|
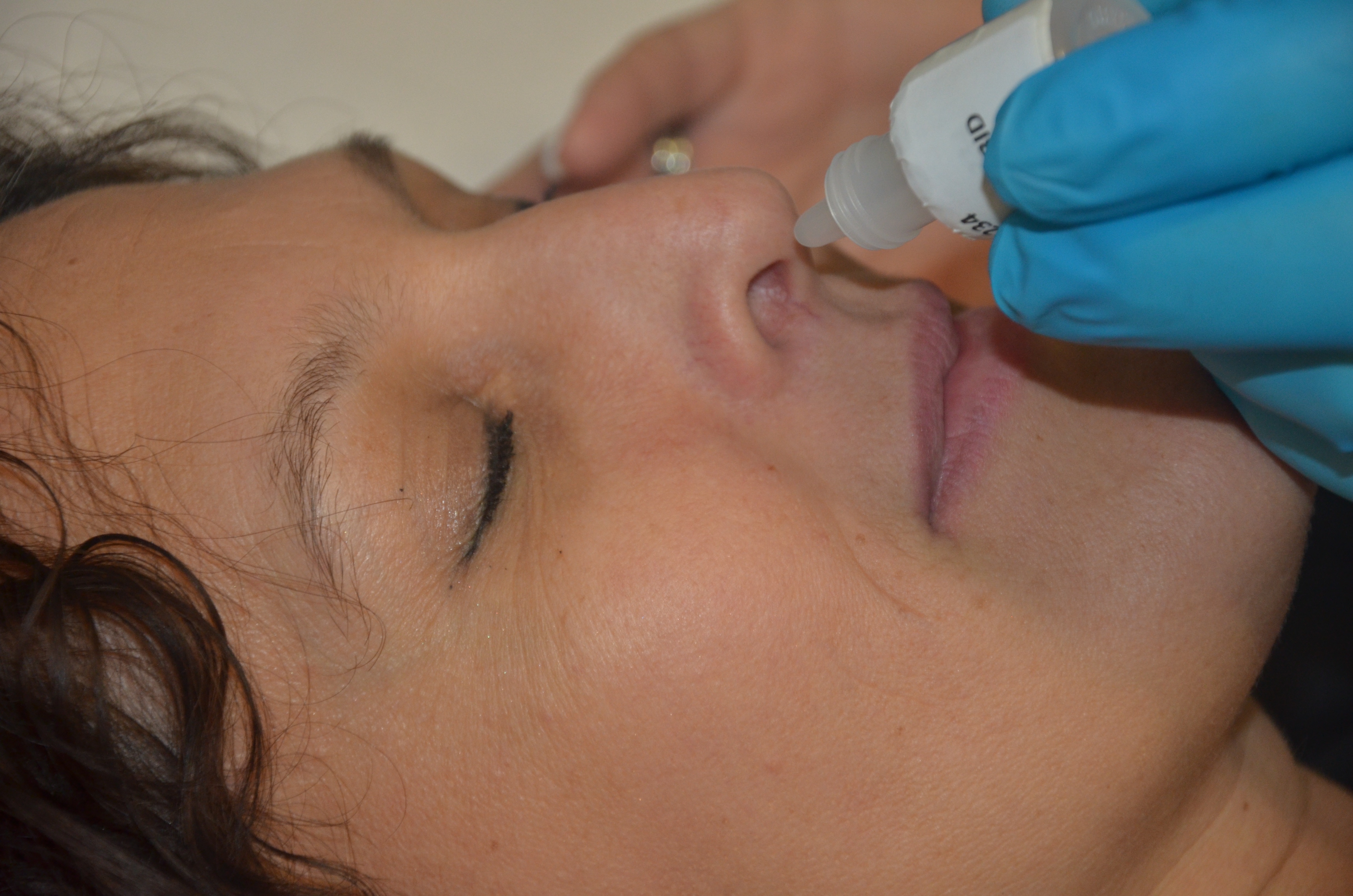
Beremagene Geperpavec
Beremagene geperpavec, sold under the brand name Vyjuvek, is a gene therapy used for the treatment of wounds. Beremagene geperpavec is the first approved gene therapy to use herpes-simplex virus type 1 as a vector. Beremagene geperpavec is a genetically modified (engineered in a laboratory) herpes-simplex virus used to deliver normal copies of the COL7A1 gene to the wounds. The most common adverse reactions include itching, chills, redness, rash, cough and runny nose. Beremagene geperpavec was approved for medical use in the United States in May 2023, and in the European Union in April 2025. Medical uses Beremagene geperpavec is indicated for the treatment of wounds in people with dystrophic epidermolysis bullosa with mutation(s) in the collagen type VII alpha 1 chain (COL7A1) gene. History The safety and effectiveness of beremagene geperpavec was established primarily in a randomized, double-blinded, placebo-controlled study involving a total of 31 subjects with dy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Topical Application
A topical medication is a medication that is applied to a particular place on or in the body. Most often topical medication means application to body surfaces such as the skin or mucous membranes to treat ailments via a large range of classes including creams, foams, gels, lotions, and ointments. Many topical medications are epicutaneous, meaning that they are applied directly to the skin. Topical medications may also be inhalational, such as asthma medications, or applied to the surface of tissues other than the skin, such as eye drops applied to the conjunctiva, or ear drops placed in the ear, or medications applied to the surface of a tooth. The word ''topical'' derives from Greek τοπικός ''topikos'', "of a place". Justification Topical drug delivery is a route of administering drugs via the skin to provide topical therapeutic effects. As skin is one of the largest and most superficial organs in the human body, pharmacists utilise it to deliver various drug ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gene Therapy
Gene therapy is Health technology, medical technology that aims to produce a therapeutic effect through the manipulation of gene expression or through altering the biological properties of living cells. The first attempt at modifying human DNA was performed in 1980, by Martin Cline, but the first successful nuclear gene transfer in humans, approved by the National Institutes of Health, was performed in May 1989. The first therapeutic use of gene transfer as well as the first direct insertion of human DNA into the nuclear genome was performed by French Anderson in a trial starting in September 1990. Between 1989 and December 2018, over 2,900 clinical trials were conducted, with more than half of them in Phases of clinical research, phase I. In 2003, Gendicine became the first gene therapy to receive regulatory approval. Since that time, further gene therapy drugs were approved, such as alipogene tiparvovec (2012), Strimvelis (2016), tisagenlecleucel (2017), voretigene neparvovec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Food And Drug Administration
The United States Food and Drug Administration (FDA or US FDA) is a List of United States federal agencies, federal agency of the United States Department of Health and Human Services, Department of Health and Human Services. The FDA is responsible for protecting and promoting public health through the control and supervision of food safety, tobacco products, caffeine products, dietary supplements, Prescription drug, prescription and Over-the-counter drug, over-the-counter pharmaceutical drugs (medications), vaccines, biopharmaceuticals, blood transfusions, medical devices, electromagnetic radiation emitting devices (ERED), cosmetics, Animal feed, animal foods & feed and Veterinary medicine, veterinary products. The FDA's primary focus is enforcement of the Federal Food, Drug, and Cosmetic Act (FD&C). However, the agency also enforces other laws, notably Section 361 of the Public Health Service Act as well as associated regulations. Much of this regulatory-enforcement work is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Herpes Simplex Virus 1
Herpes simplex virus 1 (HHV-1 or HSV-1) is a species of virus in the genus ''Simplexvirus'', subfamily ''Alphaherpesvirinae'', family ''Herpesviridae'', and order ''Herpesvirales''. Evolution Herpes simplex virus 1 can be classified into six clades. Four of these occur in East Africa, one in East Asia and one in Europe and North America. This suggests that the virus may have originated in East Africa. The most recent common ancestor of the Eurasian strains appears to have evolved around 60,000 years ago. Pathology Herpes simplex virus 1 infects humans, most often as cold sores. It is very common and contagious; about 67% of the world population under the age of 50 has Herpes simplex virus 1. It is often acquired orally during childhood. It may also be sexually transmitted, including contact with saliva, such as kissing and mouth-to-genital contact (oral sex). The virus tends to reside in the trigeminal ganglia. This species appears to be particularly damaging to the nervous ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nature Biotechnology
''Nature Biotechnology'' is a monthly peer-reviewed scientific journal published by Nature Portfolio. The editor-in-chief is Barbara Cheifet who heads an in-house team of editors. The focus of the journal is biotechnology including research results and the commercial business sector of this field. Coverage includes the related biological, biomedical, agricultural and environmental sciences. Also of interest are the commercial, political, legal, and societal influences that affect this field. The journal continues serial publication of the title "Bio/Technology", which had a publication period of 1983 to 1996. Abstracting and indexing This journal is indexed in Accessed 2012-06-29 * BIOBASE * BIOSIS * Chemical Abstracts Service * CSA Illumina * CAB Abstracts * EMBASE * Scopus * Current Contents * Science Citation Index * Medline (PubMed) A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
COL7A1
Collagen alpha-1(VII) chain is a protein that in humans is encoded by the ''COL7A1'' gene. It is composed of a triple helical, collagenous domain flanked by two non-collagenous domains, and functions as an anchoring fibril between the dermal-epidermal junction in the basement membrane. Mutations in COL7A1 cause all types of dystrophic epidermolysis bullosa, and the exact mutations vary based on the specific type or subtype. It has been shown that interactions between the NC-1 domain of collagen VII and several other proteins, including laminin-5 and collagen IV, contribute greatly to the overall stability of the basement membrane. Structure Type VII collagen is composed of three main domains in the following order: a non-collagenous domain, abbreviated NC-1; a collagenous domain; and a second non-collagenous domain, NC-2. The NC-1 domain has a cartilage matrix protein (CMP), nine fibronectin III (FNIII)-like subdomains, and a von Willebrand Factor A-like subdomain (VWFA1); a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Indicated
In medicine, an indication is a valid reason to use a certain test, medication, procedure, or surgery. There can be multiple indications to use a procedure or medication. An indication can commonly be confused with the term diagnosis. A diagnosis is the assessment that a particular medical condition is present while an indication is a reason for use. The opposite of an indication is a contraindication, a reason to withhold a certain medical treatment because the risks of treatment clearly outweigh the benefits. In the United States, indications for prescription drugs are approved by the FDA. Indications are included in the Indications and Usage section of the Prescribing Information. The primary role of this section of labeling is to enable health care practitioners to readily identify appropriate therapies for patients by clearly communicating the drug's approved indication(s). The Indications and Usage section states the disease or condition, or manifestation or symptoms thereof ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dystrophic Epidermolysis Bullosa
Dystrophic lakes, also known as humic lakes, are lakes that contain high amounts of humic substances and organic acids. The presence of these substances causes the water to be brown in colour and have a generally low pH of around 4.0-6.0. The presence of humic substances are mainly due to certain plants in the watersheds of the lakes, such as peat mosses and conifers. Due to these acidic conditions, few taxa are able to survive, consisting mostly of aquatic plants, algae, phytoplankton, picoplankton, and bacteria.Drzymulska, D., Fiłoc, M., Kupryjanowicz, M., Szeroczyńska, K., & Zieliński, P. 2015. Postglacial shifts in lake trophic status based on a multiproxy study of a humic lake. Holocene, 25(3), 495-507.Jasser, I. 1997. The dynamics and importance of picoplankton in shallow, dystrophic lake in comparison with surface waters of two deep lakes with contrasting trophic status. Hydrobiologia, 342/343(1), 87-93. Dystrophic lakes can be found in many areas of the world, especial ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Orphan Drug
An orphan drug is a medication, pharmaceutical agent that is developed to treat certain rare medical conditions. An orphan drug would not be profitable to produce without government assistance, due to the small population of patients affected by the conditions. The conditions that orphan drugs are used to treat are referred to as orphan diseases. The assignment of orphan status to a disease and to drugs developed to treat it is a matter of public policy that depends on the legislation (if there is any) of the country. Designation of a drug as an orphan drug has yielded medical breakthroughs that might not otherwise have been achieved, due to the economics of drug medical research, research and development. Examples of this can be that in the U.S. and the EU, it is easier to gain marketing approval for an orphan drug. There may be other financial incentives, such as an extended period of exclusivity, during which the producer has sole rights to market the drug. All are intended to en ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
FDA Fast Track
Fast track is a designation by the United States Food and Drug Administration (FDA) of an investigational drug for expedited review to facilitate development of drugs that treat a serious or life-threatening condition and fill an unmet medical need. Fast track designation must be requested by the drug company. The request can be initiated at any time during the drug development process. FDA will review the request and attempt to make a decision within sixty days. Purpose Fast track is one of five FDA approaches to make new drugs available as rapidly as possible: the others are priority review, breakthrough therapy, accelerated approval and regenerative medicine advanced therapy. Fast track was introduced by the FDA Modernization Act of 1997. Requirements Fast track designation is designed to aid in the development and expedite the review of drugs which show promise in treating a serious or life-threatening disease and address an unmet medical need. Serious condition: determ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Priority Review
Priority review is a program of the United States Food and Drug Administration (FDA) to expedite the review process for drugs that are expected to have a particularly great impact on the treatment of a disease. The priority review voucher program is a program that grants a voucher for priority review to a drug developer as an incentive to develop treatments for disease indications with limited profitability. Priority review vouchers are currently earned by pharmaceutical companies for the development and approval of drugs treating neglected tropical diseases, rare pediatric diseases, and "medical countermeasures" for terrorism. The voucher can be used for future drugs that could have wider indications for use, but the company is required to pay a fee (approximately $2.8 million) to use the voucher. When seeking approval for a drug, manufacturers can apply to the FDA for priority review. This is granted when a drug is intended to treat a serious condition and would "provide a sig ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Committee For Medicinal Products For Human Use
The Committee for Medicinal Products for Human Use (CHMP), formerly known as the Committee for Proprietary Medicinal Products (CPMP), is the European Medicines Agency's committee responsible for elaborating the agency's opinions on all issues regarding medicinal product Medication (also called medicament, medicine, pharmaceutical drug, medicinal product, medicinal drug or simply drug) is a drug used to diagnose, cure, treat, or prevent disease. Drug therapy ( pharmacotherapy) is an important part of the ...s for human use. See also * Committee for Medicinal Products for Veterinary Use References External links Committee for Medicinal Products for Human Use (CHMP) Health and the European Union {{eu-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |