|
Ethyl Nitrate
Ethyl nitrate is the ethyl ester of nitric acid and has the chemical formula . It is a colourless, volatile, explosive, and extremely flammable liquid. It is used in organic synthesis with use as a nitrating agent and as an intermediate in the preparation of some drugs, dyes, and perfumes. Like nitroglycerin, it's a vasodilator. Ethyl nitrate is found in the atmosphere, where it can react with other gases to form smog. The pollutant was originally thought to have been formed mainly by the combustion of fossil fuels. However recent analysis of ocean water samples reveal that in places where cool water rises from the deep, the water is saturated with alkyl nitrates, likely formed by natural processes. History and synthesis Reaction of ethanol with nitric acid was investigated since the Middle Ages, but the fact that it produces mostly ethyl nitrite was not discovered until the 19th century. Eugène Millon was the first to synthesize ethyl nitrate in 1843 by adding urea to the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkyl Nitrate
In organic chemistry, a nitrate ester is an organic functional group with the formula , where R stands for any organyl group. They are the esters of nitric acid and alcohols. A well-known example is nitroglycerin, which is not a ''nitro'' compound, despite its name. : Synthesis and reactions Nitrate esters are typically prepared by condensation of nitric acid and the alcohol: For example, the simplest nitrate ester, methyl nitrate, is formed by reaction of methanol and nitric acid in the presence of sulfuric acid: :CH3OH + HNO3 -> CH3ONO2 + H2O Formation of a nitrate ester is called a nitrooxylation (less commonly, nitroxylation). Most commonly, "mixed acid" (nitric and sulfuric acids) are used, but in the 1980s production of the nitrocellulose with magnesium nitrate as a dehydrating agent was started in the US. In laboratory, phosphoric acid and phosphorus pentoxide or acetic acid and its anhydride may be used for the same purpose, or the nitroxylation can be conducted in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Saturated Solution
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution. The extent of the solubility of a substance in a specific solvent is generally measured as the concentration of the solute in a saturated solution, one in which no more solute can be dissolved. At this point, the two substances are said to be at the solubility equilibrium. For some solutes and solvents, there may be no such limit, in which case the two substances are said to be " miscible in all proportions" (or just "miscible"). The solute can be a solid, a liquid, or a gas, while the solvent is usually solid or liquid. Both may be pure substances, or may themselves be solutions. Gases are always miscible in all proportions, except in very extreme situations,J. de Swaan Arons and G. A. M. Diepen (1966): "Gas—Gas Equilibria". ''Journal of Chemical Physics'', ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methyl Nitrate
Methyl nitrate is the methyl ester of nitric acid and has the chemical formula CH3NO3. It is a colourless explosive volatile liquid. Synthesis It can be produced by the condensation of nitric acid and methanol: :CH3OH + HNO3 → CH3NO3 + H2O A newer method uses methyl iodide and silver nitrate: : CH3I + AgNO3 → CH3NO3 + AgI Methyl nitrate can be produced on a laboratory or industrial scale either through the distillation of a mixture of methanol and nitric acid, or by the nitration of methanol by a mixture of sulfuric and nitric acids. The first procedure is not preferred due to the great explosion danger presented by the methyl nitrate vapour. The second procedure is essentially identical to that of making nitroglycerin. However, the process is usually run at a slightly higher temperature and the mixture is stirred mechanically on an industrial scale instead of with compressed air. Electrolytic production methods have been reported involving electrolyzing sodium acet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxylammonium Chloride
Hydroxylammonium chloride is a chemical compound with the formula . It is the hydrochloric acid salt of hydroxylamine (). Hydroxylamine is a biological intermediate in nitrification (biological oxidation of ammonia with oxygen into nitrite) and in anammox (biological oxidation of nitrite and ammonium into dinitrogen gas) which are important in the nitrogen cycle in soil and in wastewater treatment plants. Applications Hydroxylammonium chloride is used in organic synthesis for preparation of oximes and hydroxamic acids from carboxylic acids, N- and O- substituted hydroxylamines, and addition reactions of carbon-carbon double bond. During the acetyl bromide method of extracting lignin from lignocellulosic biomass, hydroxylammonium chloride can be used to remove bromine and polybromide from the solution. In surface treatments, it is used in the preparation of anti-skinning agents, corrosion inhibitors, and cleaner additives. It is also a starting material for pharmaceuticals an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tin(II) Chloride
Tin(II) chloride, also known as stannous chloride, is a white crystalline solid with the formula . It forms a stable dihydrate, but aqueous solutions tend to undergo hydrolysis, particularly if hot. SnCl2 is widely used as a reducing agent (in acid solution), and in electrolytic baths for tin-plating. Tin(II) chloride should not be confused with the other chloride of tin; tin(IV) chloride or stannic chloride (SnCl4). Chemical structure SnCl2 has a lone pair of electrons, such that the molecule in the gas phase is bent. In the solid state, crystalline SnCl2 forms chains linked via chloride bridges as shown. The dihydrate has three coordinates as well, with one water on the tin and another water on the first. The main part of the molecule stacks into double layers in the crystal lattice, with the "second" water sandwiched between the layers. Chemical properties Tin(II) chloride dissolves in less than its own mass of water. Dilute solutions are subject to hydrolysis, yieldi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
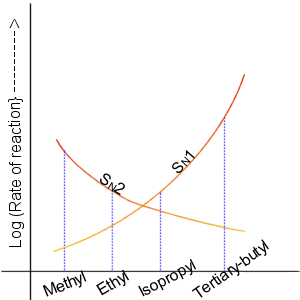
Nucleophilic Substitution
In chemistry, a nucleophilic substitution (SN) is a class of chemical reactions in which an electron-rich chemical species (known as a nucleophile) replaces a functional group within another electron-deficient molecule (known as the electrophile). The molecule that contains the electrophile and the leaving functional group is called the substrate. The most general form of the reaction may be given as the following: :\text\mathbf + \ce + \text\mathbf The electron pair (:) from the nucleophile (Nuc) attacks the substrate () and bonds with it. Simultaneously, the leaving group (LG) departs with an electron pair. The principal product in this case is . The nucleophile may be electrically neutral or negatively charged, whereas the substrate is typically neutral or positively charged. An example of nucleophilic substitution is the hydrolysis of an alkyl bromide, R-Br under basic conditions, where the attacking nucleophile is hydroxyl () and the leaving group is bromide (). :O ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Russian Chemical Bulletin
Russian(s) may refer to: *Russians (), an ethnic group of the East Slavic peoples, primarily living in Russia and neighboring countries *A citizen of Russia *Russian language, the most widely spoken of the Slavic languages *''The Russians'', a book by Hedrick Smith *Russian (comics), fictional Marvel Comics supervillain from ''The Punisher'' series *Russian (solitaire), a card game * "Russians" (song), from the album ''The Dream of the Blue Turtles'' by Sting *"Russian", from the album ''Tubular Bells 2003'' by Mike Oldfield *"Russian", from the album '' '' by Caravan Palace *Nik Russian, the perpetrator of a con committed in 2002 See also * *Russia (other) *Rus (other) *Rossiysky (other) *Russian River (other) *Rushen (other) Rushen may refer to: Places * Rushen, formally Kirk Christ Rushen, a historic parish of the Isle of Man ** Rushen (constituency), a House of Keys constituency of which the parish forms part ** Rushen (sheading ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethanol
Ethanol (also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound with the chemical formula . It is an Alcohol (chemistry), alcohol, with its formula also written as , or EtOH, where Et is the pseudoelement symbol for ethyl group, ethyl. Ethanol is a Volatility (chemistry), volatile, flammable, colorless liquid with a characteristic wine-like odor and pungent taste. As a psychoactive depressant, it is the active ingredient in alcoholic beverages, and the second most consumed drug globally behind caffeine. Ethanol is naturally produced by the fermentation process of sugars by yeasts or via petrochemical processes such as ethylene hydration. Historically it was used as a general anesthetic, and has modern medical applications as an antiseptic, disinfectant, solvent for some medications, and antidote for methanol poisoning and ethylene glycol poisoning. It is used as a chemical solvent and in the Chemical synthesis, synthesis of orga ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitryl Fluoride
Nitryl fluoride, NO2F, is a colourless gas and strong oxidizing agent, which is used as a fluorinating agent and has been proposed as an oxidiser in rocket propellants (though never flown). It is a molecular species, not ionic, consistent with its low boiling point. The structure features planar nitrogen with a short N-F bond length of 135 pm. Preparation Henri Moissan and Paul Lebeau recorded the preparation of nitryl fluoride in 1905 by the fluorination of nitrogen dioxide. This reaction is highly exothermic, which leads to contaminated products. The simplest method avoids fluorine gas but uses cobalt(III) fluoride: :NO2 + CoF3 → NO2F + CoF2 The CoF2 can be regenerated to CoF3. Other methods have been described. Thermodynamic properties The thermodynamic properties of this gas were determined by IR and Raman spectroscopy. The standard heat of formation of FNO2 is -19 ± 2 kcal/mol, but the compound becomes increasingly unstable at higher temperature. The homogeneou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrate Ester
In organic chemistry, a nitrate ester is an organic functional group with the formula , where R stands for any organyl group. They are the esters of nitric acid and alcohols. A well-known example is nitroglycerin, which is not a ''nitro'' compound, despite its name. : Synthesis and reactions Nitrate esters are typically prepared by condensation of nitric acid and the alcohol: For example, the simplest nitrate ester, methyl nitrate, is formed by reaction of methanol and nitric acid in the presence of sulfuric acid: :CH3OH + HNO3 -> CH3ONO2 + H2O Formation of a nitrate ester is called a nitrooxylation (less commonly, nitroxylation). Most commonly, "mixed acid" (nitric and sulfuric acids) are used, but in the 1980s production of the nitrocellulose with magnesium nitrate as a dehydrating agent was started in the US. In laboratory, phosphoric acid and phosphorus pentoxide or acetic acid and its anhydride may be used for the same purpose, or the nitroxylation can be conduct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrous Acid
Nitrous acid (molecular formula ) is a weak and monoprotic acid known only in solution, in the gas phase, and in the form of nitrite () salts. It was discovered by Carl Wilhelm Scheele, who called it " phlogisticated acid of niter". Nitrous acid is used to make diazonium salts from amines. The resulting diazonium salts are reagents in azo coupling reactions to give azo dyes. Structure In the gas phase, the planar nitrous acid molecule can adopt both a ''syn'' and an ''anti'' form. The ''anti'' form predominates at room temperature, and IR measurements indicate it is more stable by around 2.3 kJ/mol. p. 462. Image:Trans-nitrous-acid-2D-dimensions.png , Dimensions of the ''anti'' form(from the microwave spectrum) Image:Trans-nitrous-acid-3D-balls.png , Model of the ''anti'' form Image:Cis-nitrous-acid-3D-balls.png , ''syn'' form Preparation and decomposition Free, gaseous nitrous acid is unstable, rapidly disproportionating to nitric oxides: :2 HNO2 → NO2 + ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Urea
Urea, also called carbamide (because it is a diamide of carbonic acid), is an organic compound with chemical formula . This amide has two Amine, amino groups (–) joined by a carbonyl functional group (–C(=O)–). It is thus the simplest amide of carbamic acid. Urea serves an important role in the cellular metabolism of nitrogen-containing compounds by animals and is the main nitrogen-containing substance in the urine of mammals. ''Urea'' is Neo-Latin, , , itself from Proto-Indo-European ''*h₂worsom''. It is a colorless, odorless solid, highly soluble in water, and practically non-toxic ( is 15 g/kg for rats). Dissolved in water, it is neither acidic nor base (chemistry), alkaline. The body uses it in many processes, most notably metabolic waste#Nitrogen wastes, nitrogen excretion. The liver forms it by combining two ammonia molecules () with a carbon dioxide () molecule in the urea cycle. Urea is widely used in fertilizers as a source of nitrogen (N) and is an important ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |