|
Protease Inhibitors
Protease inhibitors (PIs) are medications that act by interfering with protease, enzymes that cleave proteins. Some of the most well known are antiviral drugs widely used to treat HIV/AIDS, hepatitis C and COVID-19. These protease inhibitors prevent viral replication by selectively binding to viral proteases (e.g. HIV-1 protease) and blocking proteolytic cleavage of protein precursors that are necessary for the production of infectious Virion, viral particles. Protease inhibitors that have been developed and are currently used in clinical practice include: * Antiretroviral drug, Antiretroviral HIV-1 protease inhibitors—class stem ** Amprenavir ** Atazanavir ** Darunavir ** Fosamprenavir ** Indinavir ** Lopinavir ** Nelfinavir ** Ritonavir ** Saquinavir ** Tipranavir * Hepatitis C virus NS3 (HCV), NS3/NS4A, 4A protease inhibitors—class stem ** Asunaprevir ** Boceprevir ** Grazoprevir ** Glecaprevir ** Paritaprevir ** Simeprevir ** Telaprevir ** Voxilaprevir * 3-chymotrypsin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Enzyme Inhibition
An enzyme inhibitor is a molecule that binds to an enzyme and blocks its Enzyme activity, activity. Enzymes are proteins that speed up chemical reactions necessary for life, in which Substrate (biochemistry), substrate molecules are converted into Product (chemistry), products. An enzyme Enzyme catalysis, facilitates a specific chemical reaction by binding the substrate to its active site, a specialized area on the enzyme that accelerates the Rate-determining step, most difficult step of the reaction. An enzyme inhibitor stops ("inhibits") this process, either by binding to the enzyme's active site (thus preventing the substrate itself from binding) or by binding to another site on the enzyme such that the enzyme's catalysis of the reaction is blocked. Enzyme inhibitors may bind Reversible reaction, reversibly or irreversibly. Irreversible inhibitors form a Covalent bond, chemical bond with the enzyme such that the enzyme is inhibited until the chemical bond is broken. By cont ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Nelfinavir
Nelfinavir, sold under the brand name Viracept, is an antiretroviral medication used in the treatment of HIV/AIDS. Nelfinavir belongs to the class of drugs known as protease inhibitor (pharmacology), protease inhibitors (PIs) and like other PIs is almost always used in combination with other antiretroviral drugs. Nelfinavir is an orally bioavailable human immunodeficiency virus HIV-1 protease inhibitor (Ki = 2 nM) and is widely prescribed in combination with HIV reverse transcriptase inhibitors for the treatment of HIV infection. It was patented in 1992 and approved for medical use in 1997. Toxicity Common (>1%) side effects include insulin resistance, hyperglycemia and lipodystrophy. Nelfinavir can produce a range of adverse side effects. Flatulence, diarrhea, or abdominal pain are common (i.e. experienced by more than one in one hundred patients). Fatigue, urination, rash, mouth ulcers, or hepatitis are less frequent effects (experienced by one in one thousand to one in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Telaprevir
Telaprevir (VX-950), marketed under the brand names Incivek and Incivo, is a pharmaceutical drug for the treatment of hepatitis C co-developed by Vertex Pharmaceuticals and Johnson & Johnson. It is a member of a class of antiviral drugs known as protease inhibitors. Specifically, telaprevir inhibits the hepatitis C viral enzyme NS3/4A serine protease. Telaprevir is only indicated for use against hepatitis C genotype 1 viral infections and has not been proven to be safe or effective when used for other genotypes of the virus. The standard therapy of pegylated interferon and ribavirin is less effective than telaprevir in those with genotype 1. Clinical trials and approvals In a randomized controlled trial (PROVE3) of patients in whom standard treatment with peginterferon alfa-2a and ribavirin had failed, repeat treatment with the addition of telaprevir was more likely to have a sustained virological response (SVR) than repeat treatment with peginterferon alfa-2a and ribavirin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Simeprevir
Simeprevir, sold under the brand name Olysio among others, is a medication used in combination with other medications for the treatment of hepatitis C. It is specifically used for hepatitis C genotype 1 and 4. Medications it is used with include sofosbuvir or ribavirin and peginterferon-alfa. Cure rates are in 80s to 90s percent. It may be used in those who also have HIV/AIDS. It is taken by mouth once daily for typically 12 weeks. Common side effects include feeling tired, headache, rash, itchiness, and sensitivity to sunlight. In those with previous hepatitis B infection, active disease may recur. It is not recommended in those with significant liver problems. During pregnancy when used with ribavirin it may cause harm to the baby while when used with sofosbuvir its safety is unclear. Simeprevir is a HCV protease inhibitor. Simeprevir was developed by Medivir AB and Janssen Pharmaceutica. It was approved for medical use in the United States in 2013. It was removed from t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Paritaprevir
Paritaprevir (previously known as ABT-450) is an acylsulfonamide inhibitor of the NS3- 4A serine protease manufactured by Abbott Laboratories that shows promising results as a treatment of hepatitis C. When given in combination with ritonavir and ribavirin for 12 weeks, the rate of sustained virologic response at 24 weeks after treatment has been estimated to be 95% for those with hepatitis C virus genotype 1. Resistance to treatment with paritaprevir is uncommon, because it targets the binding site In biochemistry and molecular biology, a binding site is a region on a macromolecule such as a protein that binds to another molecule with specificity. The binding partner of the macromolecule is often referred to as a ligand. Ligands may includ ..., but has been seen to arise due to mutations at positions 155 and 168 in NS3. Paritaprevir was a component of Viekira Pak and Technivie. In May 2018, the FDA announced that Technivie and Viekira were to be discontinued. The discon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Glecaprevir
Glecaprevir ( INN,) is a hepatitis C virus (HCV) nonstructural (NS) protein 3/ 4A protease inhibitor that was identified jointly by AbbVie and Enanta Pharmaceuticals. It is being developed as a treatment of chronic hepatitis C infection in co-formulation with an HCV NS5A inhibitor pibrentasvir. Together they demonstrated potent antiviral activity against major HCV genotypes and high barriers to resistance ''in vitro''. On 19 December 2016, AbbVie submitted a new drug application to the U.S. Food and Drug Administration for the glecaprevir/pibrentasvir (trade name ''Mavyret'') regimen for the treatment of all major genotypes (1–6) of chronic hepatitis C. On 3 August 2017 the FDA approved the combination for hepatitis C treatment. In Europe, it was approved on 17 August 2017 for the same indication, under the trade name ''Maviret''. See also * Protease inhibitor (pharmacology) Protease inhibitors (PIs) are medications that act by interfering with enzymes that cleave p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Grazoprevir
Grazoprevir is a drug approved for the treatment of hepatitis C. It was developed by Merck and completed Phase III trials, used in combination with the NS5A replication complex inhibitor elbasvir under the trade name '' Zepatier'', either with or without ribavirin. Grazoprevir is a second generation hepatitis C virus protease inhibitor acting at the NS3/ 4A protease targets. It has good activity against a range of HCV genotype variants, including some that are resistant to most currently used antiviral medications. Side effects Side effects have only been assessed in the combination with elbasvir. Common side effects of the combination include feeling tired, nausea, reduced appetite, and headache. Low red blood cell count has occurred when co-administered with ribavirin in some cases. The most important risks are alanine transaminase elevation, hyperbilirubinemia, drug resistance development and drug interactions. Interactions Grazoprevir is transported by the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Boceprevir
Boceprevir ( INN, trade name Victrelis) is a protease inhibitor used to treat hepatitis caused by hepatitis C virus (HCV) genotype 1. It binds to the HCV nonstructural protein 3 active site. It was initially developed by Schering-Plough, then by Merck after it acquired Schering in 2009. It was approved by the FDA in May 2011. In January 2015, Merck announced that they would be voluntarily withdrawing Victrelis from the market due to the overwhelming superiority of newer direct-acting antiviral agents, such as ledipasvir/sofosbuvir. Clinical trials SPRINT-1 trial The SPRINT-1 trial was a phase-II trial of boceprevir in difficult-to-treat patients with HCV genotype 1. Study results were announced at the 44th annual meeting of the European Association for the Study of the Liver in Copenhagen in April, 2009. When used in combination with peginterferon alfa-2b and ribavirin, boceprevir resulted in significantly higher sustained viral response (SVR) rates in the most diff ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
Asunaprevir
Asunaprevir (formerly BMS-650032, brand name in Japan and Russia Sunvepra) is an experimental drug candidate for the treatment of hepatitis C. It was undergoing development by Bristol-Myers Squibb and has completed Phase III clinical trials in 2013. Asunaprevir is an inhibitor of the hepatitis C virus enzyme serine protease NS3. Asunaprevir is being tested in combination with pegylated interferon and ribavirin, as well as in interferon-free regimens with other direct-acting antiviral agents including daclatasvir Daclatasvir, sold under the brand name Daklinza, is an antiviral medication used in combination with other medications to treat hepatitis C (HCV). The other medications used in combination include sofosbuvir, ribavirin, and Interferon#Drug formu .... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
NS4A
Nonstructural protein 4A (NS4A) is a viral protein found in the hepatitis C virus The hepatitis C virus (HCV) is a small (55–65 nm in size), enveloped, positive-sense single-stranded RNA virus of the family ''Flaviviridae''. The hepatitis C virus is the cause of hepatitis C and some cancers such as liver cancer ( hepatoc .... It acts as a cofactor for the enzyme NS3. References Viral nonstructural proteins Hepatitis C virus {{virus-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
NS3 (HCV)
Nonstructural protein 3 (NS3), also known as p-70, is a viral nonstructural protein that is 70 kDa cleavage product of the hepatitis C virus polyprotein. It acts as a serine protease. C-terminal two-thirds of the protein also acts as helicase and nucleoside triphosphatase. First (N-terminal) 180 aminoacids of NS3 has additional role as cofactor domains for NS2 protein. See also * Boceprevir, sovaprevir, paritaprevir and telaprevir Telaprevir (VX-950), marketed under the brand names Incivek and Incivo, is a pharmaceutical drug for the treatment of hepatitis C co-developed by Vertex Pharmaceuticals and Johnson & Johnson. It is a member of a class of antiviral drugs known as ... - drugs targeting this protein References External links * http://www.uniprot.org/uniprot/Q91RS4 Viral nonstructural proteins Hepatitis C virus {{virus-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
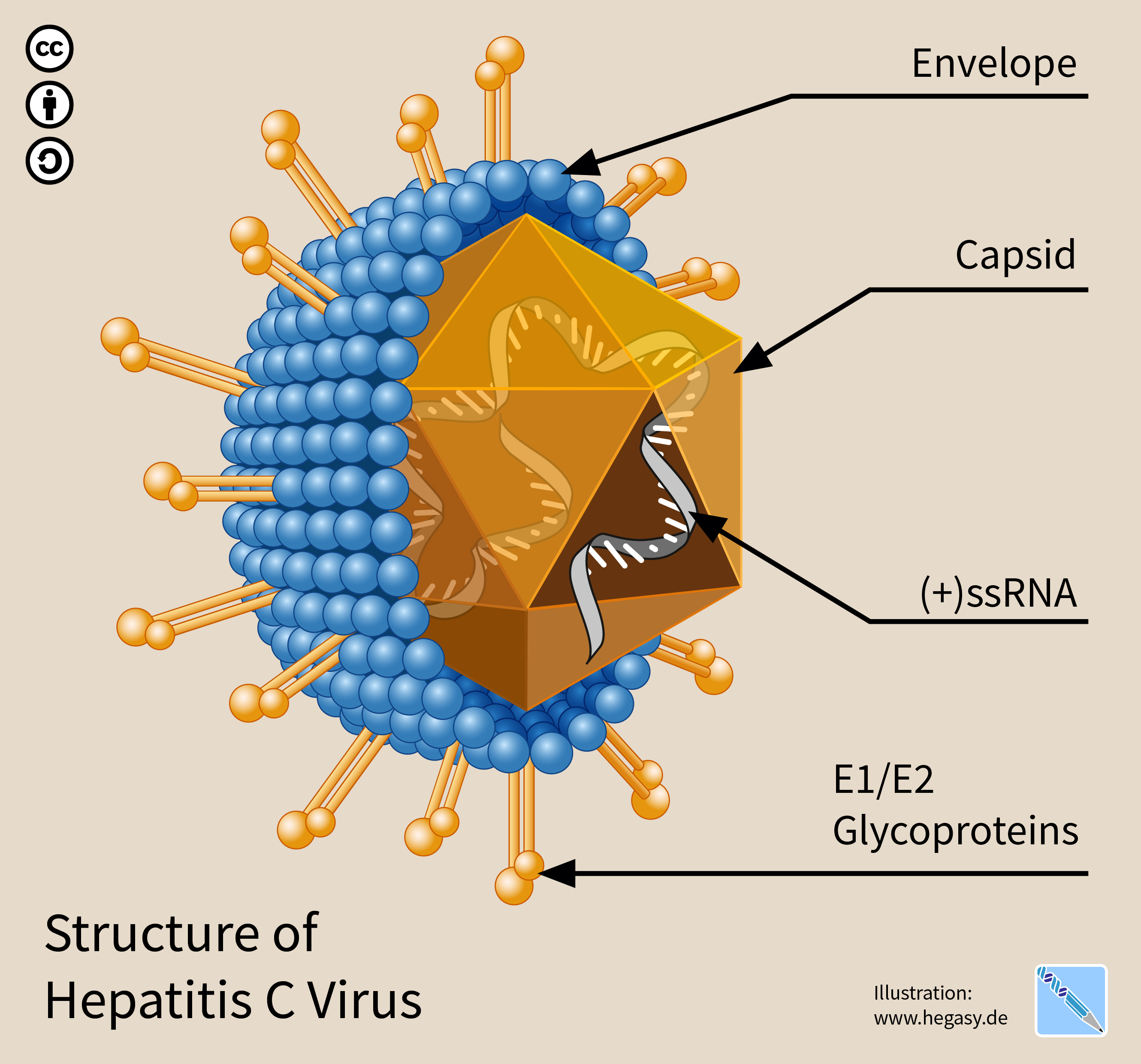
Hepatitis C Virus
The hepatitis C virus (HCV) is a small (55–65 nm in size), enveloped, positive-sense single-stranded RNA virus of the family ''Flaviviridae''. The hepatitis C virus is the cause of hepatitis C and some cancers such as liver cancer ( hepatocellular carcinoma, abbreviated HCC) and lymphomas in humans. Taxonomy The hepatitis C virus belongs to the genus '' Hepacivirus'', a member of the family ''Flaviviridae''. Before 2011, it was considered to be the only member of this genus. However a member of this genus has been discovered in dogs: canine hepacivirus. There is also at least one virus in this genus that infects horses. Several additional viruses in the genus have been described in bats and rodents. Structure The hepatitis C virus particle consists of a lipid membrane envelope that is 55 to 65 nm in diameter. Two viral envelope glycoproteins, E1 and E2, are embedded in the lipid envelope. They take part in viral attachment and entry into the cell. Within the e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |