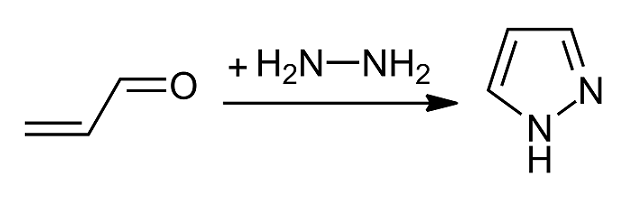|
Azine
Azines are a functional class of organic compounds with the connectivity . These compounds are the product of the condensation of hydrazine with ketones and aldehydes, although in practice they are often made by alternative routes. Ketazines are azines derived from ketones. For example, acetone azine is the simplest ketazine. Aldazines are azines derived from aldehydes. Preparation The usual method of industrial production is the peroxide process, starting from the ketone, ammonia, and hydrogen peroxide. : In the laboratory, azines are typically prepared by condensation of hydrazine with two equivalents of a carbonyl. Azines are also produced when chalcone reacts with a hydrazone to produce 3,5-diphenyl-1''H''-pyrazole, in a conversion also carried out with hydrazine hydrate. : Reactions Azines characteristically undergo hydrolysis to hydrazines. The reaction proceeds by the intermediacy of a hydrazone: : : Azines have been used as precursors to hydrazones: : They are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrazine Hydrate
Hydrazine is an inorganic compound with the chemical formula . It is a simple pnictogen hydride, and is a colourless flammable liquid with an ammonia-like odour. Hydrazine is highly hazardous unless handled in solution as, for example, hydrazine hydrate (). Hydrazine is mainly used as a foaming agent in preparing polymer foams, but applications also include its uses as a precursor to pharmaceuticals and agrochemicals, as well as a long-term storable propellant for in-space spacecraft propulsion. Additionally, hydrazine is used in various rocket fuels and to prepare the gas precursors used in airbags. Hydrazine is used within both nuclear and conventional electrical power plant steam cycles as an oxygen scavenger to control concentrations of dissolved oxygen in an effort to reduce corrosion. , approximately 120,000 tons of hydrazine hydrate (corresponding to a 64% solution of hydrazine in water by weight) were manufactured worldwide per year. Hydrazines are a class of organ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acetone Azine
Acetone azine is the simplest ketazine. It is an intermediate in some hydrazine manufacturing processes. Synthesis Acetone azine can be prepared from acetone and hydrazine: :2 (CH3)2CO + N2H4 → 2 H2O + CH3)2C=Nsub>2 It can also be produced from acetone (2 eq.), ammonia (2 eq.) and hydrogen peroxide (1 eq.). The first step is the formation of acetone imine, Me2C=NH; this is then oxidized by hydrogen peroxide through a complex mechanism to give 3,3-dimethyloxaziridine, which reacts with a further molecule of ammonia to produce acetone hydrazone. The hydrazone then condenses with a further molecule of acetone to produce the azine. The acetone azine product is distilled out of the reaction mixture as a 1:6 azeotrope An azeotrope () or a constant heating point mixture is a mixture of two or more liquids whose proportions cannot be changed by simple distillation.Moore, Walter J. ''Physical Chemistry'', 3rd e Prentice-Hall 1962, pp. 140–142 This happens beca ... with wat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Peroxide Process
The peroxide process is a method for the industrial production of hydrazine. In this process hydrogen peroxide is used as an oxidant instead of sodium hypochlorite, which is traditionally used to generate hydrazine. The main advantage of the peroxide process to hydrazine relative to the traditional Olin Raschig process is that it does not coproduce salt. In this respect, the peroxide process is an example of green chemistry. Since many millions of kilograms of hydrazine are produced annually, this method is of both commercial and environmental significance.Jean-Pierre Schirmann, Paul Bourdauducq "Hydrazine" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2002. . Production Ketazine formation In the usual implementation, hydrogen peroxide is used together with acetamide. This mixture does not react with ammonia directly but does so in the presence of methyl ethyl ketone to give the oxaziridine. : Balanced equations for the individual steps are as follows ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pechiney-Ugine-Kuhlmann Process
The peroxide process is a method for the industrial production of hydrazine. In this process hydrogen peroxide is used as an oxidant instead of sodium hypochlorite, which is traditionally used to generate hydrazine. The main advantage of the peroxide process to hydrazine relative to the traditional Olin Raschig process is that it does not coproduce salt. In this respect, the peroxide process is an example of green chemistry. Since many millions of kilograms of hydrazine are produced annually, this method is of both commercial and environmental significance.Jean-Pierre Schirmann, Paul Bourdauducq "Hydrazine" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2002. . Production Ketazine formation In the usual implementation, hydrogen peroxide is used together with acetamide. This mixture does not react with ammonia directly but does so in the presence of methyl ethyl ketone to give the oxaziridine. : Balanced equations for the individual steps are as follows. I ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Herbicide
Herbicides (, ), also commonly known as weed killers, are substances used to control undesired plants, also known as weeds.EPA. February 201Pesticides Industry. Sales and Usage 2006 and 2007: Market Estimates. Summary in press releasMain page for EPA reports on pesticide use ihere Selective herbicides control specific weed species while leaving the desired crop relatively unharmed, while non-selective herbicides (sometimes called "total weed killers") kill plants indiscriminately. The combined effects of herbicides, nitrogen fertilizer, and improved cultivars has increased yields (per acre) of major crops by three to six times from 1900 to 2000. In the United States in 2012, about 91% of all herbicide usage, was determined by weight applied, in agriculture. In 2012, world pesticide expenditures totaled nearly US$24.7 billion; herbicides were about 44% of those sales and constituted the biggest portion, followed by insecticides, fungicides, and fumigants. Herbicide is also used ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrazone
Hydrazones are a class of organic compounds with the structure . They are related to ketones and aldehydes by the replacement of the oxygen =O with the = functional group. They are formed usually by the action of hydrazine on ketones or aldehydes. Synthesis Hydrazine, organohydrazines, and 1,1-diorganohydrazines react with aldehydes and ketones to give hydrazones. : Phenylhydrazine reacts with reducing sugars to form hydrazones known as osazones, which was developed by German chemist Emil Fischer as a test to differentiate monosaccharides. Uses image:Pigment Yellow 97.svg, left, Pigment Yellow 97, a popular yellow colorant, is a hydrazone., 160px Hydrazones are the basis for various analyses of ketones and aldehydes. For example, dinitrophenylhydrazine coated onto a silica sorbent is the basis of an adsorption cartridge. The hydrazones are then eluted and analyzed by high-performance liquid chromatography (HPLC) using a ultraviolet, UV detector. The compound carbonyl cyanide-p-t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Peroxide
Hydrogen peroxide is a chemical compound with the formula . In its pure form, it is a very pale blue liquid that is slightly more viscosity, viscous than Properties of water, water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution (3%–6% by weight) in water for consumer use and in higher concentrations for industrial use. Concentrated hydrogen peroxide, or "high-test peroxide", decomposes explosively when heated and has been used as both a monopropellant and an oxidizer in rocketry. Hydrogen peroxide is a reactive oxygen species and the simplest peroxide, a compound having an oxygen–oxygen single bond. It decomposes slowly into water and elemental oxygen when exposed to light, and rapidly in the presence of organic or reactive compounds. It is typically stored with a Stabilizer (chemistry), stabilizer in a weakly acidic solution in an opaque bottle. Hydrogen peroxide is found in biological systems including the human body. Enzymes that u ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldehydes
In organic chemistry, an aldehyde () (lat. ''al''cohol ''dehyd''rogenatum, dehydrogenated alcohol) is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl group. Aldehydes are a common motif in many chemicals important in technology and biology. Structure and bonding Aldehyde molecules have a central carbon atom that is connected by a double bond to oxygen, a single bond to hydrogen and another single bond to a third substituent, which is carbon or, in the case of formaldehyde, hydrogen. The central carbon is often described as being sp2- hybridized. The aldehyde group is somewhat polar. The bond length is about 120–122 picometers. Physical properties and characterization Aldehydes have properties that are diverse and that depend on the remainder of the molecule. Smaller aldehydes such as formaldehyde and acetaldehyde are soluble ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bayer
Bayer AG (English: , commonly pronounced ; ) is a German multinational pharmaceutical and biotechnology company and is one of the largest pharmaceutical companies and biomedical companies in the world. Headquartered in Leverkusen, Bayer's areas of business include: pharmaceuticals, consumer healthcare products, agricultural chemicals, seeds and biotechnology products. The company is a component of the EURO STOXX 50 stock market index. Bayer was founded in 1863 in Barmen as a partnership between dye salesman Friedrich Bayer (1825–1880) and dyer Friedrich Weskott (1821–1876). The company was established as a dyestuffs producer, but the versatility of aniline chemistry led Bayer to expand its business into other areas. In 1899, Bayer launched the compound acetylsalicylic acid under the trademarked name Aspirin. Aspirin is on the World Health Organization's List of Essential Medicines. In 2021, it was the 34th most commonly prescribed medication in the United State ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyrazole
Pyrazole is an organic compound with the chemical formula, formula . It is a heterocycle characterized as an azole with a 5-membered ring of three carbon atoms and two adjacent nitrogen atoms, which are in Arene substitution pattern, ortho-substitution. Pyrazoles are also a class of compounds that have the ring C3N2 with adjacent nitrogen atoms. Pyrazole itself has few applications but many substituted pyrazoles are of commercial interest. Notable drugs containing a pyrazole ring are celecoxib (celebrex) and the anabolic steroid stanozolol. Properties Pyrazole is a weak base, with p''K''b 11.5 (p''K''a of the conjugate acid 2.49 at 25 °C). According to X-ray crystallography, the compound is planar. The two C-N distances are similar, both near 1.33 Å History The term pyrazole was given to this class of compounds by German Chemist Ludwig Knorr in 1883. In a classical method developed by German chemist Hans von Pechmann in 1898, pyrazole was synthesized from acetylene and di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ketones
In organic chemistry, a ketone is an organic compound with the structure , where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond C=O). The simplest ketone is acetone (where R and R' are methyl), with the formula . Many ketones are of great importance in biology and industry. Examples include many sugars (ketoses), many steroids, ''e.g.'', testosterone, and the solvent acetone. Nomenclature and etymology The word ''ketone'' is derived from ''Aketon'', an old German word for ''acetone''. According to the rules of IUPAC nomenclature, ketone names are derived by changing the suffix ''-ane'' of the parent alkane to ''-anone''. Typically, the position of the carbonyl group is denoted by a number, but traditional nonsystematic names are still generally used for the most important ketones, for example acetone and benzophenone. These nonsystematic names are considered retained IUPAC names, although some introd ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |