|
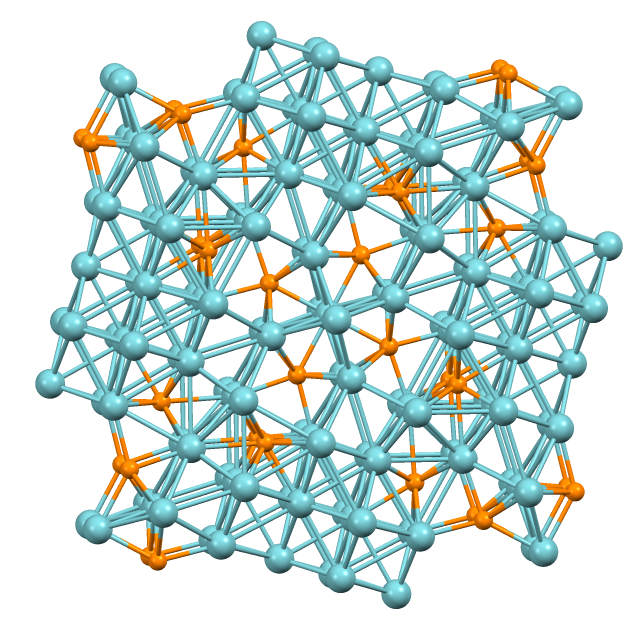
Chalcogel
A chalcogel or properly metal chalcogenide aerogel is an aerogel made from chalcogenides. Chalcogels preferentially absorb heavy metals, such as mercury, lead, and cadmium, from water. Sulfide chalcogels are also very good at desulfurization. Metal chalcogenide aerogels can be prepared from thiolysis or nanoparticle condensation and contain crystalline nanoparticles in the structure. The synthetic method can be extended to many thioanions, including tetrathiomolybdate-based chalcogels. Different metal ions have been used as linkers Co2+, Ni2+, Pb2+, Cd2+, Bi3+, Cr3+. When the gels are dried aerogels with high surface areas are obtained and the materials have multifunctional nature. For example, chalcogels are especially promising for gas separation. They were reported to exhibit high selectivity in CO2 and C2H6 over H2 and CH4 adsorption. The latter is relevant to exit gas stream composition of water gas shift reaction and steam reforming reactions (reactions widely used for H2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aerogels
Aerogels are a class of synthetic porous ultralight material derived from a gel, in which the liquid component for the gel has been replaced with a gas, without significant collapse of the gel structure. The result is a solid with extremely low density and extremely low thermal conductivity. Aerogels can be made from a variety of chemical compounds. Silica aerogels feel like fragile styrofoam to the touch, while some polymer-based aerogels feel like rigid foams. Aerogels are produced by extracting the liquid component of a gel through supercritical drying or freeze-drying. This allows the liquid to be slowly dried off without causing the solid matrix in the gel to collapse from capillary action, as would happen with conventional evaporation. The first aerogels were produced from silica gels. Kistler's later work involved aerogels based on alumina, chromia, and tin dioxide. Carbon aerogels were first developed in the late 1980s. History The first documented example of an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aerogel
Aerogels are a class of manufacturing, synthetic porous ultralight material derived from a gel, in which the liquid component for the gel has been replaced with a gas, without significant collapse of the gel structure. The result is a solid with extremely low density and extremely low thermal conductivity. Aerogels can be made from a variety of chemical compounds. Silica aerogels feel like fragile expanded polystyrene, styrofoam to the touch, while some polymer-based aerogels feel like rigid foams. Aerogels are produced by extracting the liquid component of a gel through supercritical drying or freeze-drying. This allows the liquid to be slowly dried off without causing the solid matrix in the gel to collapse from capillary action, as would happen with conventional evaporation. The first aerogels were produced from silica gels. Kistler's later work involved aerogels based on alumina, Chromium(III) oxide, chromia, and tin dioxide. Carbon aerogels were first developed in the late ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chalcogenide
: 220px, Cadmium sulfide, a prototypical metal chalcogenide, is used as a yellow pigment. A chalcogenide is a chemical compound consisting of at least one chalcogen anion and at least one more electropositive element. Although all group 16 elements of the periodic table are defined as chalcogens, the term chalcogenide is more commonly reserved for sulfides, selenides, tellurides, and polonides, rather than oxides. Many metal ores exist as chalcogenides. Photoconductive chalcogenide glasses are used in xerography. Some pigments and catalysts are also based on chalcogenides. The metal dichalcogenide MoS2 is a common solid lubricant. Alkali metal and alkaline earth chalcogenides Alkali metal and alkaline earth monochalcogenides are salt-like, being colourless and often water-soluble. The sulfides tend to undergo hydrolysis to form derivatives containing bisulfide (SH−) anions. The alkali metal chalcogenides often crystallize with the antifluorite structure and the alkal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mercury (element)
Mercury is a chemical element; it has Symbol (chemistry), symbol Hg and atomic number 80. It is commonly known as quicksilver. A Heavy metal element, heavy, silvery d-block element, mercury is the only metallic element that is known to be liquid at standard temperature and pressure; the only other element that is liquid under these conditions is the halogen bromine, though metals such as caesium, gallium, and rubidium melt just above room temperature. Mercury occurs in deposits throughout the world mostly as cinnabar (mercuric sulfide). The red pigment vermilion is obtained by Mill (grinding), grinding natural cinnabar or synthetic mercuric sulfide. Exposure to mercury and mercury-containing organic compounds is toxic to the nervous system, immune system and kidneys of humans and other animals; mercury poisoning can result from exposure to water-soluble forms of mercury (such as mercuric chloride or methylmercury) either directly or through mechanisms of biomagnification. Mercu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lead
Lead () is a chemical element; it has Chemical symbol, symbol Pb (from Latin ) and atomic number 82. It is a Heavy metal (elements), heavy metal that is density, denser than most common materials. Lead is Mohs scale, soft and Ductility, malleable, and also has a relatively low melting point. When freshly cut, lead is a shiny gray with a hint of blue. It tarnishes to a dull gray color when exposed to air. Lead has the highest atomic number of any stable nuclide, stable element and three of its isotopes are endpoints of major nuclear decay chains of heavier elements. Lead is a relatively unreactive post-transition metal. Its weak metallic character is illustrated by its Amphoterism, amphoteric nature; lead and lead oxides react with acids and base (chemistry), bases, and it tends to form covalent bonds. Lead compounds, Compounds of lead are usually found in the +2 oxidation state rather than the +4 state common with lighter members of the carbon group. Exceptions are mostly limited ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cadmium
Cadmium is a chemical element; it has chemical symbol, symbol Cd and atomic number 48. This soft, silvery-white metal is chemically similar to the two other stable metals in group 12 element, group 12, zinc and mercury (element), mercury. Like zinc, it demonstrates oxidation state +2 in most of its compounds, and like mercury, it has a lower melting point than the transition metals in group 3 element, groups 3 through group 11 element, 11. Cadmium and its Congener (chemistry), congeners in group 12 are often not considered transition metals, in that they do not have partly filled ''d'' or ''f'' electron shells in the elemental or common oxidation states. The average concentration of cadmium in Earth's crust is between 0.1 and 0.5 parts per million (ppm). It was discovered in 1817 simultaneously by Friedrich Stromeyer, Stromeyer and Karl Samuel Leberecht Hermann, Hermann, both in Germany, as an impurity in zinc carbonate. Cadmium occurs as a minor component in most zinc ores and i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Desulfurization
Desulfurization or desulphurisation is a chemical process for the removal of sulfur from a material. The term usually refers to the removal of sulfur from a molecule or a material by hydrogenolysis: : Hydrogen is the ultimate sulfur acceptor. As applied to oil refinery streams, the conversion is known as hydrodesulfurization. These processes are of industrial and environmental importance as they provide the bulk of sulfur used in industry (Claus process and Contact process), sulfur-free compounds that could otherwise not be used in a great number of catalytic processes, and also reduce the release of harmful sulfur compounds into the environment. A laboratory scale hydrogenolysis is the desulfurization of thioketals by Raney nickel. In such cases, the hydrogen is contained within the reagent. Desulfurization without hydrogenolysis Desulfurization without hydrogenolysis is uncommon. Some trithiocarbonates can be coupled by desulfurization with phosphites or phosphines as S-ato ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrathiomolybdate
Tetrathiomolybdate, also spelled tiomolibdate (USAN 280px, "Samguk Sagi" Book 04. Silla's Records. In 512, Usan-guk(于山國)was Ulleungdo(鬱陵島) Usan-guk, or the State of Usan, occupied Ulleung-do and the adjacent islands during the Korean Three Kingdoms period. According to t ...), is the anion of the following salts: * Ammonium tetrathiomolybdate, a building block in bioinorganic chemistry * Bis-choline tetrathiomolybdate, a drug for the treatment of Wilson's disease {{sia Thiometallates ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Water Gas Shift Reaction
Water is an inorganic compound with the chemical formula . It is a transparent, tasteless, odorless, and nearly colorless chemical substance. It is the main constituent of Earth's hydrosphere and the fluids of all known living organisms (in which it acts as a solvent). It is vital for all known forms of life, despite not providing food energy or organic micronutrients. Its chemical formula, , indicates that each of its molecules contains one oxygen and two hydrogen atoms, connected by covalent bonds. The hydrogen atoms are attached to the oxygen atom at an angle of 104.45°. In liquid form, is also called "water" at standard temperature and pressure. Because Earth's environment is relatively close to water's triple point, water exists on Earth as a solid, a liquid, and a gas. It forms precipitation in the form of rain and aerosols in the form of fog. Clouds consist of suspended droplets of water and ice, its solid state. When finely divided, crystalline ice may preci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Waste
Radioactive waste is a type of hazardous waste that contains radioactive material. It is a result of many activities, including nuclear medicine, nuclear research, nuclear power generation, nuclear decommissioning, rare-earth mining, and nuclear weapons reprocessing. The storage and disposal of radioactive waste is regulated by government agencies in order to protect human health and the environment. Radioactive waste is broadly classified into 3 categories: low-level waste (LLW), such as paper, rags, tools, clothing, which contain small amounts of mostly short-lived radioactivity; intermediate-level waste (ILW), which contains higher amounts of radioactivity and requires some shielding; and high-level waste (HLW), which is highly radioactive and hot due to decay heat, thus requiring cooling and shielding. Spent nuclear fuel can be processed in nuclear reprocessing plants. One third of the total amount have already been reprocessed. With nuclear reprocessing 96% of the spent fue ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Technetium-99
Technetium-99 (99Tc) is an isotope of technetium that decays with a half-life of 211,000 years to stable ruthenium-99, emitting beta particles, but no gamma rays. It is the most significant long-lived fission product of uranium fission, producing the largest fraction of the total long-lived radiation emissions of nuclear waste. Technetium-99 has a fission product yield of 6.0507% for thermal neutron fission of uranium-235. The metastable technetium-99m (99mTc) is a short-lived (half-life about 6 hours) nuclear isomer used in nuclear medicine, produced from molybdenum-99. It decays by isomeric transition to technetium-99, a desirable characteristic, since the very long half-life and type of decay of technetium-99 imposes little further radiation burden on the body. Radiation The weak beta emission is stopped by the walls of laboratory glassware. Soft X-rays are emitted when the beta particles are stopped, but as long as the body is kept more than 30 cm away these should p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uranium-238
Uranium-238 ( or U-238) is the most common isotope of uranium found in nature, with a relative abundance of 99%. Unlike uranium-235, it is non-fissile, which means it cannot sustain a chain reaction in a thermal-neutron reactor. However, it is fissionable by fast neutrons, and is ''fertile'', meaning it can be transmuted to fissile plutonium-239. 238U cannot support a chain reaction because inelastic scattering reduces neutron energy below the range where fast fission of one or more next-generation nuclei is probable. Doppler broadening of 238U's neutron absorption resonances, increasing absorption as fuel temperature increases, is also an essential negative feedback mechanism for reactor control. Around 99.284% of natural uranium's mass is uranium-238, which has a half-life of 1.41 seconds (4.468 years, or 4.468 billion years). Due to its natural abundance and half-life relative to other radioactive elements, 238U produces ~40% of the radioactive heat produced wit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |