|
Tungsten Dichloride Dioxide
Tungsten dichloride dioxide, or Tungstyl chloride is the chemical compound with the formula W O2 Cl2. It is a yellow-colored solid. It is used as a precursor to other tungsten compounds. Like other tungsten halides, WO2Cl2 is sensitive to moisture, undergoing hydrolysis. Preparation WO2Cl2 is prepared by ligand redistribution reaction from tungsten trioxide and tungsten hexachloride: : 2 WO3 + WCl6 → 3 WO2Cl2 Using a two-zone tube furnace, a vacuum-sealed tube containing these solids is heated to 350 °C. The yellow product sublimes to the cooler end of the reaction tube. No redox occurs in this process. An alternative route highlights the oxophilicity of tungsten: :WCl6 + 2 O(Si(CH3)3)2 → 3 WO2Cl2 + 4 ClSi(CH3)3 This reaction, like the preceding one, proceeds via the intermediacy of WOCl4. Structure The compound is a polymer consisting of distorted octahedral W centres. The monomer is characterized by two short W-O distances, typical for a multiple W-O ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a hydroxyl group). Ethanol is a volatile, flammable, colorless liquid with a characteristic wine-like odor and pungent taste. It is a psychoactive recreational drug, the active ingredient in alcoholic drinks. Ethanol is naturally produced by the fermentation process of sugars by yeasts or via petrochemical processes such as ethylene hydration. It has medical applications as an antiseptic and disinfectant. It is used as a chemical solvent and in the synthesis of organic compounds, and as a fuel source. Ethanol also can be dehydrated to make ethylene, an important chemical feedstock. As of 2006, world production of ethanol was , coming mostly from Brazil and the U.S. Etymology ''Ethanol'' is the systematic name defined by the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyhedron (journal)
''Polyhedron'' is a peer-reviewed scientific journal covering the field of inorganic chemistry. It was established in 1955 as the ''Journal of Inorganic and Nuclear Chemistry'' and is published by Elsevier. Abstracting and indexing ''Polyhedron'' is abstracted and indexed in: According to the ''Journal Citation Reports'', the journal has a 2020 impact factor The impact factor (IF) or journal impact factor (JIF) of an academic journal is a scientometric index calculated by Clarivate that reflects the yearly mean number of citations of articles published in the last two years in a given journal, as ... of 3.052. References External links * {{Official Website, http://www.journals.elsevier.com/polyhedron/ Inorganic chemistry journals Elsevier academic journals Publications established in 1955 English-language journals ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethoxyethane
Dimethoxyethane, also known as glyme, monoglyme, dimethyl glycol, ethylene glycol dimethyl ether, dimethyl cellosolve, and DME, is a colorless, aprotic, and liquid ether that is used as a solvent, especially in batteries. Dimethoxyethane is miscible with water. Production Monoglyme is produced industrially by the reaction of dimethylether with ethylene oxide: :CH3OCH3 + CH2CH2O → CH3OCH2CH2OCH3 Applications as solvent and ligand left, 144px, Structure of the coordination complex NbCl3(dimethoxyethane)(3-hexyne).{{cite journal , doi=10.1021/ja8100837, title=New Tantalum Ligand-Free Catalyst System for Highly Selective Trimerization of Ethylene Affording 1-Hexene: New Evidence of a Metallacycle Mechanism, year=2009, last1=Arteaga-Müller, first1=Rocío, last2=Tsurugi, first2=Hayato, last3=Saito, first3=Teruhiko, last4=Yanagawa, first4=Masao, last5=Oda, first5=Seiji, last6=Mashima, first6=Kazushi, journal=Journal of the American Chemical Society, volume=131, issue=15, pages=53 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
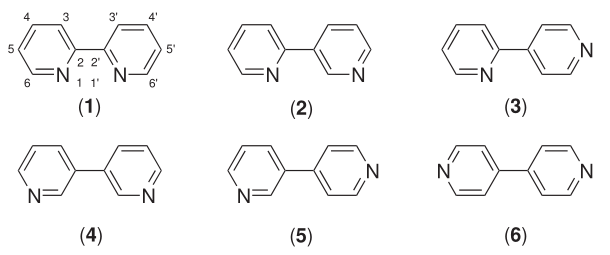
Bipyridine
Bipyridines also known as bipyridyls, dipyridyls, and dipyridines, are a family of chemical compounds with the formula (C5H4N)2, consisting of two pyridyl (C5H4N) rings. Pyridine is an aromatic nitrogen-containing heterocycle. Bipyridines are of significance in pesticides. Six isomers of bipyridine exist, but two are prominent: 2,2′-bipyridine is a popular ligand. 4,4'-Bipyridine is a precursor to the commercial herbicide paraquat. The bipyridines are all colourless solids, which are soluble in organic solvents and slightly soluble in water. 2,2′-Bipyridine 2,2′-Bipyridine (2,2′-bipy) is a chelating ligand that forms complexes with most transition metal ions that are of broad academic interest. Many of these complexes have distinctive optical properties, and some are of interest for analysis. Its complexes are used in studies of electron and energy transfer, supramolecular and materials chemistry, and catalysis. 2,2′-Bipyridine is used in the manufacture of diqu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lewis Acid
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any species that has a filled orbital containing an electron pair which is not involved in bonding but may form a dative bond with a Lewis acid to form a Lewis adduct. For example, NH3 is a Lewis base, because it can donate its lone pair of electrons. Trimethylborane (Me3B) is a Lewis acid as it is capable of accepting a lone pair. In a Lewis adduct, the Lewis acid and base share an electron pair furnished by the Lewis base, forming a dative bond. In the context of a specific chemical reaction between NH3 and Me3B, a lone pair from NH3 will form a dative bond with the empty orbital of Me3B to form an adduct NH3•BMe3. The terminology refers to the contributions of Gilbert N. Lewis. From p. 142: "We are inclined to think of substances as poss ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
WO2Cl2(dme)
Tungsten dichloride dioxide, or Tungstyl chloride is the chemical compound with the formula W O2 Cl2. It is a yellow-colored solid. It is used as a precursor to other tungsten compounds. Like other tungsten halides, WO2Cl2 is sensitive to moisture, undergoing hydrolysis. Preparation WO2Cl2 is prepared by ligand redistribution reaction from tungsten trioxide and tungsten hexachloride: : 2 WO3 + WCl6 → 3 WO2Cl2 Using a two-zone tube furnace, a vacuum-sealed tube containing these solids is heated to 350 °C. The yellow product sublimes to the cooler end of the reaction tube. No redox occurs in this process. An alternative route highlights the oxophilicity of tungsten: :WCl6 + 2 O(Si(CH3)3)2 → 3 WO2Cl2 + 4 ClSi(CH3)3 This reaction, like the preceding one, proceeds via the intermediacy of WOCl4. Structure The compound is a polymer consisting of distorted octahedral W centres. The monomer is characterized by two short W-O distances, typical for a multiple W-O b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tungsten(VI) Oxytetrachloride
Tungsten(VI) oxytetrachloride is the inorganic compound with the formula W O Cl4. This diamagnetic solid is used to prepare other complexes of tungsten. The yellow-green compound is soluble in nonpolar solvents but it reacts with alcohols and water and forms adducts with Lewis bases. Structure The solid consists of weakly associated square pyramidal monomers. The compound is classified as an oxyhalide. Synthesis and reactions WOCl4 is prepared from tungsten trioxide Tungsten(VI) oxide, also known as tungsten trioxide is a chemical compound of oxygen and the transition metal tungsten, with formula WO3. The compound is also called tungstic anhydride, reflecting its relation to tungstic acid . It is a light ...: :WO3 + 2 SOCl2 → WOCl4 + 2 SO2 :WCl6 + (Me3Si)2O → WOCl4 + 2 Me3SiCl WOCl4 is Lewis acidic. It is a precursor to catalysts used for polymerization of alkynes. References Chlorides Metal halides Oxychlorides Tungsten compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxyhalide
In chemistry, molecular oxohalides (oxyhalides) are a group of chemical compounds in which both oxygen and halogen atoms are attached to another chemical element A in a single molecule. They have the general formula , where X = fluorine (F), chlorine (Cl), bromine (Br), and/or iodine (I). The element A may be a main group element, a transition element or an actinide. The term ''oxohalide'', or ''oxyhalide'', may also refer to minerals and other crystalline substances with the same overall chemical formula, but having an ionic structure. Synthesis Oxohalides can be seen as compounds intermediate between oxides and halides. There are three general methods of synthesis: *Partial oxidation of a halide: *:2 PCl3 + O2 -> 2 POCl3 **In this example, the oxidation state increases by two and the electrical charge is unchanged. *Partial halogenation of an oxide: *:2 V2O5 + 6 Cl2 + 3 C -> 4 VOCl3 + 3 CO2 *Oxide replacement: *: rO42- + 2 Cl- + 4 H+ -> CrO2Cl2 + 4 H2O In addition, various ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oxophilicity
Oxophilicity is the tendency of certain chemical compounds to form oxides by hydrolysis or abstraction of an oxygen atom from another molecule, often from organic compounds. The term is often used to describe metal centers, commonly the early transition metals such as titanium, niobium, and tungsten. Oxophilicity is often stated to be related to the hardness of the element, within the HSAB theory ( hard and soft (Lewis) acids and bases), but it has been shown that oxophilicity depends more on the electronegativity and effective nuclear charge of the element than on its hardness. This explains why the early transition metals, whose electronegativities and effective nuclear charges are low, are very oxophilic. Many main group compounds are also oxophilic, such as derivatives of aluminium, silicon, and phosphorus(III). The handling of oxophilic compounds often requires air-free techniques. Examples Complexes of oxophilic metals typically are prone to hydrolysis. For example, the high v ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Compound
A chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. A molecule consisting of atoms of only one element is therefore not a compound. A compound can be transformed into a different substance by a chemical reaction, which may involve interactions with other substances. In this process, bonds between atoms may be broken and/or new bonds formed. There are four major types of compounds, distinguished by how the constituent atoms are bonded together. Molecular compounds are held together by covalent bonds; ionic compounds are held together by ionic bonds; intermetallic compounds are held together by metallic bonds; coordination complexes are held together by coordinate covalent bonds. Non-stoichiometric compounds form a disputed marginal case. A chemical formula specifies the number of atoms of each element in a compound molecule, using ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tube Furnace
A tube furnace is an electric heating device used to conduct syntheses and purifications of inorganic compounds and occasionally in organic synthesis. One possible design consists of a cylindrical cavity surrounded by heating coils that are embedded in a thermally insulating matrix. Temperature can be controlled via feedback from a thermocouple. More elaborate tube furnaces have two (or more) heating zones useful for transport experiments. Some digital temperature controllers provide an RS-232 interface, and permit the operator to program segments for uses like ramping, soaking, sintering, and more. Advanced materials in the heating elements, such as molybdenum disilicide (MoSi2) offered in certain models can now produce working temperatures up to 1800 °C. This facilitates more sophisticated applications. Common material for the reaction tubes include alumina, Pyrex, and fused quartz, or in the case of corrosive materials molybdenum or tungsten tubes can be used. The tub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
.png)