Organocopper on:
[Wikipedia]
[Google]
[Amazon]

 Organocopper chemistry is the study of the physical properties, reactions, and synthesis of organocopper compounds, which are organometallic compounds containing a
Organocopper chemistry is the study of the physical properties, reactions, and synthesis of organocopper compounds, which are organometallic compounds containing a




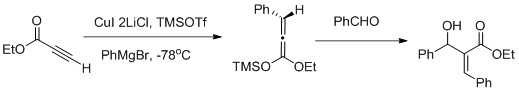 Muller and collaborators reported a vicinal functionalization of α,β-acetylenic esters using a carbocupration/Mukaiyama aldol reaction sequence (as shown in the figure above) carbocupration favors the formation of the Z-aldol.
Muller and collaborators reported a vicinal functionalization of α,β-acetylenic esters using a carbocupration/Mukaiyama aldol reaction sequence (as shown in the figure above) carbocupration favors the formation of the Z-aldol.

 Organocopper chemistry is the study of the physical properties, reactions, and synthesis of organocopper compounds, which are organometallic compounds containing a
Organocopper chemistry is the study of the physical properties, reactions, and synthesis of organocopper compounds, which are organometallic compounds containing a carbon
Carbon () is a chemical element; it has chemical symbol, symbol C and atomic number 6. It is nonmetallic and tetravalence, tetravalent—meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 ...
to copper
Copper is a chemical element; it has symbol Cu (from Latin ) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orang ...
chemical bond
A chemical bond is the association of atoms or ions to form molecules, crystals, and other structures. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds or through the sharing of electrons a ...
. They are reagents
In chemistry, a reagent ( ) or analytical reagent is a substance or compound added to a system to cause a chemical reaction, or test if one occurs. The terms ''reactant'' and ''reagent'' are often used interchangeably, but reactant specifies a ...
in organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the science, scientific study of the structure, properties, and reactions of organic compounds and organic matter, organic materials, i.e., matter in its various forms that contain ...
.
The first organocopper compound, the explosive copper(I) acetylide (), was synthesized by Rudolf Christian Böttger in 1859 by passing acetylene
Acetylene (Chemical nomenclature, systematic name: ethyne) is a chemical compound with the formula and structure . It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is u ...
gas through a solution of copper(I) chloride
Copper(I) chloride, commonly called cuprous chloride, is the lower chloride of copper, with the formula CuCl. The substance is a white solid sparingly soluble in water, but very soluble in concentrated hydrochloric acid. Impure samples appear gr ...
:
:
Structure and bonding
Organocopper compounds are diverse in structure and reactivity, but almost all are based on copper with anoxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical Electrical charge, charge of an atom if all of its Chemical bond, bonds to other atoms are fully Ionic bond, ionic. It describes the degree of oxidation (loss of electrons ...
of +1, sometimes denoted Cu(I) or . With 10 electrons in its valence shell
In chemistry and physics, valence electrons are electrons in the outermost shell of an atom, and that can participate in the formation of a chemical bond if the outermost shell is not closed. In a single covalent bond, a shared pair forms with b ...
, the bonding behavior of Cu(I) is similar to Ni(0), but owing to its higher oxidation state, it engages in less pi-backbonding. Organic derivatives of copper's higher oxidation states of +2 and +3 are sometimes encountered as reaction intermediate
In chemistry, a reaction intermediate, or intermediate, is a molecular entity arising within the sequence of a stepwise chemical reaction. It is formed as the reaction product of an elementary step, from the reactants and/or preceding interme ...
s, but rarely isolated or even observed.
Organocopper compounds form complexes with a variety of soft ligand
In coordination chemistry, a ligand is an ion or molecule with a functional group that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's el ...
s such as alkylphosphines (), thioethers
In organic chemistry, a sulfide (British English sulphide) or thioether is an organosulfur functional group with the connectivity as shown on right. Like many other sulfur-containing compounds, volatile sulfides have foul odors. A sulfide is s ...
(), and cyanide
In chemistry, cyanide () is an inorganic chemical compound that contains a functional group. This group, known as the cyano group, consists of a carbon atom triple-bonded to a nitrogen atom.
Ionic cyanides contain the cyanide anion . This a ...
().
Due to the spherical electronic shell of , copper(I) complexes have symmetrical structures - either linear
In mathematics, the term ''linear'' is used in two distinct senses for two different properties:
* linearity of a '' function'' (or '' mapping'');
* linearity of a '' polynomial''.
An example of a linear function is the function defined by f(x) ...
, trigonal planar
In chemistry, trigonal planar is a molecular geometry model with one atom at the center and three atoms at the corners of an equilateral triangle, called peripheral atoms, all in one plane. In an ideal trigonal planar species, all three ligands a ...
or tetrahedral
In geometry, a tetrahedron (: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular Face (geometry), faces, six straight Edge (geometry), edges, and four vertex (geometry), vertices. The tet ...
, depending on the number of ligands
In coordination chemistry, a ligand is an ion or molecule with a functional group that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's ...
.
Simple complexes with CO, alkene, and Cp ligands
Copper(I) salts have long been known to bind CO, albeit weakly. A representative complex is carbonylcopper(I) chloride CuCl(CO), which is polymeric. In contrast to classicalmetal carbonyl
A metal () is a material that, when polished or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. These properties are all associated with having electrons available at the Fermi level, as against n ...
s, pi backbonding
In chemistry, pi backbonding or π backbonding is a π-bonding interaction between a filled (or half filled) orbital of a transition metal atom and a vacant orbital on an adjacent ion or molecule. In this type of interaction, electrons from the ...
is not strong in these compounds.

Alkenes
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as Alpha-olefin, α-olefins.
The Internationa ...
bind to copper(I), although again generally weakly. The binding of ethylene
Ethylene (IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon–carbon bond, carbon–carbon doub ...
to Cu in proteins
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, re ...
has a broad significance in plant biology
Botany, also called plant science, is the branch of natural science
Natural science or empirical science is one of the branches of science concerned with the description, understanding and prediction of natural phenomena, based on empiri ...
so much so that ethylene is classified as a plant hormone
Plant hormones (or phytohormones) are signal molecules, produced within plants, that occur in extremely low concentrations. Plant hormones control all aspects of plant growth and development, including embryogenesis, the regulation of Organ (anat ...
. Its presence, detected by the Cu-protein, affects fruit ripening and many other developments.
Copper forms no metallocene
A metallocene is a compound typically consisting of two cyclopentadienyl anions (, abbreviated Cp) bound to a metallic element, metal center (M) in the oxidation state II, with the resulting general formula Closely related to the metallocenes are ...
, and adds cyclopentadiene only in the presence of "soft" Lewis bases to give half-sandwich complex
Half sandwich compounds, also known as piano stool complexes, are Organometallic chemistry, organometallic complexes that feature a cyclic hapticity, polyhapto ligand bound to an MLn center, where L is a unidentate ligand. Thousands of such complex ...
es. One such derivative is π-cyclopentadienyl(triethylphosphine)copper(I), .
Alkyl and aryl copper compounds
Alkyl and aryl copper(I) compounds
Copper halides react withorganolithium reagent
In organometallic chemistry, organolithium reagents are chemical compounds that contain carbon–lithium (C–Li) bonds. These reagents are important in organic synthesis, and are frequently used to transfer the organic group or the lithium atom ...
s to give organocopper compounds. The area was pioneered by Henry Gilman
Henry Gilman (May 9, 1893 – November 7, 1986) was an American organic chemist known as the father of organometallic chemistry. He discovered the Gilman reagent, which bears his name.
Early life and education
Henry Gilman was born in Boston ...
, who reported methylcopper in 1936. Thus, phenylcopper
Phenylcopper is an organometallic chemical compound of copper. Its chemical formula is , where copper is in the oxidation state of +1.
Synthesis
Phenylcopper was the first known organocopper compound and was first prepared in 1923 from phenylmagn ...
is prepared by reaction of phenyllithium
Phenyllithium is an organometallic agent with the empirical formula . It is most commonly used as a metalating agent in organic syntheses and a substitute for Grignard reagents for introducing phenyl groups in organic syntheses. Crystalline phenyl ...
with copper(I) bromide
Copper(I) bromide is the chemical compound with the formula CuBr. This white diamagnetic solid adopts a polymeric structure akin to that for zinc sulfide. The compound is widely used in the synthesis of organic compounds and as a lasing medium ...
in diethyl ether
Diethyl ether, or simply ether, is an organic compound with the chemical formula , sometimes abbreviated as . It is a colourless, highly Volatility (chemistry), volatile, sweet-smelling ("ethereal odour"), extremely flammable liquid. It belongs ...
. Grignard reagents can be used in place of organolithium compounds. Gilman also investigated the dialkylcuprates. These are obtained by combining two equivalent of RLi with Cu(I) salts. Alternatively, these cuprates are prepared from oligomeric neutral organocopper compounds by treatment with one equivalent of organolithium reagent.
Compounds of the type are reactive towards oxygen and water, forming copper(I) oxide
Copper(I) oxide or cuprous oxide is the inorganic compound with the formula . It is one of the principal oxides of copper, the other being copper(II) oxide or cupric oxide (CuO). The compound can appear either yellow or red, depending on the size ...
. They also tend to be thermally unstable, which can be useful in certain coupling reactions. Despite or because of these difficulties, organocopper reagents are frequently generated and consumed in situ
is a Latin phrase meaning 'in place' or 'on site', derived from ' ('in') and ' ( ablative of ''situs'', ). The term typically refers to the examination or occurrence of a process within its original context, without relocation. The term is use ...
with no attempt to isolate them. They are used in organic synthesis
Organic synthesis is a branch of chemical synthesis concerned with the construction of organic compounds. Organic compounds are molecules consisting of combinations of covalently-linked hydrogen, carbon, oxygen, and nitrogen atoms. Within the gen ...
as alkylating reagents because they exhibit greater functional group
In organic chemistry, a functional group is any substituent or moiety (chemistry), moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions r ...
tolerance than corresponding Grignard and organolithium reagents. The electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the ...
of copper is much higher than its next-door neighbor in the group 12 element
Group 12, by modern IUPAC numbering, is a group of chemical elements in the periodic table. It includes zinc (Zn), cadmium (Cd), mercury (Hg), and copernicium (Cn). Formerly this group was named ''IIB'' (pronounced as "group two B", as the "I ...
s, zinc
Zinc is a chemical element; it has symbol Zn and atomic number 30. It is a slightly brittle metal at room temperature and has a shiny-greyish appearance when oxidation is removed. It is the first element in group 12 (IIB) of the periodic tabl ...
, suggesting diminished nucleophilicity
In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. Because nucleophiles donate electrons, they are ...
for its carbon ligands.
Copper salts react with terminal alkyne
\ce
\ce
Acetylene
\ce
\ce
\ce
Propyne
\ce
\ce
\ce
\ce
1-Butyne
In organic chemistry, an alkyne is an unsaturated hydrocarbon containing at least one carbon—carbon triple bond. The simplest acyclic alkynes with only one triple bond and n ...
s to form the acetylide
In chemistry, an acetylide is a compound that can be viewed as the result of replacing one or both hydrogen atoms of acetylene (ethyne) by metallic or other cations. Calcium carbide is an important industrial compound, which has long been used ...
s.
Alkyl halides react with organocopper compounds with inversion of configuration. On the other hand, reactions of organocopper compound with alkenyl halides proceed with retention of subtrate’s configuration.
Organocopper compounds couple with aryl halides (see Ullmann condensation
The Ullmann condensation or Ullmann-type reaction is the copper-promoted conversion of aryl halides to aryl ethers, aryl thioethers, aryl nitriles, and aryl amines. These reactions are examples of cross-coupling reactions.
Ullmann-type reaction ...
and Ullmann reaction
The Ullmann reaction or Ullmann coupling, named after Fritz Ullmann, couples two aryl or alkyl groups with the help of copper. The reaction was first reported by Ullmann and his student Bielecki in 1901. It has been later shown that palladium and ...
):
:
:
:
Structures
Alkyl and aryl copper complexes aggregate both in crystalline form and in solution. Aggregation is especially evident for charge-neutral organocopper compounds, i.e. species with theempirical formula
In chemistry, the empirical formula of a chemical compound is the simplest whole number ratio of atoms present in a compound. A simple example of this concept is that the empirical formula of sulfur monoxide, or SO, is simply SO, as is the empir ...
(RCu), which adopt cyclic structures. Since each copper center requires at least two ligands, the organic group is a bridging ligand
In coordination chemistry, a bridging ligand is a ligand that connects two or more atoms, usually metal ions. The ligand may be atomic or polyatomic. Virtually all complex organic compounds can serve as bridging ligands, so the term is usually r ...
. This effect is illustrated by the structure of mesitylcopper, which is a pentamer. A cyclic structure is also seen for , where Me stands for methyl group
In organic chemistry, a methyl group is an alkyl derived from methane, containing one carbon atom bonded to three hydrogen atoms, having chemical formula (whereas normal methane has the formula ). In formulas, the group is often abbreviated a ...
, the first 1:1 organocopper compound to be analyzed by X-ray crystallography
Crystallography is the branch of science devoted to the study of molecular and crystalline structure and properties. The word ''crystallography'' is derived from the Ancient Greek word (; "clear ice, rock-crystal"), and (; "to write"). In J ...
(1972 by Lappert). This compound is relatively stable because the bulky trimethylsilyl
A trimethylsilyl group (abbreviated TMS) is a functional group in organic chemistry. This group consists of three methyl groups bonded to a silicon atom minus;Si(CH3)3 which is in turn bonded to the rest of a molecule. This structural group i ...
groups provide steric protection. It is a tetramer
A tetramer () (''tetra-'', "four" + '' -mer'', "parts") is an oligomer formed from four monomers or subunits. The associated property is called ''tetramery''. An example from inorganic chemistry is titanium methoxide with the empirical formula ...
, forming an 8-membered ring with alternating Cu-C bonds. In addition the four copper atoms form a planar ring based on three-center two-electron bond
A three-center two-electron (3c–2e) bond is an electron-deficient chemical bond where three atoms share two electrons. The combination of three atomic orbitals form three molecular orbitals: one bonding, one ''non''-bonding, and one ''anti''- ...
s. The copper to copper bond length
In molecular geometry, bond length or bond distance is defined as the average distance between Atomic nucleus, nuclei of two chemical bond, bonded atoms in a molecule. It is a Transferability (chemistry), transferable property of a bond between at ...
is 242 pm compared to 256 pm in bulk copper. In ''pentamesitylpentacopper'' a 5-membered copper ring is formed, similar to (2,4,6-trimethylphenyl)gold, and ''pentafluorophenylcopper'' is a tetramer.
:
Lithium dimethylcuprate(I) is a dimer
Dimer may refer to:
* Dimer (chemistry), a chemical structure formed from two similar sub-units
** Protein dimer, a protein quaternary structure
** d-dimer
** TH-dimer
* Dimer model, an item in statistical mechanics, based on ''domino tiling''
* ...
in diethyl ether
Diethyl ether, or simply ether, is an organic compound with the chemical formula , sometimes abbreviated as . It is a colourless, highly Volatility (chemistry), volatile, sweet-smelling ("ethereal odour"), extremely flammable liquid. It belongs ...
, forming an 8-membered ring with two lithium atoms linking two methyl groups, . Similarly, lithium diphenylcuprate(I) forms a dimeric etherate, , in the solid state.
Alkyl and aryl copper(III) compounds
The involvement of the otherwise rare Cu(III) oxidation state has been demonstrated in theconjugate addition
Nucleophilic conjugate addition is a type of organic reaction. Ordinary nucleophilic additions or 1,2-nucleophilic additions deal mostly with additions to carbonyl compounds. Simple alkene compounds do not show 1,2 reactivity due to lack of polar ...
of the Gilman reagent
A Gilman reagent is a diorganocopper compound with the formula Li uR2 where R is an alkyl or aryl. They are colorless solids.
Use in organic chemistry
These reagents are useful because, unlike related Grignard reagents and organolithium reage ...
to an enone: In a so-called rapid-injection NMR
Nuclear magnetic resonance (NMR) is a physical phenomenon in which atomic nucleus, nuclei in a strong constant magnetic field are disturbed by a weak oscillating magnetic field (in the near and far field, near field) and respond by producing ...
experiment at −100 °C, the Gilman reagent (stabilized by lithium iodide
Lithium iodide, or LiI, is a compound of lithium and iodine. When exposed to air, it becomes yellow in color, due to the oxidation of iodide to iodine. It crystallizes in the NaCl motif. It can participate in various hydrates.Wietelmann, Ulrich a ...
) was introduced to cyclohexenone (1) enabling the detection of the copper — alkene pi complex
The number (; spelled out as pi) is a mathematical constant, approximately equal to 3.14159, that is the ratio of a circle's circumference to its diameter. It appears in many formulae across mathematics and physics, and some of these formu ...
2. On subsequent addition of trimethylsilyl cyanide
Trimethylsilyl cyanide is the chemical compound with the chemical formula, formula . This volatile liquid consists of a cyanide group, that is CN, attached to a trimethylsilyl group. The molecule is used in organic synthesis as the equivalent of h ...
the Cu(III) species 3 is formed (indefinitely stable at that temperature) and on increasing the temperature to −80 °C the conjugate addition product 4. According to an accompanying in silico
In biology and other experimental sciences, an ''in silico'' experiment is one performed on a computer or via computer simulation software. The phrase is pseudo-Latin for 'in silicon' (correct ), referring to silicon in computer chips. It was c ...
experiments the Cu(III) intermediate has a square planar molecular geometry
In chemistry, the square planar molecular geometry describes the stereochemistry (spatial arrangement of atoms) that is adopted by certain chemical compounds. As the name suggests, molecules of this geometry have their atoms positioned at the co ...
with the cyano
In chemistry, cyanide () is an inorganic chemical compound that contains a functional group. This group, known as the cyano group, consists of a carbon atom triple-bonded to a nitrogen atom.
Ionic cyanides contain the cyanide anion . This a ...
group in ''cis'' orientation with respect to the cyclohexenyl methine
In organic chemistry, a methine group or methine bridge is a trivalent functional group , derived formally from methane. It consists of a carbon atom bound by two single bonds and one double bond, where one of the single bonds is to a hydrogen. ...
group and anti-parallel to the methine proton. With other ligands than the cyano group
In chemistry, cyanide () is an inorganic chemical compound that contains a functional group. This group, known as the cyano group, consists of a carbon atom triple-bonded to a nitrogen atom.
Ionic cyanides contain the cyanide anion . This an ...
this study predicts room temperature
Room temperature, colloquially, denotes the range of air temperatures most people find comfortable indoors while dressed in typical clothing. Comfortable temperatures can be extended beyond this range depending on humidity, air circulation, and ...
stable Cu(III) compounds.
:
Reactions of organocuprates
Cross-coupling reactions
Prior to the development ofpalladium
Palladium is a chemical element; it has symbol Pd and atomic number 46. It is a rare and lustrous silvery-white metal discovered in 1802 by the English chemist William Hyde Wollaston. He named it after the asteroid Pallas (formally 2 Pallas), ...
-catalyzed cross coupling reaction
In organic chemistry, a cross-coupling reaction is a reaction where two different fragments are joined. Cross-couplings are a subset of the more general coupling reactions. Often cross-coupling reactions require metal catalysts. One important reac ...
s, copper
Copper is a chemical element; it has symbol Cu (from Latin ) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orang ...
was the preferred catalyst
Catalysis () is the increase in rate of a chemical reaction due to an added substance known as a catalyst (). Catalysts are not consumed by the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quick ...
for almost a century. Palladium offers a faster, more selective reaction. Copper reagents and catalysts continue to be the subject of innovation. Relative to palladium, copper is cheaper but the turnover number
In chemistry, the term "turnover number" has two distinct meanings.
In enzymology, the turnover number () is defined as the limiting number of chemical conversions of substrate molecules per second that a single active site will execute for a g ...
s are often lower with copper and the reaction conditions more vigorous.
Reactions of with alkyl halide
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents of hydrogen atom. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalka ...
s give the coupling product:
:
The reaction mechanism
In chemistry, a reaction mechanism is the step by step sequence of elementary reactions by which overall chemical reaction occurs.
A chemical mechanism is a theoretical conjecture that tries to describe in detail what takes place at each stage ...
involves oxidative addition
Oxidative addition and reductive elimination are two important and related classes of reactions in organometallic chemistry. Oxidative addition is a process that increases both the oxidation state and coordination number of a metal centre. Oxidat ...
(OA) of the alkyl halide to Cu(I), forming a planar
Planar is an adjective meaning "relating to a plane (geometry)".
Planar may also refer to:
Science and technology
* Planar (computer graphics), computer graphics pixel information from several bitplanes
* Planar (transmission line technologies), ...
Cu(III) intermediate, followed by reductive elimination
Reductive elimination is an elementary step in organometallic chemistry in which the oxidation state of the metal center decreases while forming a new covalent bond between two ligands. It is the microscopic reverse of oxidative addition, and is ...
(RE). The nucleophilic attack is the rate-determining step. In the substitution of iodide
An iodide ion is I−. Compounds with iodine in formal oxidation state −1 are called iodides. In everyday life, iodide is most commonly encountered as a component of iodized salt, which many governments mandate. Worldwide, iodine deficiency ...
, a single-electron transfer mechanism is proposed (see figure).
:
Many electrophiles participate in this reaction. The approximate order of reactivity, beginning with the most reactive, is as follows: acid chloride
In organic chemistry, an acyl chloride (or acid chloride) is an organic compound with the functional group . Their formula is usually written , where R is a side chain. They are reactive derivatives of carboxylic acids (). A specific example o ...
s > aldehyde
In organic chemistry, an aldehyde () (lat. ''al''cohol ''dehyd''rogenatum, dehydrogenated alcohol) is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred ...
s > tosylate
In organic chemistry, a toluenesulfonyl group (tosyl group, abbreviated Ts or TosIn this article, "Ts", unless otherwise stated, means tosyl, not tennessine.) is a univalent functional group with the chemical formula . It consists of a tolyl ...
s ~ epoxide
In organic chemistry, an epoxide is a cyclic ether, where the ether forms a three-atom ring: two atoms of carbon and one atom of oxygen. This triangular structure has substantial ring strain, making epoxides highly reactive, more so than other ...
s > iodide
An iodide ion is I−. Compounds with iodine in formal oxidation state −1 are called iodides. In everyday life, iodide is most commonly encountered as a component of iodized salt, which many governments mandate. Worldwide, iodine deficiency ...
s > bromide
A bromide ion is the negatively charged form (Br−) of the element bromine, a member of the halogens group on the periodic table. Most bromides are colorless. Bromides have many practical roles, being found in anticonvulsants, flame-retard ...
s > chloride
The term chloride refers to a compound or molecule that contains either a chlorine anion (), which is a negatively charged chlorine atom, or a non-charged chlorine atom covalently bonded to the rest of the molecule by a single bond (). The pr ...
s > ketone
In organic chemistry, a ketone is an organic compound with the structure , where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond C=O). The simplest ketone is acetone ( ...
s > ester
In chemistry, an ester is a compound derived from an acid (either organic or inorganic) in which the hydrogen atom (H) of at least one acidic hydroxyl group () of that acid is replaced by an organyl group (R). These compounds contain a distin ...
s > nitrile
In organic chemistry, a nitrile is any organic compound that has a functional group. The name of the compound is composed of a base, which includes the carbon of the , suffixed with "nitrile", so for example is called " propionitrile" (or pr ...
s >> alkene
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as Alpha-olefin, α-olefins.
The Internationa ...
s
Generally the OA-RE mechanism is analogous to that of palladium-catalyzed cross coupling reactions. One difference between copper and palladium is that copper can undergo single-electron transfer processes.Posner, G. H. 2011. Substitution Reactions Using Organocopper Reagents. Organic Reactions. 22:2:253–400
Coupling reactions
Oxidative coupling is the coupling of copper acetylides to conjugated alkynes in theGlaser coupling
The Glaser coupling is a type of coupling reaction. It is by far one of the oldest coupling reactions and is based on copper compounds like copper(I) chloride or copper(I) bromide and an additional oxidant like air. The base used in the original ...
(for example in the synthesis of cyclooctadecanonaene
Cyclooctadecanonaene or 8nnulene is an organic compound with chemical formula . It belongs to the class of highly conjugated compounds known as annulenes and is aromatic. The usual isomer that 8nnulene refers to is the most stable one, containi ...
) or to aryl halides in the Castro-Stephens Coupling.
Reductive coupling is a coupling reaction
In organic chemistry, a coupling reaction is a type of reaction in which two reactant molecules are bonded together. Such reactions often require the aid of a metal catalyst. In one important reaction type, a main group organometallic compound o ...
of aryl halides with a stoichiometric equivalent of copper metal that occurs in the Ullmann reaction
The Ullmann reaction or Ullmann coupling, named after Fritz Ullmann, couples two aryl or alkyl groups with the help of copper. The reaction was first reported by Ullmann and his student Bielecki in 1901. It has been later shown that palladium and ...
. A related reaction called decarboxylative cross-coupling, one coupling partner is a carboxylate. Cu(I) displaces a carboxyl
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an R-group. The general formula of a carboxylic acid is often written as or , sometimes as with R referring to an organyl group (e.g. ...
forming the arylcopper (ArCu) intermediate. Simultaneously, a palladium catalyst reacts with an aryl bromide to give an organopalladium intermediate (Ar'PdB), which undergoes transmetallation Transmetalation (alt. spelling: transmetallation) is a type of organometallic reaction that involves the transfer of ligands from one metal to another. It has the general form:
:M1–R + M2–R′ → M1–R′ + M2–R
where R and R′ can be, but ...
to give ArPdAr', which in turn reductively eliminates the biaryl
Biphenyl (also known as diphenyl, phenylbenzene, 1,1′-biphenyl, lemonene or BP) is an organic compound that forms colorless crystals. Particularly in older literature, compounds containing the functional group consisting of biphenyl less one ...
.
:
Redox neutral coupling is the coupling of terminal alkynes with halo-alkynes with a copper(I) salt in the Cadiot-Chodkiewicz coupling. Thermal coupling of two organocopper compounds is also possible.
Carbocupration
Carbocupration is anucleophilic addition
In organic chemistry, a nucleophilic addition (AN) reaction is an addition reaction where a chemical compound with an electrophilic double or triple bond reacts with a nucleophile, such that the double or triple bond is broken. Nucleophilic addit ...
of organocopper reagents () to acetylene
Acetylene (Chemical nomenclature, systematic name: ethyne) is a chemical compound with the formula and structure . It is a hydrocarbon and the simplest alkyne. This colorless gas is widely used as a fuel and a chemical building block. It is u ...
or terminal alkynes
\ce
\ce
Acetylene
\ce
\ce
\ce
Propyne
\ce
\ce
\ce
\ce
1-Butyne
In organic chemistry, an alkyne is an unsaturated hydrocarbon containing at least one carbon—carbon triple bond. The simplest acyclic alkynes with only one triple bond and no ...
resulting in an alkenylcopper compound (). It is a special case of carbometalation
A carbometallation is any reaction where a carbon-metal bond reacts with a carbon-carbon π-bond to produce a new carbon-carbon σ-bond and a carbon-metal σ-bond. The resulting carbon-metal bond can undergo further carbometallation reactions (oli ...
and also called the Normant reaction.

Synthetic applications
*The Chan-Lam coupling enables the formation of aryl carbon-hetoroatom bonds. It involves coupling ofboronic acids
A boronic acid is an organic compound related to boric acid () in which one of the three hydroxyl groups () is replaced by an alkyl or aryl group (represented by R in the general formula ). As a compound containing a carbon–boron bond, memb ...
, stannanes, or siloxanes with NH- or OH-containing substrates.
*Ullmann reaction
The Ullmann reaction or Ullmann coupling, named after Fritz Ullmann, couples two aryl or alkyl groups with the help of copper. The reaction was first reported by Ullmann and his student Bielecki in 1901. It has been later shown that palladium and ...
involves copper-mediated reactions of aryl halides. Two types of Ullmann reaction are recognized:
*#Classic copper-promoted synthesis of symmetric biaryl compounds)
*#Copper-promoted nucleophilic aromatic substitution.
*Sonogashira coupling
The Sonogashira reaction is a cross-coupling reaction used in organic synthesis to form carbon–carbon bonds. It employs a palladium catalyst as well as copper co-catalyst to form a carbon–carbon bond between a terminal alkyne and an aryl or vi ...
reaction, which utilizes both copper and palladium, entails the coupling of aryl and/or vinyl halides with terminal alkynes.
Reducing agents
Copper hydrides are specializedreducing agent
In chemistry, a reducing agent (also known as a reductant, reducer, or electron donor) is a chemical species that "donates" an electron to an (called the , , , or ).
Examples of substances that are common reducing agents include hydrogen, carbon ...
s. The well-known copper hydride is Stryker's reagent
Stryker's reagent ( PPh3)CuHsub>6), also known as the Osborn complex, is a hexameric copper hydride ligated with triphenylphosphine. It is a brick red, air-sensitive solid. Stryker's reagent is a mildly hydridic reagent, used in homogeneous cata ...
, with the formula PPh3)CuHsub>6. It reduces the alkene portion of α,β-Unsaturated carbonyl compound
α,β-Unsaturated carbonyl compounds are organic compounds with the general structure (O=CR)−Cα=Cβ−R. Such compounds include enones and enals, but also carboxylic acids and the corresponding esters and amides. In these compounds, the carb ...
s. A related but catalytic reaction uses copper(I) NHC complex with hydride equivalents provided by a hydrosilane
Hydrosilanes are tetravalent silicon compounds containing one or more Si-H bond. The parent hydrosilane is silane (SiH4). Commonly, hydrosilane refers to organosilicon derivatives. Examples include phenylsilane (PhSiH3) and triethoxysilane ((C2H ...
.
:
Copper alkylation reaction
Generally, the alkylation reaction of organocopper reagents proceed via ''gamma''- alkylation. ''Cis''- ''gamma'' attack occurs better in cyclohexyl carbamate due to sterics. The reaction is reported to be favorable in ethereal solvents. This method was proved to be very effective for the oxidative coupling of amines and alkyl, including ''tert''-butyl, and aryl halides.Vicinal functionalization reactions
Vicinal functionalization using a carbocupration/Mukaiyama aldol reaction sequence:Muller, A.J.; Jennings, M.P. Vicinal Functionalization of propionilate Esters via Tandem Catalytic Carbocupration-Mukaiyama Aldol Reaction sequence. Org. Lett. 2008, 10, 1649-1652 Muller and collaborators reported a vicinal functionalization of α,β-acetylenic esters using a carbocupration/Mukaiyama aldol reaction sequence (as shown in the figure above) carbocupration favors the formation of the Z-aldol.
Muller and collaborators reported a vicinal functionalization of α,β-acetylenic esters using a carbocupration/Mukaiyama aldol reaction sequence (as shown in the figure above) carbocupration favors the formation of the Z-aldol.
See also
* Ethyl copperFurther reading
* *References
{{ChemicalBondsToCarbon