Intramolecular force on:
[Wikipedia]
[Google]
[Amazon]
An intramolecular force (or primary forces) is any
 An
An
 In a true
In a true

 Bonds are formed by atoms so that they are able to achieve a lower energy state. Free atoms will have more energy than a bonded atom. This is because some energy is released during bond formation, allowing the entire system to achieve a lower energy state. The bond length, or the minimum separating distance between two atoms participating in bond formation, is determined by their repulsive and attractive forces along the internuclear direction. As the two atoms get closer and closer, the positively charged nuclei repel, creating a force that attempts to push the atoms apart. As the two atoms get further apart, attractive forces work to pull them back together. Thus an equilibrium bond length is achieved and is a good measure of bond stability.
Bonds are formed by atoms so that they are able to achieve a lower energy state. Free atoms will have more energy than a bonded atom. This is because some energy is released during bond formation, allowing the entire system to achieve a lower energy state. The bond length, or the minimum separating distance between two atoms participating in bond formation, is determined by their repulsive and attractive forces along the internuclear direction. As the two atoms get closer and closer, the positively charged nuclei repel, creating a force that attempts to push the atoms apart. As the two atoms get further apart, attractive forces work to pull them back together. Thus an equilibrium bond length is achieved and is a good measure of bond stability.
 Intramolecular forces are extremely important in the field of biochemistry, where it comes into play at the most basic levels of biological structures. Intramolecular forces such as
Intramolecular forces are extremely important in the field of biochemistry, where it comes into play at the most basic levels of biological structures. Intramolecular forces such as
force
In physics, a force is an influence that can change the motion of an object. A force can cause an object with mass to change its velocity (e.g. moving from a state of rest), i.e., to accelerate. Force can also be described intuitively as a ...
that binds together the atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, a ...
s making up a molecule
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and b ...
or compound, not to be confused with intermolecular force
An intermolecular force (IMF) (or secondary force) is the force that mediates interaction between molecules, including the electromagnetic forces of attraction
or repulsion which act between atoms and other types of neighbouring particles, e.g. ...
s, which are the forces present between molecules. The subtle difference in the name comes from the Latin roots of English with inter meaning ''between or among'' and intra meaning ''inside''. Chemical bonds are considered to be intramolecular forces which are often stronger than intermolecular force
An intermolecular force (IMF) (or secondary force) is the force that mediates interaction between molecules, including the electromagnetic forces of attraction
or repulsion which act between atoms and other types of neighbouring particles, e.g. ...
s present between non-bonding atoms or molecules.
Types
The classical model identifies three main types of chemical bonds — ionic, covalent, and metallic — distinguished by the degree of charge separation between participating atoms. The characteristics of the bond formed can be predicted by the properties of constituent atoms, namely electronegativity. They differ in the magnitude of their bond enthalpies, a measure of bond strength, and thus affect the physical and chemical properties of compounds in different ways. % of ionic character is directly proportional difference in electronegitivity of bonded atom.Ionic bond
 An
An ionic bond
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, and is the primary interaction occurring in ionic compounds ...
can be approximated as complete transfer of one or more valence electrons of atoms participating in bond formation, resulting in a positive ion and a negative ion bound together by electrostatic forces. Electrons in an ionic bond tend to be mostly found around one of the two constituent atoms due to the large electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the ...
difference between the two atoms, generally more than 1.9, (greater difference in electronegativity results in a stronger bond); this is often described as one atom giving electrons to the other. This type of bond is generally formed between a metal
A metal (from ancient Greek, Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electrical resistivity and conductivity, e ...
and nonmetal
In chemistry, a nonmetal is a chemical element that generally lacks a predominance of metallic properties; they range from colorless gases (like hydrogen) to shiny solids (like carbon, as graphite). The electrons in nonmetals behave differentl ...
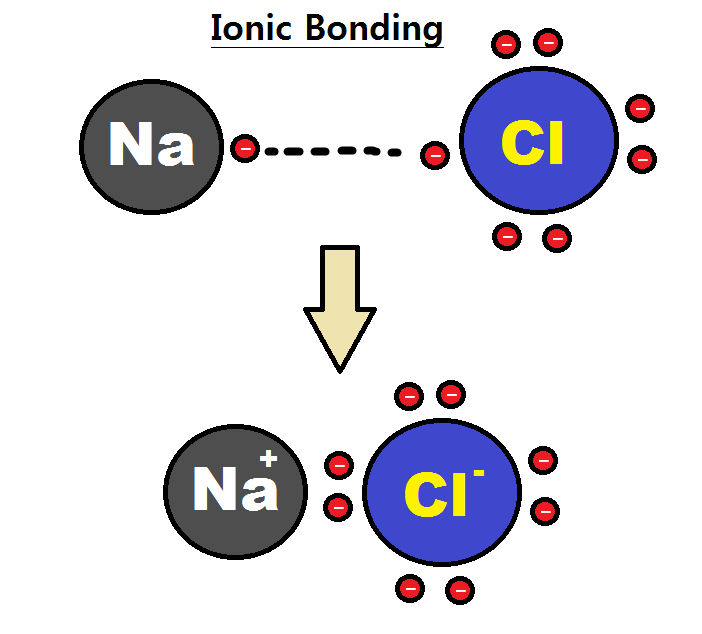
, such as sodium
Sodium is a chemical element with the symbol Na (from Latin ''natrium'') and atomic number 11. It is a soft, silvery-white, highly reactive metal. Sodium is an alkali metal, being in group 1 of the periodic table. Its only stable ...
and chlorine
Chlorine is a chemical element with the symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine i ...
in NaCl
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions. With molar masses of 22.99 and 35.45 g/ ...
. Sodium would give an electron to chlorine, forming a positively charged sodium ion and a negatively charged chloride ion.
Covalent bond
 In a true
In a true covalent bond
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between ato ...
, the electrons are shared evenly between the two atoms of the bond; there is little or no charge separation. Covalent bond
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between ato ...
s are generally formed between two nonmetals. There are several types of covalent bonds: in polar covalent bonds, electrons are more likely to be found around one of the two atoms, whereas in nonpolar covalent bonds, electrons are evenly shared. Homonuclear
Homonuclear molecules, or homonuclear species, are molecules composed of only one element. Homonuclear molecules may consist of various numbers of atoms. The size of the molecule an element can form depends on the element's properties, and some el ...
diatomic molecule
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear. O ...
s are purely covalent. The polarity of a covalent bond is determined by the electronegativities
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the d ...
of each atom and thus a polar covalent bond has a dipole moment pointing from the partial positive end to the partial negative end. Polar covalent bonds represent an intermediate type in which the electrons are neither completely transferred from one atom to another nor evenly shared.
Metallic bond
Metallic bond
Metallic bonding is a type of chemical bonding that arises from the electrostatic attractive force between conduction electrons (in the form of an electron cloud of delocalized electrons) and positively charged metal ions. It may be des ...
s generally form within a pure metal or metal alloy
An alloy is a mixture of chemical elements of which at least one is a metal. Unlike chemical compounds with metallic bases, an alloy will retain all the properties of a metal in the resulting material, such as electrical conductivity, ductili ...
. Metallic electrons are generally delocalized; the result is a large number of free electrons around positive nuclei, sometimes called an electron sea.
Bond formation
Comparison of the bond lengths between carbon and oxygen in a double and triple bond.
Biochemistry
disulfide bonds
In biochemistry, a disulfide (or disulphide in British English) refers to a functional group with the structure . The linkage is also called an SS-bond or sometimes a disulfide bridge and is usually derived by the coupling of two thiol groups. In ...
give proteins and DNA their structure. Protein
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, res ...
s derive their structure from the intramolecular forces that shape them and hold them together. The main source of structure in these molecules is the interaction between the amino acid
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although hundreds of amino acids exist in nature, by far the most important are the alpha-amino acids, which comprise proteins. Only 22 alpha ...
residues that form the foundation of proteins. The interactions between residues of the same proteins forms the secondary structure of the protein, allowing for the formation of beta sheet
The beta sheet, (β-sheet) (also β-pleated sheet) is a common motif of the regular protein secondary structure. Beta sheets consist of beta strands (β-strands) connected laterally by at least two or three backbone hydrogen bonds, forming a ge ...
s and alpha helices
The alpha helix (α-helix) is a common motif in the secondary structure of proteins and is a right hand-helix conformation in which every backbone N−H group hydrogen bonds to the backbone C=O group of the amino acid located four residues ear ...
, which are important structures for proteins and in the case of alpha helices, for DNA.
See also
*Chemical bond
A chemical bond is a lasting attraction between atoms or ions that enables the formation of molecules and crystals. The bond may result from the electrostatic force between oppositely charged ions as in ionic bonds, or through the sharing of ...
*Intermolecular force
An intermolecular force (IMF) (or secondary force) is the force that mediates interaction between molecules, including the electromagnetic forces of attraction
or repulsion which act between atoms and other types of neighbouring particles, e.g. ...
References
{{DEFAULTSORT:Intramolecular Force Chemical bonding