and properties
Although one-letter symbols are included in the table, IUPAC–IUBMB recommend[ that "Use of the one-letter symbols should be restricted to the comparison of long sequences".
Two additional amino acids are in some species coded for by ]codons
The genetic code is the set of rules used by living cells to translate information encoded within genetic material ( DNA or RNA sequences of nucleotide triplets, or codons) into proteins. Translation is accomplished by the ribosome, which links ...
that are usually interpreted as stop codon
In molecular biology (specifically protein biosynthesis), a stop codon (or termination codon) is a codon (nucleotide triplet within messenger RNA) that signals the termination of the translation process of the current protein. Most codons in m ...
s:
In addition to the specific amino acid codes, placeholders are used in cases where chemical or crystallographic analysis of a peptide or protein cannot conclusively determine the identity of a residue. They are also used to summarise conserved protein sequence motifs. The use of single letters to indicate sets of similar residues is similar to the use of abbreviation codes for degenerate bases.
Unk is sometimes used instead of Xaa, but is less standard.
Ter or * (from termination) is used in notation for mutations in proteins when a stop codon occurs. It correspond to no amino acid at all.Bortezomib
Bortezomib, sold under the brand name Velcade among others, is an anti-cancer medication used to treat multiple myeloma and mantle cell lymphoma. This includes multiple myeloma in those who have and have not previously received treatment. It is ...
and MG132, are artificially synthesized and retain their protecting groups, which have specific codes. Bortezomib is Pyz–Phe–boroLeu, and MG132 is Z–Leu–Leu–Leu–al. To aid in the analysis of protein structure, photo-reactive amino acid analog Photo-reactive amino acid analogs are artificial analogs of natural amino acids that can be used for crosslinking of protein complexes. Photo-reactive amino acid analogs may be incorporated into proteins and peptides ''in vivo'' or in ''vitro''. Pho ...
s are available. These include photoleucine (pLeu) and photomethionine (pMet).
Occurrence and functions in biochemistry
Amino acids which have the amine group attached to the (alpha-) carbon atom next to the carboxyl group have primary importance in living organisms since they participate in protein synthesis.stereoisomers
In stereochemistry, stereoisomerism, or spatial isomerism, is a form of isomerism in which molecules have the same molecular formula and sequence of bonded atoms (constitution), but differ in the three-dimensional orientations of their atoms in ...
("left-handed" enantiomer
In chemistry, an enantiomer ( /ɪˈnænti.əmər, ɛ-, -oʊ-/ ''ih-NAN-tee-ə-mər''; from Ancient Greek ἐνάντιος ''(enántios)'' 'opposite', and μέρος ''(méros)'' 'part') – also called optical isomer, antipode, or optical ant ...
s), although a few D-amino acids ("right-handed") occur in bacterial envelopes, as a neuromodulator (D- serine), and in some antibiotic
An antibiotic is a type of antimicrobial substance active against bacteria. It is the most important type of antibacterial agent for fighting bacterial infections, and antibiotic medications are widely used in the treatment and prevention ...
s.
Many proteinogenic and non-proteinogenic amino acids have biological functions. For example, in the human brain, glutamate (standard glutamic acid
Glutamic acid (symbol Glu or E; the ionic form is known as glutamate) is an α-amino acid that is used by almost all living beings in the biosynthesis of proteins. It is a non-essential nutrient for humans, meaning that the human body can syn ...
) and gamma-aminobutyric acid ("GABA", nonstandard gamma-amino acid) are, respectively, the main excitatory and inhibitory neurotransmitters.Glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid ( carbamic acid is unstable), with the chemical formula NH2‐ CH2‐ COOH. Glycine is one of the proteinogen ...
is a biosynthetic precursor to porphyrins used in red blood cells. Carnitine
Carnitine is a quaternary ammonium compound involved in metabolism in most mammals, plants, and some bacteria. In support of energy metabolism, carnitine transports long-chain fatty acids into mitochondria to be oxidized for energy production, an ...
is used in lipid transport. Nine proteinogenic amino acids are called " essential" for humans because they cannot be produced from other compounds by the human body and so must be taken in as food. Others may be conditionally essential for certain ages or medical conditions. Essential amino acids may also vary from species
In biology, a species is the basic unit of Taxonomy (biology), classification and a taxonomic rank of an organism, as well as a unit of biodiversity. A species is often defined as the largest group of organisms in which any two individuals of ...
to species. Because of their biological significance, amino acids are important in nutrition and are commonly used in nutritional supplement
A dietary supplement is a manufactured product intended to supplement one's diet by taking a pill, capsule, tablet, powder, or liquid. A supplement can provide nutrients either extracted from food sources or that are synthetic in order ...
s, fertilizers, feed, and food technology. Industrial uses include the production of drugs, biodegradable plastic
Biodegradable plastics are plastics that can be decomposed by the action of living organisms, usually microbes, into water, carbon dioxide, and biomass. Biodegradable plastics are commonly produced with renewable raw materials, micro-organisms, ...
s, and chiral catalysts.
Proteinogenic amino acids
Amino acids are the precursors to proteins. They join by condensation reactions to form short polymer chains called peptides or longer chains called either polypeptides or proteins. These chains are linear and unbranched, with each amino acid residue within the chain attached to two neighboring amino acids. In Nature, the process of making proteins encoded by DNA/RNA genetic material is called '' translation'' and involves the step-by-step addition of amino acids to a growing protein chain by a ribozyme that is called a ribosome. The order in which the amino acids are added is read through the genetic code
The genetic code is the set of rules used by living cells to translate information encoded within genetic material ( DNA or RNA sequences of nucleotide triplets, or codons) into proteins. Translation is accomplished by the ribosome, which links ...
from an mRNA template, which is an RNA copy of one of the organism's genes.
Twenty-two amino acids are naturally incorporated into polypeptides and are called proteinogenic or natural amino acids.SECIS element
In biology, the SECIS element (SECIS: ''selenocysteine insertion sequence'') is an RNA element around 60 nucleotides in length that adopts a stem-loop structure. This structural motif (pattern of nucleotides) directs the cell to translate UGA ...
, which causes the UGA codon to encode selenocysteine instead of a stop codon. Pyrrolysine is used by some methanogenic archaea in enzymes that they use to produce methane. It is coded for with the codon UAG, which is normally a stop codon in other organisms. This UAG codon is followed by a PYLIS downstream sequence
In biology, the PYLIS downstream sequence (PYLIS: ''pyrrolysine insertion sequence'') is a stem-loop structure that appears on some mRNA sequences. This structural motif was previously thought to cause the UAG (amber) stop codon to be translated ...
.
Standard vs nonstandard amino acids
The 20 amino acids that are encoded directly by the codons of the universal genetic code are called ''standard'' or ''canonical'' amino acids. A modified form of methionine ( ''N''-formylmethionine) is often incorporated in place of methionine as the initial amino acid of proteins in bacteria, mitochondria and chloroplasts. Other amino acids are called ''nonstandard'' or ''non-canonical''. Most of the nonstandard amino acids are also non-proteinogenic (i.e. they cannot be incorporated into proteins during translation), but two of them are proteinogenic, as they can be incorporated translationally into proteins by exploiting information not encoded in the universal genetic code.
The two nonstandard proteinogenic amino acids are selenocysteine (present in many non-eukaryotes as well as most eukaryotes, but not coded directly by DNA) and pyrrolysine (found only in some archaea and at least one bacterium
Bacteria (; singular: bacterium) are ubiquitous, mostly free-living organisms often consisting of one biological cell. They constitute a large domain of prokaryotic microorganisms. Typically a few micrometres in length, bacteria were among ...
). The incorporation of these nonstandard amino acids is rare. For example, 25 human proteins include selenocysteine in their primary structure, and the structurally characterized enzymes (selenoenzymes) employ selenocysteine as the catalytic moiety
Moiety may refer to:
Chemistry
* Moiety (chemistry), a part or functional group of a molecule
** Moiety conservation, conservation of a subgroup in a chemical species
Anthropology
* Moiety (kinship), either of two groups into which a society is ...
in their active sites. Pyrrolysine and selenocysteine are encoded via variant codons. For example, selenocysteine is encoded by stop codon and SECIS element
In biology, the SECIS element (SECIS: ''selenocysteine insertion sequence'') is an RNA element around 60 nucleotides in length that adopts a stem-loop structure. This structural motif (pattern of nucleotides) directs the cell to translate UGA ...
.chloroplast
A chloroplast () is a type of membrane-bound organelle known as a plastid that conducts photosynthesis mostly in plant and algal cells. The photosynthetic pigment chlorophyll captures the energy from sunlight, converts it, and stores it in ...
s) is generally considered as a form of methionine rather than as a separate proteinogenic amino acid. Codon–tRNA
Transfer RNA (abbreviated tRNA and formerly referred to as sRNA, for soluble RNA) is an adaptor molecule composed of RNA, typically 76 to 90 nucleotides in length (in eukaryotes), that serves as the physical link between the mRNA and the amino a ...
combinations not found in nature can also be used to "expand" the genetic code and form novel proteins known as alloprotein
An alloprotein is a novel synthetic protein containing one or more "non-natural" amino acids. Non-natural in the context means an amino acid either not occurring in nature (novel and synthesised amino acids), s incorporating non-proteinogenic amino acids.
Non-proteinogenic amino acids
Aside from the 22 proteinogenic amino acid
Proteinogenic amino acids are amino acids that are incorporated biosynthetically into proteins during translation. The word "proteinogenic" means "protein creating". Throughout known life, there are 22 genetically encoded (proteinogenic) amino ac ...
s, many ''non-proteinogenic'' amino acids are known. Those either are not found in proteins (for example carnitine
Carnitine is a quaternary ammonium compound involved in metabolism in most mammals, plants, and some bacteria. In support of energy metabolism, carnitine transports long-chain fatty acids into mitochondria to be oxidized for energy production, an ...
, GABA, levothyroxine) or are not produced directly and in isolation by standard cellular machinery (for example, hydroxyproline and selenomethionine).
Non-proteinogenic amino acids that are found in proteins are formed by post-translational modification
Post-translational modification (PTM) is the covalent and generally enzymatic modification of proteins following protein biosynthesis. This process occurs in the endoplasmic reticulum and the golgi apparatus. Proteins are synthesized by ribosom ...
, which is modification after translation during protein synthesis. These modifications are often essential for the function or regulation of a protein. For example, the carboxylation
Carboxylation is a chemical reaction in which a carboxylic acid is produced by treating a substrate with carbon dioxide. The opposite reaction is decarboxylation. In chemistry, the term carbonation is sometimes used synonymously with carboxylation ...
of glutamate allows for better binding of calcium cations, and collagen contains hydroxyproline, generated by hydroxylation of proline. Another example is the formation of hypusine
Hypusine is an uncommon amino acid found in all eukaryotes and in some archaea, but not in bacteria. The only known proteins containing the hypusine residue is eukaryotic translation initiation factor 5A (eIF-5A) and a similar protein found in ar ...
in the translation initiation factor Initiation factors are proteins that bind to the small subunit of the ribosome during the initiation of translation, a part of protein biosynthesis.
Initiation factors can interact with repressors to slow down or prevent translation. They have th ...
EIF5A
Eukaryotic translation initiation factor 5A-1 is a protein that in humans is encoded by the ''EIF5A'' gene.
It is the only known protein to contain the unusual amino acid hypusine 'N''ε-(4-amino-2-hydroxybutyl)-lysine which is synthesized on e ...
, through modification of a lysine residue. Such modifications can also determine the localization of the protein, e.g., the addition of long hydrophobic groups can cause a protein to bind to a phospholipid
Phospholipids, are a class of lipids whose molecule has a hydrophilic "head" containing a phosphate group and two hydrophobic "tails" derived from fatty acids, joined by an alcohol residue (usually a glycerol molecule). Marine phospholipids typ ...
membrane.
Some non-proteinogenic amino acids are not found in proteins. Examples include 2-aminoisobutyric acid
2-Aminoisobutyric acid (also known as α-aminoisobutyric acid, AIB, α-methylalanine, or 2-methylalanine) is the non-proteinogenic amino acid with the structural formula H2N-C(CH3)2-COOH. It is rare in nature, having been only found in meteorites, ...
and the neurotransmitter gamma-aminobutyric acid. Non-proteinogenic amino acids often occur as intermediates in the metabolic pathways for standard amino acids – for example, ornithine
Ornithine is a non-proteinogenic amino acid that plays a role in the urea cycle. Ornithine is abnormally accumulated in the body in ornithine transcarbamylase deficiency. The radical is ornithyl.
Role in urea cycle
L-Ornithine is one of the produc ...
and citrulline
The organic compound citrulline is an α-amino acid. Its name is derived from '' citrullus'', the Latin word for watermelon. Although named and described by gastroenterologists since the late 19th century, it was first isolated from watermelon in ...
occur in the urea cycle, part of amino acid catabolism
Catabolism () is the set of metabolic pathways that breaks down molecules into smaller units that are either oxidized to release energy or used in other anabolic reactions. Catabolism breaks down large molecules (such as polysaccharides, lipids, ...
(see below). A rare exception to the dominance of α-amino acids in biology is the β-amino acid beta alanine (3-aminopropanoic acid), which is used in plants and microorganisms in the synthesis of pantothenic acid
Pantothenic acid, also called vitamin B5 is a water-soluble B vitamin and therefore an essential nutrient. All animals require pantothenic acid in order to synthesize coenzyme A (CoA) – essential for fatty acid metabolism – as well as to, i ...
(vitamin B5), a component of coenzyme A
Coenzyme A (CoA, SHCoA, CoASH) is a coenzyme, notable for its role in the synthesis and oxidation of fatty acids, and the oxidation of pyruvate in the citric acid cycle. All genomes sequenced to date encode enzymes that use coenzyme A as a substr ...
.
In human nutrition
 When taken up into the human body from the diet, the 20 standard amino acids either are used to synthesize proteins, other biomolecules, or are oxidized to
When taken up into the human body from the diet, the 20 standard amino acids either are used to synthesize proteins, other biomolecules, or are oxidized to urea
Urea, also known as carbamide, is an organic compound with chemical formula . This amide has two amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest amide of carbamic acid.
Urea serves an important ...
and carbon dioxide as a source of energy. The oxidation pathway starts with the removal of the amino group by a transaminase; the amino group is then fed into the urea cycle. The other product of transamidation is a keto acid
In organic chemistry, keto acids or ketoacids (also called oxo acids or oxoacids) are organic compounds that contain a carboxylic acid group () and a ketone group ().Franz Dietrich Klingler, Wolfgang Ebertz "Oxocarboxylic Acids" in Ullmann's ...
that enters the citric acid cycle
The citric acid cycle (CAC)—also known as the Krebs cycle or the TCA cycle (tricarboxylic acid cycle)—is a series of chemical reactions to release stored energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and protei ...
. Glucogenic amino acids can also be converted into glucose, through gluconeogenesis. Of the 20 standard amino acids, nine (His
His or HIS may refer to:
Computing
* Hightech Information System, a Hong Kong graphics card company
* Honeywell Information Systems
* Hybrid intelligent system
* Microsoft Host Integration Server
Education
* Hangzhou International School, ...
, Ile, Leu
Leu may refer to:
Businesses and organisations
* LEU, NYSE American stock symbol for Centrus Energy Corp.
* London Ecology Unit, a former body (1986-2000) which advised London boroughs on environmental matters
* Free and Equal (''LeU - Liberi e ...
, Lys, Met, Phe, Thr, Trp and Val
Val may refer to: Val-a
Film
* ''Val'' (film), an American documentary about Val Kilmer, directed by Leo Scott and Ting Poo
Military equipment
* Aichi D3A, a Japanese World War II dive bomber codenamed "Val" by the Allies
* AS Val, a Sov ...
) are called essential amino acid
An essential amino acid, or indispensable amino acid, is an amino acid that cannot be synthesized from scratch by the organism fast enough to supply its demand, and must therefore come from the diet. Of the 21 amino acids common to all life form ...
s because the human body
The human body is the structure of a human being. It is composed of many different types of cells that together create tissues and subsequently organ systems. They ensure homeostasis and the viability of the human body.
It comprises a head ...
cannot synthesize them from other compounds at the level needed for normal growth, so they must be obtained from food. In addition, cysteine, tyrosine, and arginine
Arginine is the amino acid with the formula (H2N)(HN)CN(H)(CH2)3CH(NH2)CO2H. The molecule features a guanidino group appended to a standard amino acid framework. At physiological pH, the carboxylic acid is deprotonated (−CO2−) and both the a ...
are considered semiessential amino acids, and taurine a semiessential aminosulfonic acid in children. The metabolic pathways that synthesize these monomers are not fully developed. The amounts required also depend on the age and health of the individual, so it is hard to make general statements about the dietary requirement for some amino acids. Dietary exposure to the nonstandard amino acid BMAA has been linked to human neurodegenerative diseases, including ALS
Amyotrophic lateral sclerosis (ALS), also known as motor neuron disease (MND) or Lou Gehrig's disease, is a neurodegenerative disease that results in the progressive loss of motor neurons that control voluntary muscles. ALS is the most comm ...
.
Non-protein functions
In humans, non-protein amino acids also have important roles as metabolic intermediates, such as in the biosynthesis of the neurotransmitter gamma-aminobutyric acid (GABA). Many amino acids are used to synthesize other molecules, for example:
* Tryptophan
Tryptophan (symbol Trp or W)
is an α-amino acid that is used in the biosynthesis of proteins. Tryptophan contains an α-amino group, an α-carboxylic acid group, and a side chain indole, making it a polar molecule with a non-polar aromatic ...
is a precursor of the neurotransmitter serotonin.
* Tyrosine (and its precursor phenylalanine) are precursors of the catecholamine
A catecholamine (; abbreviated CA) is a monoamine neurotransmitter, an organic compound that has a catechol (benzene with two hydroxyl side groups next to each other) and a side-chain amine.
Catechol can be either a free molecule or a substi ...
neurotransmitters dopamine, epinephrine
Adrenaline, also known as epinephrine, is a hormone and medication which is involved in regulating visceral functions (e.g., respiration). It appears as a white microcrystalline granule. Adrenaline is normally produced by the adrenal glands and ...
and norepinephrine and various trace amines.
* Phenylalanine is a precursor of phenethylamine and tyrosine in humans. In plants, it is a precursor of various phenylpropanoids, which are important in plant metabolism.
* Glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid ( carbamic acid is unstable), with the chemical formula NH2‐ CH2‐ COOH. Glycine is one of the proteinogen ...
is a precursor of porphyrins such as heme
Heme, or haem (pronounced / hi:m/ ), is a precursor to hemoglobin, which is necessary to bind oxygen in the bloodstream. Heme is biosynthesized in both the bone marrow and the liver.
In biochemical terms, heme is a coordination complex "consist ...
.
* Arginine
Arginine is the amino acid with the formula (H2N)(HN)CN(H)(CH2)3CH(NH2)CO2H. The molecule features a guanidino group appended to a standard amino acid framework. At physiological pH, the carboxylic acid is deprotonated (−CO2−) and both the a ...
is a precursor of nitric oxide
Nitric oxide (nitrogen oxide or nitrogen monoxide) is a colorless gas with the formula . It is one of the principal oxides of nitrogen. Nitric oxide is a free radical: it has an unpaired electron, which is sometimes denoted by a dot in its ch ...
.
* Ornithine
Ornithine is a non-proteinogenic amino acid that plays a role in the urea cycle. Ornithine is abnormally accumulated in the body in ornithine transcarbamylase deficiency. The radical is ornithyl.
Role in urea cycle
L-Ornithine is one of the produc ...
and ''S''-adenosylmethionine are precursors of polyamine
A polyamine is an organic compound having more than two amino groups. Alkyl polyamines occur naturally, but some are synthetic. Alkylpolyamines are colorless, hygroscopic, and water soluble. Near neutral pH, they exist as the ammonium derivatives. ...
s.
* Aspartate
Aspartic acid (symbol Asp or D; the ionic form is known as aspartate), is an α-amino acid that is used in the biosynthesis of proteins. Like all other amino acids, it contains an amino group and a carboxylic acid. Its α-amino group is in the pro ...
, glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid ( carbamic acid is unstable), with the chemical formula NH2‐ CH2‐ COOH. Glycine is one of the proteinogen ...
, and glutamine are precursors of nucleotides.arginine
Arginine is the amino acid with the formula (H2N)(HN)CN(H)(CH2)3CH(NH2)CO2H. The molecule features a guanidino group appended to a standard amino acid framework. At physiological pH, the carboxylic acid is deprotonated (−CO2−) and both the a ...
that is found in many legumes,Canavalia gladiata
''Canavalia gladiata'', the sword bean or scimitar bean, is a domesticated plant species in the legume family Fabaceae. It is used as a vegetable in interior central and south central India, though not commercially farmed. The unripe pods are al ...
'' (sword bean). This amino acid protects the plants from predators such as insects and can cause illness in people if some types of legumes are eaten without processing. The non-protein amino acid mimosine is found in other species of legume, in particular '' Leucaena leucocephala''. This compound is an analogue of tyrosine and can poison animals that graze on these plants.
Uses in industry
Fertilizer
The chelating ability of amino acids is sometimes used in fertilizers to facilitate the delivery of minerals to plants in order to correct mineral deficiencies, such as iron chlorosis. These fertilizers are also used to prevent deficiencies from occurring and to improve the overall health of the plants.
Animal feed
Amino acids are sometimes added to animal feed
Animal feed is food given to domestic animals, especially livestock, in the course of animal husbandry. There are two basic types: fodder and forage. Used alone, the word ''feed'' more often refers to fodder. Animal feed is an important input to ...
because some of the components of these feeds, such as soybeans, have low levels of some of the essential amino acid
An essential amino acid, or indispensable amino acid, is an amino acid that cannot be synthesized from scratch by the organism fast enough to supply its demand, and must therefore come from the diet. Of the 21 amino acids common to all life form ...
s, especially of lysine, methionine, threonine, and tryptophan.
Food
The food industry
The food industry is a complex, global network of diverse businesses that supplies most of the food consumed by the world's population. The food industry today has become highly diversified, with manufacturing ranging from small, traditional, ...
is a major consumer of amino acids, especially glutamic acid
Glutamic acid (symbol Glu or E; the ionic form is known as glutamate) is an α-amino acid that is used by almost all living beings in the biosynthesis of proteins. It is a non-essential nutrient for humans, meaning that the human body can syn ...
, which is used as a flavor enhancer,aspartame
Aspartame is an artificial non-saccharide sweetener 200 times sweeter than sucrose and is commonly used as a sugar substitute in foods and beverages. It is a methyl ester of the aspartic acid/phenylalanine dipeptide with the trade nam ...
(aspartylphenylalanine 1-methyl ester), which is used as an artificial sweetener
A sugar substitute is a food additive that provides a sweetness like that of sugar while containing significantly less food energy than sugar-based sweeteners, making it a zero-calorie () or low-calorie sweetener. Artificial sweeteners may b ...
. Amino acids are sometimes added to food by manufacturers to alleviate symptoms of mineral deficiencies, such as anemia, by improving mineral absorption and reducing negative side effects from inorganic mineral supplementation.
Pharmaceuticals and cosmetics
Similarly, some amino acids derivatives are used in pharmaceutical industry. They include 5-HTP
5-Hydroxytryptophan (5-HTP), also known as oxitriptan, is a naturally occurring amino acid and chemical precursor as well as a metabolic intermediate in the biosynthesis of the neurotransmitter serotonin.
Uses
5-HTP is sold over the counter ...
(5-hydroxytryptophan) used for experimental treatment of depression, L-DOPA (L-dihydroxyphenylalanine) for Parkinson's treatment, and eflornithine
Eflornithine, sold under the brand name Vaniqa among others, is a medication used to treat African trypanosomiasis (sleeping sickness) and excessive hair growth on the face in women. Specifically it is used for the 2nd stage of sleeping sickness ...
drug that inhibits ornithine decarboxylase
The enzyme ornithine decarboxylase (, ODC) catalyzes the decarboxylation of ornithine (a product of the urea cycle) to form putrescine. This reaction is the committed step in polyamine synthesis. In humans, this protein has 461 amino acids and fo ...
and used in the treatment of sleeping sickness
African trypanosomiasis, also known as African sleeping sickness or simply sleeping sickness, is an insect-borne parasitic infection of humans and other animals. It is caused by the species ''Trypanosoma brucei''. Humans are infected by two ty ...
. Amino acids are used in the synthesis of some cosmetics
Cosmetics are constituted mixtures of chemical compounds derived from either natural sources, or synthetically created ones. Cosmetics have various purposes. Those designed for personal care and skin care can be used to cleanse or protect ...
.[
]
Expanded genetic code
Since 2001, 40 non-natural amino acids have been added into protein by creating a unique codon (recoding) and a corresponding transfer-RNA:aminoacyl – tRNA-synthetase pair to encode it with diverse physicochemical and biological properties in order to be used as a tool to exploring protein structure
Protein structure is the three-dimensional arrangement of atoms in an amino acid-chain molecule. Proteins are polymers specifically polypeptides formed from sequences of amino acids, the monomers of the polymer. A single amino acid monomer ma ...
and function or to create novel or enhanced proteins.
Nullomers
Nullomers are codons that in theory code for an amino acid, however, in nature there is a selective bias against using this codon in favor of another, for example bacteria prefer to use CGA instead of AGA to code for arginine. This creates some sequences that do not appear in the genome. This characteristic can be taken advantage of and used to create new selective cancer-fighting drugs and to prevent cross-contamination of DNA samples from crime-scene investigations.
Chemical building blocks
Amino acids are important as low-cost feedstocks. These compounds are used in chiral pool synthesis The chiral pool is a "collection of abundant enantiopure building blocks provided by nature" used in synthesis. In other words, a chiral pool would be a large quantity of common organic enantiomers. Contributors to the chiral pool are amino acids, s ...
as enantiomerically pure building blocks.hydrogenation
Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to reduce or saturate organic c ...
reactions, although no commercial applications exist.
Biodegradable plastics
Amino acids have been considered as components of biodegradable polymers, which have applications as environmentally friendly packaging and in medicine in drug delivery
Drug delivery refers to approaches, formulations, manufacturing techniques, storage systems, and technologies involved in transporting a pharmaceutical compound to its target site to achieve a desired therapeutic effect. Principles related to d ...
and the construction of prosthetic implants.diaper
A diaper /ˈdaɪpə(r)/ ( American and Canadian English) or a nappy (Australian English, British English, and Hiberno-English) is a type of underwear that allows the wearer to urinate or defecate without using a toilet, by absorbing or conta ...
s and agriculture.chelate
Chelation is a type of bonding of ions and molecules to metal ions. It involves the formation or presence of two or more separate coordinate bonds between a polydentate (multiple bonded) ligand and a single central metal atom. These ligands are ...
metal ions, polyaspartate is also being used as a biodegradable antiscaling
Scaling may refer to:
Science and technology
Mathematics and physics
* Scaling (geometry), a linear transformation that enlarges or diminishes objects
* Scale invariance, a feature of objects or laws that do not change if scales of length, energ ...
agent and a corrosion inhibitor
In chemistry, a corrosion inhibitor or anti-corrosive is a chemical compound that, when added to a liquid or gas, decreases the corrosion rate of a material, typically a metal or an alloy, that comes into contact with the fluid. The effectivene ...
.phenols
In organic chemistry, phenols, sometimes called phenolics, are a class of chemical compounds consisting of one or more hydroxyl groups (— O H) bonded directly to an aromatic hydrocarbon group. The simplest is phenol, . Phenolic compounds are ...
such as bisphenol A
Bisphenol A (BPA) is a chemical compound primarily used in the manufacturing of various plastics. It is a colourless solid which is soluble in most common organic solvents, but has very poor solubility in water. BPA is produced on an industrial s ...
in the manufacture of polycarbonates.
Synthesis
Chemical synthesis
The commercial production of amino acids usually relies on mutant bacteria that overproduce individual amino acids using glucose as a carbon source. Some amino acids are produced by enzymatic conversions of synthetic intermediates. 2-Aminothiazoline-4-carboxylic acid
2-Aminothiazoline-4-carboxylic acid (ACTA) is the organosulfur compound and a heterocycle with the formula HO2CCHCH2SCNH2N. This derivative of thiazoline is an intermediate in the industrial synthesis of L- cysteine, an amino acid. ACTA exists i ...
is an intermediate in one industrial synthesis of L-cysteine for example. Aspartic acid
Aspartic acid (symbol Asp or D; the ionic form is known as aspartate), is an α-amino acid that is used in the biosynthesis of proteins. Like all other amino acids, it contains an amino group and a carboxylic acid. Its α-amino group is in the pro ...
is produced by the addition of ammonia to fumarate using a lyase.
Biosynthesis
In plants, nitrogen is first assimilated into organic compounds in the form of glutamate, formed from alpha-ketoglutarate and ammonia in the mitochondrion. For other amino acids, plants use transaminases to move the amino group from glutamate to another alpha-keto acid. For example, aspartate aminotransferase converts glutamate and oxaloacetate to alpha-ketoglutarate and aspartate. Other organisms use transaminases for amino acid synthesis, too.
Nonstandard amino acids are usually formed through modifications to standard amino acids. For example, homocysteine is formed through the transsulfuration pathway
The transsulfuration pathway is a metabolic pathway involving the interconversion of cysteine and homocysteine through the intermediate cystathionine. Two transsulfurylation pathways are known: the ''forward'' and the ''reverse''.
The ''forward ...
or by the demethylation of methionine via the intermediate metabolite ''S''-adenosylmethionine,2-aminoisobutyric acid
2-Aminoisobutyric acid (also known as α-aminoisobutyric acid, AIB, α-methylalanine, or 2-methylalanine) is the non-proteinogenic amino acid with the structural formula H2N-C(CH3)2-COOH. It is rare in nature, having been only found in meteorites, ...
and lanthionine
Lanthionine is a nonproteinogenic amino acid with the chemical formula (HOOC-CH(NH2)-CH2-S-CH2-CH(NH2)-COOH). It is typically formed by a cysteine residue and a dehydrated serine residue. Despite its name, lanthionine does not contain the element ...
, which is a sulfide-bridged derivative of alanine. Both of these amino acids are found in peptidic lantibiotics
Lantibiotics are a class of polycyclic peptide antibiotics that contain the characteristic thioether amino acids lanthionine or methyllanthionine, as well as the unsaturated amino acids dehydroalanine, and 2-aminoisobutyric acid. They belong to ...
such as alamethicin. However, in plants, 1-aminocyclopropane-1-carboxylic acid
1-Aminocyclopropane-1-carboxylic acid (ACC) is a disubstituted cyclic α-amino acid in which a cyclopropane ring is fused to the C atom of the amino acid. It is a white solid. Many cyclopropane-substituted amino acids are known, but this one occ ...
is a small disubstituted cyclic amino acid that is an intermediate in the production of the plant hormone ethylene
Ethylene ( IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon-carbon double bonds).
Ethylene ...
.
Primordial synthesis
The formation of amino acids and peptides are assumed to precede and perhaps induce the emergence of life on earth. Amino acids can form from simple precursors under various conditions.Strecker synthesis
The Strecker amino acid synthesis, also known simply as the Strecker synthesis, is a method for the synthesis of amino acids by the reaction of an aldehyde with ammonia in the presence of potassium cyanide. The condensation reaction yields an α- ...
whereby hydrogen cyanide, simple aldehydes, ammonia, and water produce amino acids.RNA world
The RNA world is a hypothetical stage in the evolutionary history of life on Earth, in which self-replicating RNA molecules proliferated before the evolution of DNA and proteins. The term also refers to the hypothesis that posits the existence ...
' and the ' DNA world'. Codon
The genetic code is the set of rules used by living cells to translate information encoded within genetic material ( DNA or RNA sequences of nucleotide triplets, or codons) into proteins. Translation is accomplished by the ribosome, which links p ...
–amino acids mappings may be the biological
Biology is the scientific study of life. It is a natural science with a broad scope but has several unifying themes that tie it together as a single, coherent field. For instance, all organisms are made up of cells that process hereditary in ...
information system at the primordial origin of life on Earth. While amino acids and consequently simple peptides must have formed under different experimentally probed geochemical scenarios, the transition from an abiotic world to the first life forms is to a large extent still unresolved.
Reactions
Amino acids undergo the reactions expected of the constituent functional groups.
Peptide bond formation
 As both the amine and carboxylic acid groups of amino acids can react to form amide bonds, one amino acid molecule can react with another and become joined through an amide linkage. This polymerization of amino acids is what creates proteins. This
As both the amine and carboxylic acid groups of amino acids can react to form amide bonds, one amino acid molecule can react with another and become joined through an amide linkage. This polymerization of amino acids is what creates proteins. This condensation reaction
In organic chemistry, a condensation reaction is a type of chemical reaction in which two molecules are combined to form a single molecule, usually with the loss of a small molecule such as water. If water is lost, the reaction is also known as a ...
yields the newly formed peptide bond and a molecule of water. In cells, this reaction does not occur directly; instead, the amino acid is first activated by attachment to a transfer RNA molecule through an ester bond. This aminoacyl-tRNA is produced in an ATP-dependent reaction carried out by an aminoacyl tRNA synthetase
An aminoacyl-tRNA synthetase (aaRS or ARS), also called tRNA-ligase, is an enzyme that attaches the appropriate amino acid onto its corresponding tRNA. It does so by catalyzing the transesterification of a specific cognate amino acid or its pre ...
. This aminoacyl-tRNA is then a substrate for the ribosome, which catalyzes the attack of the amino group of the elongating protein chain on the ester bond. As a result of this mechanism, all proteins made by ribosomes are synthesized starting at their ''N''-terminus and moving toward their ''C''-terminus.
However, not all peptide bonds are formed in this way. In a few cases, peptides are synthesized by specific enzymes. For example, the tripeptide glutathione
Glutathione (GSH, ) is an antioxidant in plants, animals, fungi, and some bacteria and archaea. Glutathione is capable of preventing damage to important cellular components caused by sources such as reactive oxygen species, free radicals, per ...
is an essential part of the defenses of cells against oxidative stress. This peptide is synthesized in two steps from free amino acids. In the first step, gamma-glutamylcysteine synthetase
γ -L-Glutamyl-L-cysteine, also known as γ-glutamylcysteine (GGC), is a dipeptide found in animals, plants, fungi, some bacteria, and archaea. It has a relatively unusual γ-bond between the constituent amino acids, L-glutamic acid and L-cyst ...
condenses cysteine and glutamate through a peptide bond formed between the side chain carboxyl of the glutamate (the gamma carbon of this side chain) and the amino group of the cysteine. This dipeptide is then condensed with glycine by glutathione synthetase
Glutathione synthetase (GSS) () is the second enzyme in the glutathione (GSH) biosynthesis pathway. It catalyses the condensation of gamma-glutamylcysteine and glycine, to form glutathione. Glutathione synthetase is also a potent antioxidant ...
to form glutathione.
In chemistry, peptides are synthesized by a variety of reactions. One of the most-used in solid-phase peptide synthesis uses the aromatic oxime derivatives of amino acids as activated units. These are added in sequence onto the growing peptide chain, which is attached to a solid resin support. Libraries of peptides are used in drug discovery through high-throughput screening
High-throughput screening (HTS) is a method for scientific experimentation especially used in drug discovery and relevant to the fields of biology, materials science and chemistry. Using robotics, data processing/control software, liquid handling ...
.
The combination of functional groups allow amino acids to be effective polydentate ligands for metal–amino acid chelates.
The multiple side chains of amino acids can also undergo chemical reactions.
Catabolism
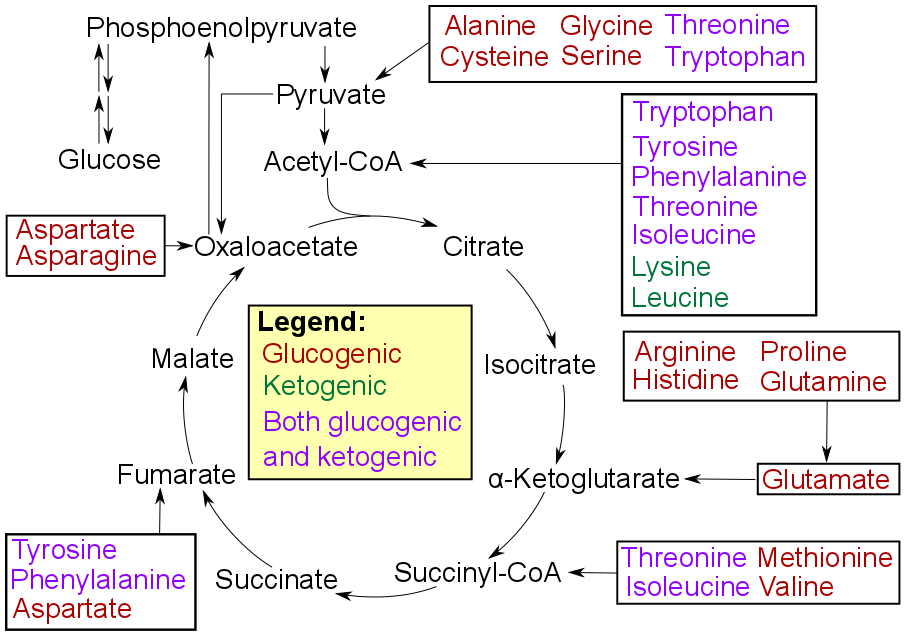 Degradation of an amino acid often involves deamination by moving its amino group to alpha-ketoglutarate, forming glutamate. This process involves transaminases, often the same as those used in amination during synthesis. In many vertebrates, the amino group is then removed through the urea cycle and is excreted in the form of
Degradation of an amino acid often involves deamination by moving its amino group to alpha-ketoglutarate, forming glutamate. This process involves transaminases, often the same as those used in amination during synthesis. In many vertebrates, the amino group is then removed through the urea cycle and is excreted in the form of urea
Urea, also known as carbamide, is an organic compound with chemical formula . This amide has two amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest amide of carbamic acid.
Urea serves an important ...
. However, amino acid degradation can produce uric acid
Uric acid is a heterocyclic compound of carbon, nitrogen, oxygen, and hydrogen with the formula C5H4N4O3. It forms ions and salts known as urates and acid urates, such as ammonium acid urate. Uric acid is a product of the metabolic breakdown o ...
or ammonia instead. For example, serine dehydratase converts serine to pyruvate and ammonia.glycolysis
Glycolysis is the metabolic pathway that converts glucose () into pyruvate (). The free energy released in this process is used to form the high-energy molecules adenosine triphosphate (ATP) and reduced nicotinamide adenine dinucleotide (NADH) ...
or the citric acid cycle
The citric acid cycle (CAC)—also known as the Krebs cycle or the TCA cycle (tricarboxylic acid cycle)—is a series of chemical reactions to release stored energy through the oxidation of acetyl-CoA derived from carbohydrates, fats, and protei ...
, as detailed in image at right.
Complexation
Amino acids are bidentate ligands, forming transition metal amino acid complexes.
:
Chemical analysis
The total nitrogen content of organic matter is mainly formed by the amino groups in proteins. The Total Kjeldahl Nitrogen (TKN
The Kjeldahl method or Kjeldahl digestion () in analytical chemistry is a method for the quantitative determination of nitrogen contained in organic substances plus the nitrogen contained in the inorganic compounds ammonia and ammonium (NH3/NH4+) ...
) is a measure of nitrogen widely used in the analysis of (waste) water, soil, food, feed and organic matter in general. As the name suggests, the Kjeldahl method
The Kjeldahl method or Kjeldahl digestion () in analytical chemistry is a method for the quantitative determination of nitrogen contained in organic substances plus the nitrogen contained in the inorganic compounds ammonia and ammonium (NH3/NH4+). ...
is applied. More sensitive methods are available.
See also
* Amino acid dating
* Beta-peptide
* Degron
* Erepsin
* Homochirality
* Hyperaminoacidemia Hyperaminoacidemia refers to the condition of having an excess of amino acids in the bloodstream. There is evidence that hyperaminoacidemia increases protein synthesis and anabolism
Anabolism () is the set of metabolic pathways that construct molec ...
* Leucines
The leucines are primarily the four isomeric amino acids: leucine, isoleucine, ''tert''-leucine ( terleucine, pseudoleucine) and norleucine. Being compared with the four butanols, they could be classified as butyl-substituted glycines; they re ...
* Miller–Urey experiment
The Miller–Urey experiment (or Miller experiment) is a famous chemistry experiment that simulated the conditions thought at the time (1952) to be present in the atmosphere of the early, prebiotic Earth, in order to test the hypothesis of the ...
* Nucleic acid sequence
* RNA codon table
Notes
References
Further reading
*
*
*
*
External links
*
{{DEFAULTSORT:Amino Acid
Nitrogen cycle
Zwitterions
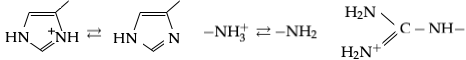 There are three amino acids with side-chains that are cations at neutral pH (though in one, histidine, cationic and neutral forms both exist). They are commonly called ''basic amino acids'', but this term is misleading: histidine can act both as a Brønsted acid and as a Brønsted base at neutral pH, lysine acts as a Brønsted acid, and arginine has a fixed positive charge and does not ionize in neutral conditions. The names ''histidinium, lysinium'' and ''argininium'' would be more accurate names for the structures, but have essentially no currency.
*
There are three amino acids with side-chains that are cations at neutral pH (though in one, histidine, cationic and neutral forms both exist). They are commonly called ''basic amino acids'', but this term is misleading: histidine can act both as a Brønsted acid and as a Brønsted base at neutral pH, lysine acts as a Brønsted acid, and arginine has a fixed positive charge and does not ionize in neutral conditions. The names ''histidinium, lysinium'' and ''argininium'' would be more accurate names for the structures, but have essentially no currency.
* In aqueous solution at pH close to neutrality, amino acids exist as zwitterions, i.e. as dipolar ions with both and in charged states, so the overall structure is . At physiological pH the so-called "neutral forms" are not present to any measurable degree. Although the two charges in the zwitterion structure add up to zero it is misleading to call a species with a net charge of zero "uncharged".
In strongly acidic conditions (pH below 3), the carboxylate group becomes protonated and the structure becomes an ammonio carboxylic acid, . This is relevant for enzymes like pepsin that are active in acidic environments such as the mammalian stomach and
In aqueous solution at pH close to neutrality, amino acids exist as zwitterions, i.e. as dipolar ions with both and in charged states, so the overall structure is . At physiological pH the so-called "neutral forms" are not present to any measurable degree. Although the two charges in the zwitterion structure add up to zero it is misleading to call a species with a net charge of zero "uncharged".
In strongly acidic conditions (pH below 3), the carboxylate group becomes protonated and the structure becomes an ammonio carboxylic acid, . This is relevant for enzymes like pepsin that are active in acidic environments such as the mammalian stomach and  For amino acids with uncharged side-chains the zwitterion predominates at pH values between the two p''K''a values, but coexists in equilibrium with small amounts of net negative and net positive ions. At the midpoint between the two p''K''a values, the trace amount of net negative and trace of net positive ions balance, so that average net charge of all forms present is zero. This pH is known as the
For amino acids with uncharged side-chains the zwitterion predominates at pH values between the two p''K''a values, but coexists in equilibrium with small amounts of net negative and net positive ions. At the midpoint between the two p''K''a values, the trace amount of net negative and trace of net positive ions balance, so that average net charge of all forms present is zero. This pH is known as the  When taken up into the human body from the diet, the 20 standard amino acids either are used to synthesize proteins, other biomolecules, or are oxidized to
When taken up into the human body from the diet, the 20 standard amino acids either are used to synthesize proteins, other biomolecules, or are oxidized to  Degradation of an amino acid often involves deamination by moving its amino group to alpha-ketoglutarate, forming glutamate. This process involves transaminases, often the same as those used in amination during synthesis. In many vertebrates, the amino group is then removed through the urea cycle and is excreted in the form of
Degradation of an amino acid often involves deamination by moving its amino group to alpha-ketoglutarate, forming glutamate. This process involves transaminases, often the same as those used in amination during synthesis. In many vertebrates, the amino group is then removed through the urea cycle and is excreted in the form of 