|
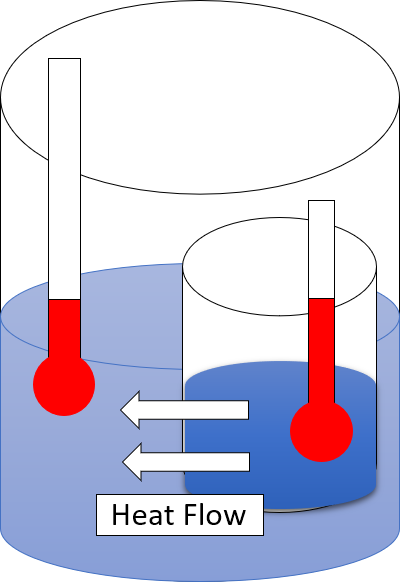
Zeroth Law Of Thermodynamics
The zeroth law of thermodynamics is one of the four principal laws of thermodynamics. It provides an independent definition of temperature without reference to entropy, which is defined in the second law. The law was established by Ralph H. Fowler in the 1930s, long after the first, second, and third laws were widely recognized. The zeroth law states that if two thermodynamic systems are in thermal equilibrium with each other, and also separately in thermal equilibrium with a third system, then the three systems are in thermal equilibrium with each other.Buchdahl, H.A. (1966). ''The Concepts of Classical Thermodynamics'', Cambridge University Press, Cambridge, p. 29: "... if each of two systems is in equilibrium with a third system then they are in equilibrium with each other." Two systems are said to be in thermal equilibrium if they are linked by a wall permeable only to heat, and they do not change over time. Another formulation by Maxwell is "All heat is of the same kind". ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Laws Of Thermodynamics
The laws of thermodynamics are a set of scientific laws which define a group of physical quantities, such as temperature, energy, and entropy, that characterize thermodynamic systems in thermodynamic equilibrium. The laws also use various parameters for thermodynamic processes, such as thermodynamic work and heat, and establish relationships between them. They state empirical facts that form a basis of precluding the possibility of certain phenomena, such as perpetual motion. In addition to their use in thermodynamics, they are important fundamental laws of physics in general, and are applicable in other natural sciences. Traditionally, thermodynamics has recognized three fundamental laws, simply named by an ordinal identification, the first law, the second law, and the third law.Guggenheim, E.A. (1985). ''Thermodynamics. An Advanced Treatment for Chemists and Physicists'', seventh edition, North Holland, Amsterdam, .Kittel, C. Kroemer, H. (1980). ''Thermal Physics'', second edit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Symmetry
Symmetry (from grc, συμμετρία "agreement in dimensions, due proportion, arrangement") in everyday language refers to a sense of harmonious and beautiful proportion and balance. In mathematics, "symmetry" has a more precise definition, and is usually used to refer to an object that is invariant under some transformations; including translation, reflection, rotation or scaling. Although these two meanings of "symmetry" can sometimes be told apart, they are intricately related, and hence are discussed together in this article. Mathematical symmetry may be observed with respect to the passage of time; as a spatial relationship; through geometric transformations; through other kinds of functional transformations; and as an aspect of abstract objects, including theoretic models, language, and music. This article describes symmetry from three perspectives: in mathematics, including geometry, the most familiar type of symmetry for many people; in science and nature ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
James Serrin
James Burton Serrin (1 November 1926, Chicago, Illinois – 23 August 2012, Minneapolis, Minnesota) was an American mathematician, and a professor at University of Minnesota. Life He received his doctorate from Indiana University in 1951 under the supervision of David Gilbarg. From 1954 till 1995 he was on the faculty of the University of Minnesota. Work He is known for his contributions to continuum mechanics, nonlinear analysis, and partial differential equations. Awards and honors He was elected a member of the National Academy of Sciences in 1980. Selected works *. *. *. References External Links Donald G. Aronson and Hans F. Weinberger, "James B. Serrin", Biographical Memoirs of the National Academy of Sciences (2016) See also *American mathematicians American(s) may refer to: * American, something of, from, or related to the United States of America, commonly known as the "United States" or "America" ** Americans, citizens and nationals of the United States o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Meghnad Saha
Meghnad Saha (6 October 1893 – 16 February 1956) was an Indian astrophysicist who developed the Saha ionization equation, used to describe chemical and physical conditions in stars. His work allowed astronomers to accurately relate the spectral classes of stars to their actual temperatures. He was elected to the Parliament of India in 1952 from Calcutta. Biography Meghnad Saha was born in 1893 in a very poor family in Shaoratoli, a village near Dhaka-Bikrampur, in the former Bengal Presidency of British India (in present-day Village- Shaoratoli, Thana- Kaliakair, District- Gazipur, Bangladesh). Son of Jagannath Saha (a grocer) and Smt. Bhubneshwari Devi, Meghnad struggled to rise in life. During his early schooling he was forced to leave Dhaka Collegiate School because he participated in the Swadeshi movement. He earned his Indian School Certificate from Dhaka College. He was also a student at the Presidency College, Kolkata and Rajabazar Science College CU. As a stude ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Arnold Sommerfeld
Arnold Johannes Wilhelm Sommerfeld, (; 5 December 1868 – 26 April 1951) was a German theoretical physicist who pioneered developments in atomic and quantum physics, and also educated and mentored many students for the new era of theoretical physics. He served as doctoral supervisor for many Nobel Prize winners in physics and chemistry (only J. J. Thomson's record of mentorship is comparable to his). He introduced the second quantum number (azimuthal quantum number) and the third quantum number (magnetic quantum number). He also introduced the fine-structure constant and pioneered X-ray wave theory. Early life and education Sommerfeld was born in 1868 to a family with deep ancestral roots in Prussia. His mother Cäcilie Matthias (1839–1902) was the daughter of a Potsdam builder. His father Franz Sommerfeld (1820–1906) was a physician from a leading family in Königsberg, where Arnold's grandfather had resettled from the hinterland in 1822 for a career as Court Postal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mole (unit)
The mole, symbol mol, is the unit of amount of substance in the International System of Units (SI). The quantity amount of substance is a measure of how many elementary entities of a given substance are in an object or sample. The mole is defined as containing exactly elementary entities. Depending on what the substance is, an elementary entity may be an atom, a molecule, an ion, an ion pair, or a subatomic particle such as an electron. For example, 10 moles of water (a chemical compound) and 10 moles of mercury (a chemical element), contain equal amounts of substance and the mercury contains exactly one atom for each molecule of the water, despite the two having different volumes and different masses. The number of elementary entities in one mole is known as the Avogadro number, which is the approximate number of nucleons (protons or neutrons) in one gram of ordinary matter. The previous definition of a mole was simply the number of elementary entities equal to that of 12 gram ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimension
In physics and mathematics, the dimension of a Space (mathematics), mathematical space (or object) is informally defined as the minimum number of coordinates needed to specify any Point (geometry), point within it. Thus, a Line (geometry), line has a dimension of one (1D) because only one coordinate is needed to specify a point on itfor example, the point at 5 on a number line. A Surface (mathematics), surface, such as the Boundary (mathematics), boundary of a Cylinder (geometry), cylinder or sphere, has a dimension of two (2D) because two coordinates are needed to specify a point on itfor example, both a latitude and longitude are required to locate a point on the surface of a sphere. A two-dimensional Euclidean space is a two-dimensional space on the Euclidean plane, plane. The inside of a cube, a cylinder or a sphere is three-dimensional (3D) because three coordinates are needed to locate a point within these spaces. In classical mechanics, space and time are different categ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermodynamic Temperature
Thermodynamic temperature is a quantity defined in thermodynamics as distinct from kinetic theory or statistical mechanics. Historically, thermodynamic temperature was defined by Kelvin in terms of a macroscopic relation between thermodynamic work and heat transfer as defined in thermodynamics, but the kelvin was redefined by international agreement in 2019 in terms of phenomena that are now understood as manifestations of the kinetic energy of free motion of microscopic particles such as atoms, molecules, and electrons. From the thermodynamic viewpoint, for historical reasons, because of how it is defined and measured, this microscopic kinetic definition is regarded as an "empirical" temperature. It was adopted because in practice it can generally be measured more precisely than can Kelvin's thermodynamic temperature. A thermodynamic temperature reading of zero is of particular importance for the third law of thermodynamics. By convention, it is reported on the ''Kelvin scale'' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Second Law Of Thermodynamics
The second law of thermodynamics is a physical law based on universal experience concerning heat and Energy transformation, energy interconversions. One simple statement of the law is that heat always moves from hotter objects to colder objects (or "downhill"), unless energy in some form is supplied to reverse the direction of heat flow. Another definition is: "Not all heat energy can be converted into Work (thermodynamics), work in a cyclic process."Young, H. D; Freedman, R. A. (2004). ''University Physics'', 11th edition. Pearson. p. 764. The second law of thermodynamics in other versions establishes the concept of entropy as a physical property of a thermodynamic system. It can be used to predict whether processes are forbidden despite obeying the requirement of conservation of energy as expressed in the first law of thermodynamics and provides necessary criteria for spontaneous processes. The second law may be formulated by the observation that the entropy of isolated systems ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Scale Of Temperature
Scale of temperature is a methodology of calibrating the physical quantity temperature in metrology. Empirical scales measure temperature in relation to convenient and stable parameters, such as the freezing and boiling point of water. Absolute temperature is based on thermodynamic principles: using the lowest possible temperature as the zero point, and selecting a convenient incremental unit. Celsius, Kelvin, and Fahrenheit are common temperature scales. Other scales used throughout history include Centigrade, Rankine, Rømer, Newton, Delisle, Réaumur, Gas Mark, Leiden and Wedgwood. Definition The zeroth law of thermodynamics describes thermal equilibrium between thermodynamic systems in form of an equivalence relation. Accordingly, all thermal systems may be divided into a quotient set, denoted as M. If the set M has the cardinality of c, then one can construct an injective function , by which every thermal system has a parameter associated with it such that when ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thermometer
A thermometer is a device that temperature measurement, measures temperature or a temperature gradient (the degree of hotness or coldness of an object). A thermometer has two important elements: (1) a temperature sensor (e.g. the bulb of a mercury-in-glass thermometer or the pyrometric sensor in an infrared thermometer) in which some change occurs with a change in temperature; and (2) some means of converting this change into a numerical value (e.g. the visible scale that is marked on a mercury-in-glass thermometer or the digital readout on an infrared model). Thermometers are widely used in technology and industry to monitor processes, in meteorology, in medicine, and in scientific research. History While an individual thermometer is able to measure degrees of hotness, the readings on two thermometers cannot be compared unless they conform to an agreed scale. Today there is an absolute thermodynamic temperature scale. Internationally agreed temperature scales are designed to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Real Number System
In mathematics, a real number is a number that can be used to measure a ''continuous'' one-dimensional quantity such as a distance, duration or temperature. Here, ''continuous'' means that values can have arbitrarily small variations. Every real number can be almost uniquely represented by an infinite decimal expansion. The real numbers are fundamental in calculus (and more generally in all mathematics), in particular by their role in the classical definitions of limits, continuity and derivatives. The set of real numbers is denoted or \mathbb and is sometimes called "the reals". The adjective ''real'' in this context was introduced in the 17th century by René Descartes to distinguish real numbers, associated with physical reality, from imaginary numbers (such as the square roots of ), which seemed like a theoretical contrivance unrelated to physical reality. The real numbers include the rational numbers, such as the integer and the fraction . The rest of the real numbers ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |