|
Allyl Chloride
Allyl chloride is the organic compound with the formula C H2=CHCH2 Cl. This colorless liquid is insoluble in water but soluble in common organic solvents. It is mainly converted to epichlorohydrin, used in the production of plastics. It is a chlorinated derivative of propylene. It is an alkylating agent, which makes it both useful and hazardous to handle. Production Laboratory scale Allyl chloride was first produced in 1857 by Auguste Cahours and August Hofmann by reacting allyl alcohol with phosphorus trichloride.Ludger Krähling, Jürgen Krey, Gerald Jakobson, Johann Grolig, Leopold Miksche “Allyl Compounds” in Ullmann's Encyclopedia of Industrial Chemistry Wiley-VCH, Weinheim, 2005. Published online: 15 June 2000. Modern preparation protocols economize this approach, replacing relatively expensive phosphorus trichloride with hydrochloric acid and a catalyst such as copper(I) chloride. Industrial scale Allyl chloride is produced by the chlorination of propylene. At low ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diethyl Ether
Diethyl ether, or simply ether, is an organic compound in the ether class with the formula , sometimes abbreviated as (see Pseudoelement symbols). It is a colourless, highly volatile, sweet-smelling ("ethereal odour"), extremely flammable liquid. It is commonly used as a solvent in laboratories and as a starting fluid for some engines. It was formerly used as a general anesthetic, until non-flammable drugs were developed, such as halothane. It has been used as a recreational drug to cause intoxication. Production Most diethyl ether is produced as a byproduct of the vapor-phase hydration of ethylene to make ethanol. This process uses solid-supported phosphoric acid catalysts and can be adjusted to make more ether if the need arises. Vapor-phase dehydration of ethanol over some alumina catalysts can give diethyl ether yields of up to 95%. Diethyl ether can be prepared both in laboratories and on an industrial scale by the acid ether synthesis. Ethanol is mixed with a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allyl Alcohol
Allyl alcohol ( IUPAC name: prop-2-en-1-ol) is an organic compound with the structural formula . Like many alcohols, it is a water-soluble, colourless liquid. It is more toxic than typical small alcohols. Allyl alcohol is used as a raw material for the production of glycerol, but is also used as a precursor to many specialized compounds such as flame-resistant materials, drying oils, and plasticizers. Allyl alcohol is the smallest representative of the allylic alcohols. Production Allyl alcohol can be obtained by many methods. It was first prepared in 1856 by Auguste Cahours and August Hofmann by hydrolysis of allyl iodide. Today allyl alcohol is produced commercially by the Olin and Shell corporations through the hydrolysis of allyl chloride: :CH2=CHCH2Cl + NaOH -> CH2=CHCH2OH + NaCl Allyl alcohol can also be made by the rearrangement of propylene oxide, a reaction that is catalyzed by potassium alum at high temperature. The advantage of this method relative to the all ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyanation
In organic synthesis, cyanation is the attachment or substitution of a cyanide group on various substrates. Such transformations are high-value because they generate C-C bond. Furthermore nitriles are versatile functional groups. Cyanation to form sp3 nitriles Typically, alkyl nitriles are formed ''via'' SN1 or SN2-type cyanation with alkyl electrophiles. Illustrative is the synthesis of benzyl cyanide by the reaction of benzyl chloride and sodium cyanide. In some cases cuprous cyanide is used instead of sodium cyanide. Cyanation of ketones or aldehydes yields the corresponding cyanohydrins, which can be done directly with the cyanide ion (the cyanohydrin reaction) or by using bisulfite, followed by displacement of sulfite: A related reaction is hydrocyanation, which installs the elements of H-CN. Cyanation of arenes Cyanation of arenes offers access to benzoic acid derivatives, as well as the utility of aryl nitriles themselves in as fine chemicals: A variety of mechanisti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pesticide
Pesticides are substances that are meant to control pests. This includes herbicide, insecticide, nematicide, molluscicide, piscicide, avicide, rodenticide, bactericide, insect repellent, animal repellent, microbicide, fungicide, and lampricide. The most common of these are herbicides which account for approximately 80% of all pesticide use. Most pesticides are intended to serve as plant protection products (also known as crop protection products), which in general, protect plants from weeds, fungi, or insects. As an example, the fungus '' Alternaria solani'' is used to combat the aquatic weed '' Salvinia''. In general, a pesticide is a chemical (such as carbamate) or biological agent (such as a virus, bacterium, or fungus) that deters, incapacitates, kills, or otherwise discourages pests. Target pests can include insects, plant pathogens, weeds, molluscs, birds, mammals, fish, nematodes (roundworms), and microbes that destroy property, cause nuisance, or spread ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
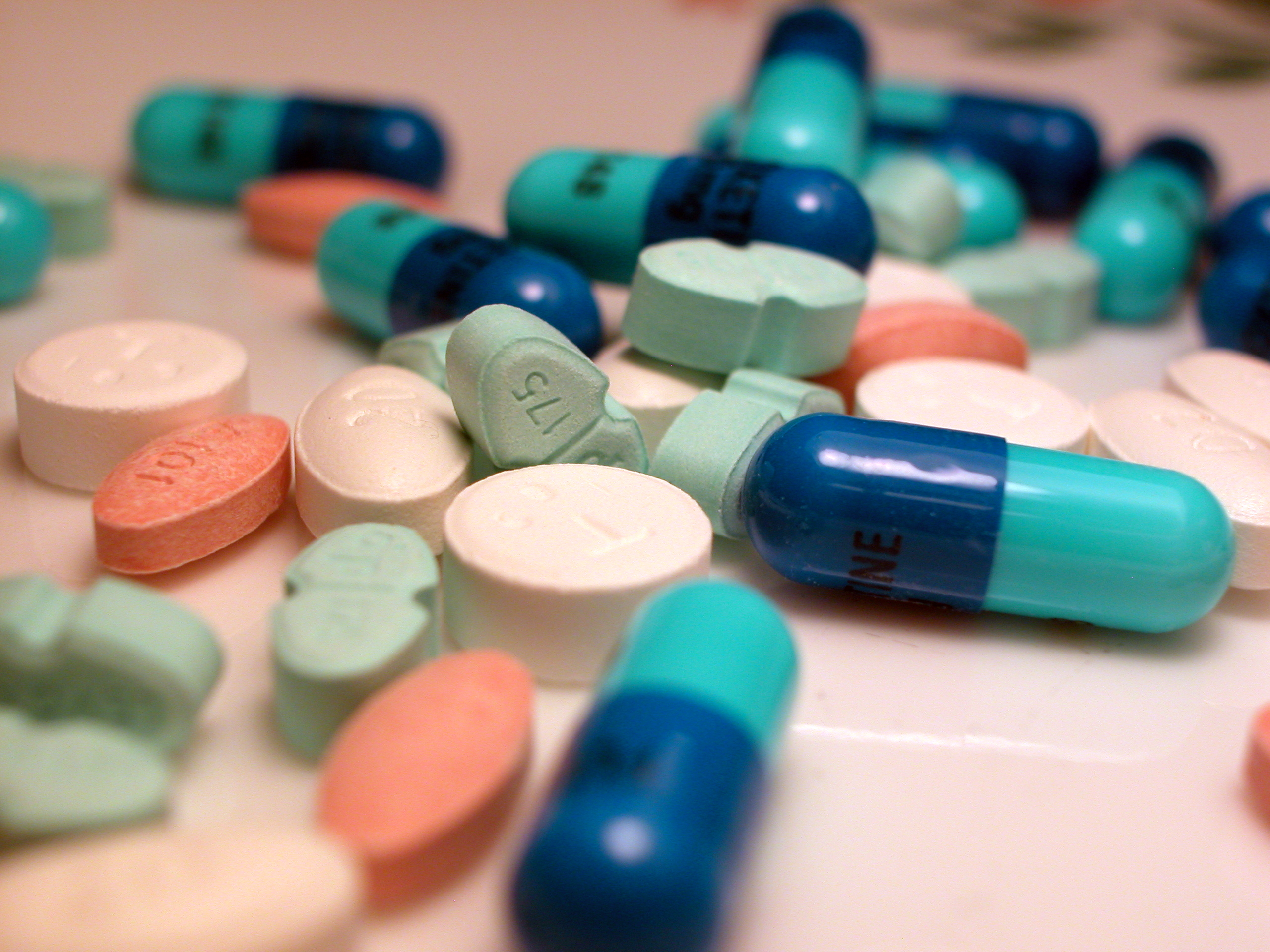
Pharmaceutical
A medication (also called medicament, medicine, pharmaceutical drug, medicinal drug or simply drug) is a drug used to diagnose, cure, treat, or prevent disease. Drug therapy ( pharmacotherapy) is an important part of the medical field and relies on the science of pharmacology for continual advancement and on pharmacy for appropriate management. Drugs are classified in multiple ways. One of the key divisions is by level of control, which distinguishes prescription drugs (those that a pharmacist dispenses only on the order of a physician, physician assistant, or qualified nurse) from over-the-counter drugs (those that consumers can order for themselves). Another key distinction is between traditional small molecule drugs, usually derived from chemical synthesis, and biopharmaceuticals, which include recombinant proteins, vaccines, blood products used therapeutically (such as IVIG), gene therapy, monoclonal antibodies and cell therapy (for instance, stem ce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1-Bromo-3-chloropropane
1-Bromo-3-chloropropane is an organohalogen compound with the formula Br(CH2)3Cl. It is a colorless liquid, produced by free-radical addition of hydrogen bromide to allyl chloride. It is used as an alkylating agent to install the –(CH2)3Cl and –(CH2)3– groups. For example, it is a precursor to 4-chlorobutyronitrile 4-Chlorobutyronitrile is the organic compound with the formula ClCH2CH2CH2CN. With both chloro and cyano functional groups, it is a bifunctional molecule. This colorless liquid is prepared by the reaction of sodium cyanide with 1-bromo-3-chloro .... References {{DEFAULTSORT:Bromo-3-chloropropane, 1- Organochlorides Organobromides ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allyl Isothiocyanate
Allyl isothiocyanate (AITC) is an organosulfur compound (formula CH2CHCH2NCS). This colorless oil is responsible for the pungent taste of mustard, radish, horseradish, and wasabi. This pungency and the lachrymatory effect of AITC are mediated through the TRPA1 and TRPV1 ion channels. It is slightly soluble in water, but more soluble in most organic solvents. Biosynthesis and biological functions Allyl isothiocyanate can be obtained from the seeds of black mustard (''Brassica nigra'') or brown Indian mustard (''Brassica juncea''). When these mustard seeds are broken, the enzyme myrosinase is released and acts on a glucosinolate known as sinigrin to give allyl isothiocyanate. Allyl isothiocyanate serves the plant as a defense against herbivores; since it is harmful to the plant itself , it is stored in the harmless form of the glucosinolate, separate from the myrosinase enzyme. When an animal chews the plant, the allyl isothiocyanate is released, repelling the animal. Human appr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allylamine
Allylamine is an organic compound with the formula C3H5NH2. This colorless liquid is the simplest stable unsaturated amine. Production and reactions All three allylamines, mono-, di-, and triallylamine, are produced by the treating allyl chloride with ammonia followed by distillation. Or by the reaction of allyl chloride with Hexamine. Pure samples can be prepared by hydrolysis of allyl isothiocyanate. It behaves as a typical amine.Henk de Koning, W. Nico Speckamp "Allylamine" in ''Encyclopedia of Reagents for Organic Synthesis'', 2001, John Wiley & Sons, Weinheim. Article Online Posting Date: April 15, 2001 Polymerization can be used to prepare the homopolymer ( polyallylamine) or copolymers. The polymers are promising membranes for use in reverse osmosis. Other allylamines Diallylamine is a precursor to industrial products. Functionalized allylamines have pharmaceutical applications. Pharmaceutically important allylamines include flunarizine and naftifine. Flunarizine a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Chloride
The compound hydrogen chloride has the chemical formula and as such is a hydrogen halide. At room temperature, it is a colourless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric water vapor. Hydrogen chloride gas and hydrochloric acid are important in technology and industry. Hydrochloric acid, the aqueous solution of hydrogen chloride, is also commonly given the formula HCl. Reactions Hydrogen chloride is a diatomic molecule, consisting of a hydrogen atom H and a chlorine atom Cl connected by a polar covalent bond. The chlorine atom is much more electronegative than the hydrogen atom, which makes this bond polar. Consequently, the molecule has a large dipole moment with a negative partial charge (δ−) at the chlorine atom and a positive partial charge (δ+) at the hydrogen atom. In part because of its high polarity, HCl is very soluble in water (and in other polar solvents). Upon contact, and HCl combine to form hydronium catio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Free Radical
A daughter category of ''Ageing'', this category deals only with the biological aspects of ageing. Ageing Ailments of unknown cause Biogerontology Biological processes Causes of death Cellular processes Gerontology Life extension Metabolic disorders Metabolism Old age Time in life Wikipedia categories named after diseases and disorders {{CatAutoTOC ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,2-dichloropropane
1,2-Dichloropropane is an organic compound classified as a chlorocarbon. It is a colorless, flammable liquid with a sweet odor. it is obtained as a byproduct of the production of epichlorohydrin, which is produced on a large scale.Manfred Rossberg, Wilhelm Lendle, Gerhard Pfleiderer, Adolf Tögel, Eberhard-Ludwig Dreher, Ernst Langer, Heinz Rassaerts, Peter Kleinschmidt, Heinz Strack, Richard Cook, Uwe Beck, Karl-August Lipper, Theodore R. Torkelson, Eckhard Löser, Klaus K. Beutel, Trevor Mann "Chlorinated Hydrocarbons" in Ullmann's Encyclopedia of Industrial Chemistry 2006, Wiley-VCH, Weinheim. . Uses 1,2-Dichloropropane is an intermediate in the production of perchloroethylene and other chlorinated chemicals. It was once used as a soil fumigant, chemical intermediate, as well as an industrial solvent and was found in paint strippers, varnishes, and furniture finish removers but some of these uses have been discontinued. Carcinogenity Following several cases of bile duct cance ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halogenation
In chemistry, halogenation is a chemical reaction that entails the introduction of one or more halogens into a compound. Halide-containing compounds are pervasive, making this type of transformation important, e.g. in the production of polymers, drugs. This kind of conversion is in fact so common that a comprehensive overview is challenging. This article mainly deals with halogenation using elemental halogens (F2, Cl2, Br2, I2). Halides are also commonly introduced using salts of the halides and halogen acids. Many specialized reagents exist for and introducing halogens into diverse substrates, e.g. thionyl chloride. Organic chemistry Several pathways exist for the halogenation of organic compounds, including free radical halogenation, ketone halogenation, electrophilic halogenation, and halogen addition reaction. The nature of the substrate determines the pathway. The facility of halogenation is influenced by the halogen. Fluorine and chlorine are more electrophilic and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |