Azimuthal Quantum Number on:
[Wikipedia]
[Google]
[Amazon]
 The azimuthal quantum number is a quantum number for an
The azimuthal quantum number is a quantum number for an
 An atomic electron's
An atomic electron's
 Due to the
Due to the
Development of the Bohr atom
{{electron configuration navbox Angular momentum Atomic physics Quantum numbers Rotational symmetry de:Nebenquantenzahl
 The azimuthal quantum number is a quantum number for an
The azimuthal quantum number is a quantum number for an atomic orbital
In atomic theory and quantum mechanics, an atomic orbital is a function describing the location and wave-like behavior of an electron in an atom. This function can be used to calculate the probability of finding any electron of an atom in any ...
that determines its orbital angular momentum and describes the shape of the orbital. The azimuthal quantum number is the second of a set of quantum numbers that describe the unique quantum state
In quantum physics, a quantum state is a mathematical entity that provides a probability distribution for the outcomes of each possible measurement on a system. Knowledge of the quantum state together with the rules for the system's evolution i ...
of an electron (the others being the principal quantum number, the magnetic quantum number, and the spin quantum number). It is also known as the orbital angular momentum quantum number, orbital quantum number or second quantum number, and is symbolized as ℓ (pronounced ''ell'').
Derivation
Connected with the energy states of the atom's electrons are four quantum numbers: ''n'', ''ℓ'', ''m''''ℓ'', and ''m''''s''. These specify the complete, unique quantum state of a singleelectron
The electron ( or ) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary particles because they have n ...
in an atom
Every atom is composed of a nucleus and one or more electrons bound to the nucleus. The nucleus is made of one or more protons and a number of neutrons. Only the most common variety of hydrogen has no neutrons.
Every solid, liquid, gas, a ...
, and make up its wavefunction or ''orbital''. When solving to obtain the wave function, the Schrödinger equation
The Schrödinger equation is a linear partial differential equation that governs the wave function of a quantum-mechanical system. It is a key result in quantum mechanics, and its discovery was a significant landmark in the development of th ...
reduces to three equations that lead to the first three quantum numbers. Therefore, the equations for the first three quantum numbers are all interrelated. The azimuthal quantum number arose in the solution of the polar part of the wave equation as shown below , reliant on the spherical coordinate system, which generally works best with models having some glimpse of spherical symmetry.
angular momentum
In physics, angular momentum (rarely, moment of momentum or rotational momentum) is the rotational analog of linear momentum. It is an important physical quantity because it is a conserved quantity—the total angular momentum of a closed syst ...
, ''L'', is related to its quantum number ''ℓ'' by the following equation:
where ''ħ'' is the reduced Planck's constant, L2 is the orbital angular momentum operator and is the wavefunction of the electron. The quantum number ''ℓ'' is always a non-negative integer: 0, 1, 2, 3, etc. L has no real meaning except in its use as the angular momentum operator
In quantum mechanics, the angular momentum operator is one of several related operators analogous to classical angular momentum. The angular momentum operator plays a central role in the theory of atomic and molecular physics and other quantum p ...
. When referring to angular momentum, it is better to simply use the quantum number ''ℓ''.
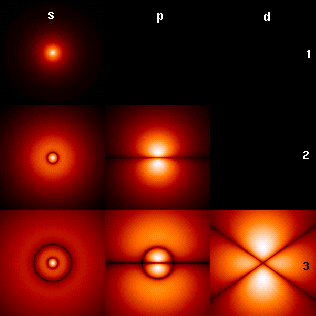
Atomic orbitals have distinctive shapes denoted by letters. In the illustration, the letters s, p, and d (a convention originating in spectroscopy) describe the shape of the atomic orbital
In atomic theory and quantum mechanics, an atomic orbital is a function describing the location and wave-like behavior of an electron in an atom. This function can be used to calculate the probability of finding any electron of an atom in any ...
.
Their wavefunctions take the form of spherical harmonic
In mathematics and physical science, spherical harmonics are special functions defined on the surface of a sphere. They are often employed in solving partial differential equations in many scientific fields.
Since the spherical harmonics form ...
s, and so are described by Legendre polynomials
In physical science and mathematics, Legendre polynomials (named after Adrien-Marie Legendre, who discovered them in 1782) are a system of complete and orthogonal polynomials, with a vast number of mathematical properties, and numerous applica ...
. The various orbitals relating to different values of ''ℓ'' are sometimes called sub-shells, and are referred to by lowercase Latin letters
The Latin script, also known as Roman script, is an alphabetic writing system based on the letters of the classical Latin alphabet, derived from a form of the Greek alphabet which was in use in the ancient Greek city of Cumae, in southern ...
(chosen for historical reasons), as follows:
Each of the different angular momentum states can take 2(2''ℓ'' + 1) electrons. This is because the third quantum number ''m''ℓ (which can be thought of loosely as the quantized projection of the angular momentum vector on the z-axis) runs from −''ℓ'' to ''ℓ'' in integer units, and so there are 2''ℓ'' + 1 possible states. Each distinct ''n'', ''ℓ'', ''m''ℓ orbital can be occupied by two electrons with opposing spins (given by the quantum number ''m''s = ±), giving 2(2''ℓ'' + 1) electrons overall. Orbitals with higher ''ℓ'' than given in the table are perfectly permissible, but these values cover all atoms so far discovered.
For a given value of the principal quantum number ''n'', the possible values of ''ℓ'' range from 0 to ; therefore, the shell only possesses an s subshell and can only take 2 electrons, the shell possesses an s and a p subshell and can take 8 electrons overall, the shell possesses s, p, and d subshells and has a maximum of 18 electrons, and so on.
A simplistic one-electron model results in energy levels depending on the principal number alone. In more complex atoms these energy levels split for all , placing states of higher ''ℓ'' above states of lower ''ℓ''. For example, the energy of 2p is higher than of 2s, 3d occurs higher than 3p, which in turn is above 3s, etc. This effect eventually forms the block structure of the periodic table. No known atom possesses an electron having ''ℓ'' higher than three (f) in its ground state.
The angular momentum quantum number, ''ℓ'', governs the number of planar nodes going through the nucleus. A planar node can be described in an electromagnetic wave as the midpoint between crest and trough, which has zero magnitudes. In an s orbital, no nodes go through the nucleus, therefore the corresponding azimuthal quantum number ''ℓ'' takes the value of 0. In a p orbital, one node traverses the nucleus and therefore ''ℓ'' has the value of 1. has the value .
Depending on the value of ''n'', there is an angular momentum quantum number ''ℓ'' and the following series. The wavelengths listed are for a hydrogen atom:
: , Lyman series (ultraviolet)
: , Balmer series (visible)
: , Ritz–Paschen series ( near infrared)
: , Brackett series ( short-wavelength infrared)
: , Pfund series
The emission spectrum of atomic hydrogen has been divided into a number of spectral series, with wavelengths given by the Rydberg formula. These observed spectral lines are due to the electron making transitions between two energy levels in an ...
( mid-wavelength infrared).
Addition of quantized angular momenta
Given a quantized total angular momentum which is the sum of two individual quantized angular momenta and , : the quantum number associated with its magnitude can range from to in integer steps where and are quantum numbers corresponding to the magnitudes of the individual angular momenta.Total angular momentum of an electron in the atom
spin–orbit interaction
In quantum physics, the spin–orbit interaction (also called spin–orbit effect or spin–orbit coupling) is a relativistic interaction of a particle's spin with its motion inside a potential. A key example of this phenomenon is the spin–orb ...
in the atom, the orbital angular momentum no longer commutes with the Hamiltonian, nor does the spin. These therefore change over time. However the total angular momentum J does commute with the one-electron Hamiltonian and so is constant. J is defined through
:
L being the orbital angular momentum and S the spin. The total angular momentum satisfies the same commutation relations as orbital angular momentum, namely
:
from which follows
:
where ''J''i stand for ''J''x, ''J''y, and ''J''z.
The quantum numbers describing the system, which are constant over time, are now ''j'' and ''m''''j'', defined through the action of J on the wavefunction
:
:
So that ''j'' is related to the norm of the total angular momentum and ''m''''j'' to its projection along a specified axis. The ''j'' number has a particular importance for relativistic quantum chemistry
Relativistic quantum chemistry combines relativistic mechanics with quantum chemistry to calculate elemental properties and structure, especially for the heavier elements of the periodic table. A prominent example is an explanation for the color of ...
, often featuring in subscript in electron configuration of superheavy elements.
As with any angular momentum in quantum mechanics, the projection of J along other axes cannot be co-defined with ''J''z, because they do not commute.
Relation between new and old quantum numbers
''j'' and ''m''''j'', together with theparity
Parity may refer to:
* Parity (computing)
** Parity bit in computing, sets the parity of data for the purpose of error detection
** Parity flag in computing, indicates if the number of set bits is odd or even in the binary representation of the ...
of the quantum state
In quantum physics, a quantum state is a mathematical entity that provides a probability distribution for the outcomes of each possible measurement on a system. Knowledge of the quantum state together with the rules for the system's evolution i ...
, replace the three quantum numbers ''ℓ'', ''m''''ℓ'' and ''m''''s'' (the projection of the spin along the specified axis). The former quantum numbers can be related to the latter.
Furthermore, the eigenvectors of ''j'', ''s'', ''m''''j'' and parity, which are also eigenvectors of the Hamiltonian, are linear combinations of the eigenvectors of ''ℓ'', ''s'', ''m''''ℓ'' and ''m''''s''.
List of angular momentum quantum numbers
* Intrinsic (or spin) angular momentum quantum number, or simply spin quantum number * orbital angular momentum quantum number (the subject of this article) * magnetic quantum number, related to the orbital momentum quantum number * total angular momentum quantum numberHistory
The azimuthal quantum number was carried over from the Bohr model of the atom, and was posited by Arnold Sommerfeld. The Bohr model was derived fromspectroscopic analysis
Spectroscopy is the field of study that measures and interprets the electromagnetic spectra that result from the interaction between electromagnetic radiation and matter as a function of the wavelength or frequency of the radiation. Matte ...
of the atom in combination with the Rutherford atomic model. The lowest quantum level was found to have an angular momentum of zero. Orbits with zero angular momentum were considered as oscillating charges in one dimension and so described as "pendulum" orbits, but were not found in nature. In three-dimensions the orbits become spherical without any nodes crossing the nucleus, similar (in the lowest-energy state) to a skipping rope that oscillates in one large circle.
See also
*Angular momentum operator
In quantum mechanics, the angular momentum operator is one of several related operators analogous to classical angular momentum. The angular momentum operator plays a central role in the theory of atomic and molecular physics and other quantum p ...
* Introduction to quantum mechanics
* Particle in a spherically symmetric potential
* Angular momentum coupling
* Clebsch–Gordan coefficients
References
External links
Development of the Bohr atom
{{electron configuration navbox Angular momentum Atomic physics Quantum numbers Rotational symmetry de:Nebenquantenzahl