|
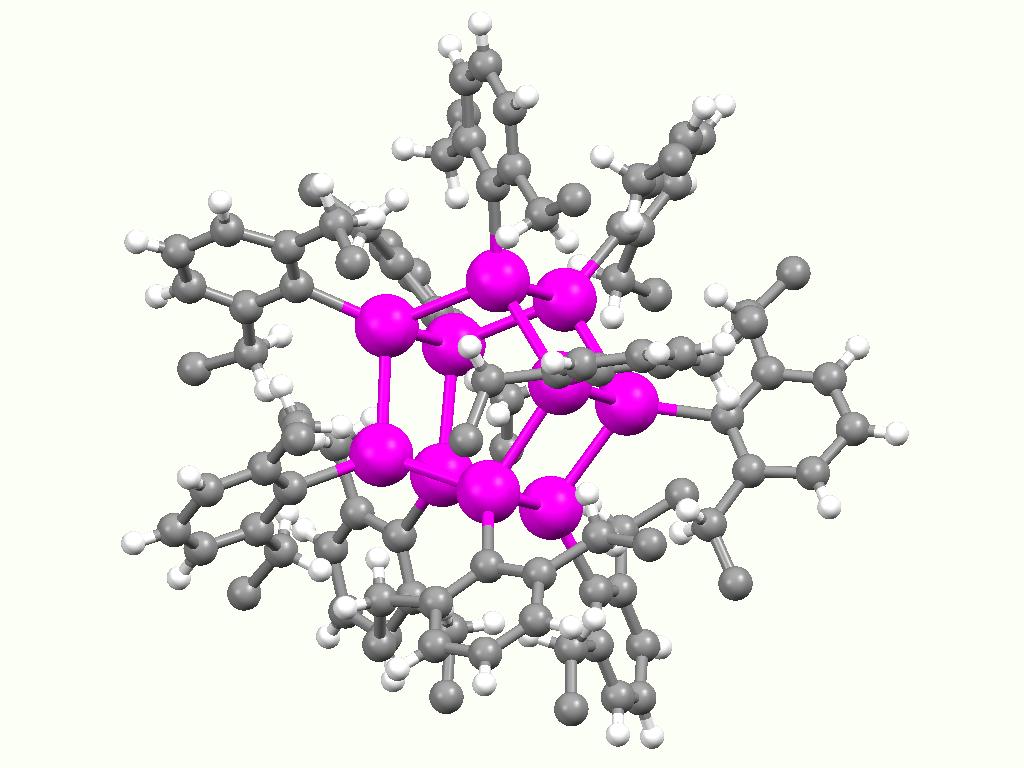
Butyltin Trichloride
Monobutyltin trichloride, also known as MBTC, is an organotin compound. It is a colorless oil that is soluble in organic solvents. Relative to other organotin compounds, MBTC is obscure and not widely used. Applications Glass coating Monobutyltin trichloride has been examined as a precursor to tin dioxide coatings on glass. Such coatings, especially when doped, are low-emissivity and transparent to visible light, reflect infrared light, and provide a high conductance and a low sheet resistance. For example, MBTC is used in the manufacturing process of glass containers such as those used for beers, spirits, and juices. These glass-making processes heat raw materials (sand, soda-ash, limestone, and recycled glass) to produce molten glass. The molten glob is cut into smaller pieces of uniform size, and are then pressed in a mold. MBTC is applied on the external surface of these containers, and then, the glass is annealed and coated with polyethylene. MBTC is a commonly used or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organotin Compound
Organotin chemistry is the scientific study of the synthesis and properties of organotin compounds or stannanes, which are organometallic compounds containing tin–carbon bonds. The first organotin compound was diethyltin diiodide (), discovered by Edward Frankland in 1849. The area grew rapidly in the 1900s, especially after the discovery of the Grignard reagents, which are useful for producing Sn–C bonds. The area remains rich with many applications in industry and continuing activity in the research laboratory. Structure Organotin compounds are generally classified according to their oxidation states. Tin(IV) compounds are much more common and more useful. Organic derivatives of tin(IV) The tetraorgano derivatives are invariably tetrahedral. Compounds of the type SnRR'R''R have been resolved into individual enantiomers. Organotin halides Organotin chlorides have the formula for values of ''n'' up to 3. Bromides, iodides, and fluorides are also known, but are less importan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trifluoroacetic Acid
Trifluoroacetic acid (TFA) is a synthetic organofluorine compound with the chemical formula CF3CO2H. It belongs to the subclass of per- and polyfluoroalkyl substances (PFASs) known as ultrashort-chain perfluoroalkyl acids (PFAAs). TFA is not produced biologically or abiotically and is commonly used in organic chemistry for various purposes. It is the most abundant PFAS found in the environment. It is a haloacetic acid, with all three of the acetyl group's hydrogen atoms replaced by fluorine atoms. It is a colorless liquid with a vinegar-like odor. TFA is a stronger acid than acetic acid, having an acid ionisation constant, ''K''a, that is approximately 34,000 times higher, as the highly electronegative fluorine atoms and consequent electron-withdrawing nature of the trifluoromethyl group weakens the oxygen-hydrogen bond (allowing for greater acidity) and stabilises the anionic conjugate base. Synthesis TFA is prepared industrially by the electrofluorination of ac ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Vapor Deposition
Chemical vapor deposition (CVD) is a vacuum deposition method used to produce high-quality, and high-performance, solid materials. The process is often used in the semiconductor industry to produce thin films. In typical CVD, the wafer (electronics), wafer (substrate) is exposed to one or more Volatility (chemistry), volatile wikt:precursor, precursors, which chemical reaction, react and/or chemical decomposition, decompose on the substrate surface to produce the desired deposit. Frequently, volatile by-products are also produced, which are removed by gas flow through the reaction chamber. Microfabrication processes widely use CVD to deposit materials in various forms, including: Single crystal, monocrystalline, polycrystalline, amorphous, and Epitaxy, epitaxial. These materials include: silicon (Silicon dioxide, dioxide, silicon carbide, carbide, silicon nitride, nitride, silicon oxynitride, oxynitride), carbon (carbon (fiber), fiber, carbon nanofibers, nanofibers, carbon nanot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Physical Vapor Deposition
Physical vapor deposition (PVD), sometimes called physical vapor transport (PVT), describes a variety of vacuum deposition methods which can be used to produce thin films and coatings on substrates including metals, ceramics, glass, and polymers. PVD is characterized by a process in which the material transitions from a condensed phase to a vapor phase and then back to a thin film condensed phase. The most common PVD processes are Sputter coating, sputtering and Evaporation (deposition), evaporation. PVD is used in the manufacturing of items which require thin films for optical, mechanical, electrical, acoustic or chemical functions. Examples include semiconductor devices such as thin-film solar cells, microelectromechanical devices such as thin film bulk acoustic resonator, aluminized Polyethylene terephthalate, PET film for food packaging and balloons, and titanium nitride coated cutting tools for metalworking. Besides PVD tools for fabrication, special smaller tools used mai ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetramethyltin
Tetramethyltin is an organometallic compound with the formula (CH3)4Sn. This liquid, one of the simplest organotin compounds, is useful for transition-metal mediated conversion of acid chlorides to methyl ketones and aryl halides to aryl methyl ketones. It is volatile and toxic, so care should be taken when using it in the laboratory. Synthesis and structure Tetramethyltin is synthesized by reaction of the Grignard reagent methylmagnesium iodide, with tin tetrachloride, which is synthesized by reacting tin metal with chlorine gas. :4 CH3MgI + SnCl4 → (CH3)4Sn + 4 MgICl In tetramethyltin, the metal surrounded by four methyl groups in a tetrahedral structure is a heavy analogue of neopentane. Applications Precursor to methyltin compounds Tetramethyltin is a precursor to trimethyltin chloride (and related methyltin halides), which are precursors to other organotin compounds. These methyltin chlorides are prepared via the so-called Kocheshkov redistribution reaction. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organotin Compounds
Organotin chemistry is the scientific study of the synthesis and properties of organotin compounds or stannanes, which are organometallic compounds containing tin–carbon bonds. The first organotin compound was diethyltin diiodide (), discovered by Edward Frankland in 1849. The area grew rapidly in the 1900s, especially after the discovery of the Grignard reagents, which are useful for producing Sn–C bonds. The area remains rich with many applications in industry and continuing activity in the research laboratory. Structure Organotin compounds are generally classified according to their oxidation states. Tin(IV) compounds are much more common and more useful. Organic derivatives of tin(IV) The tetraorgano derivatives are invariably tetrahedral. Compounds of the type SnRR'R''R have been resolved into individual enantiomers. Organotin halides Organotin chlorides have the formula for values of ''n'' up to 3. Bromides, iodides, and fluorides are also known, but are less imp ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tin(IV) Compounds
Tin is a chemical element; it has chemical symbol, symbol Sn () and atomic number 50. A silvery-colored metal, tin is soft enough to be cut with little force, and a bar of tin can be bent by hand with little effort. When bent, a bar of tin makes a sound, the so-called "tin cry", as a result of crystal twinning, twinning in tin crystals. Tin is a post-transition metal in Carbon group, group 14 of the periodic table of elements. It is obtained chiefly from the mineral cassiterite, which contains tin(IV) oxide, stannic oxide, . Tin shows a chemical similarity to both of its neighbors in group 14, germanium and lead, and has two main oxidation states, +2 and the slightly more stable +4. Tin is the 49th most abundance of the chemical elements, abundant element on Earth, making up 0.00022% of its crust, and with 10 stable isotopes, it has the largest number of stable isotopes in the periodic table, due to its magic number (physics), magic number of protons. It has two main allotrop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |