|
Fluorescence Intensity Decay Shape Microscopy
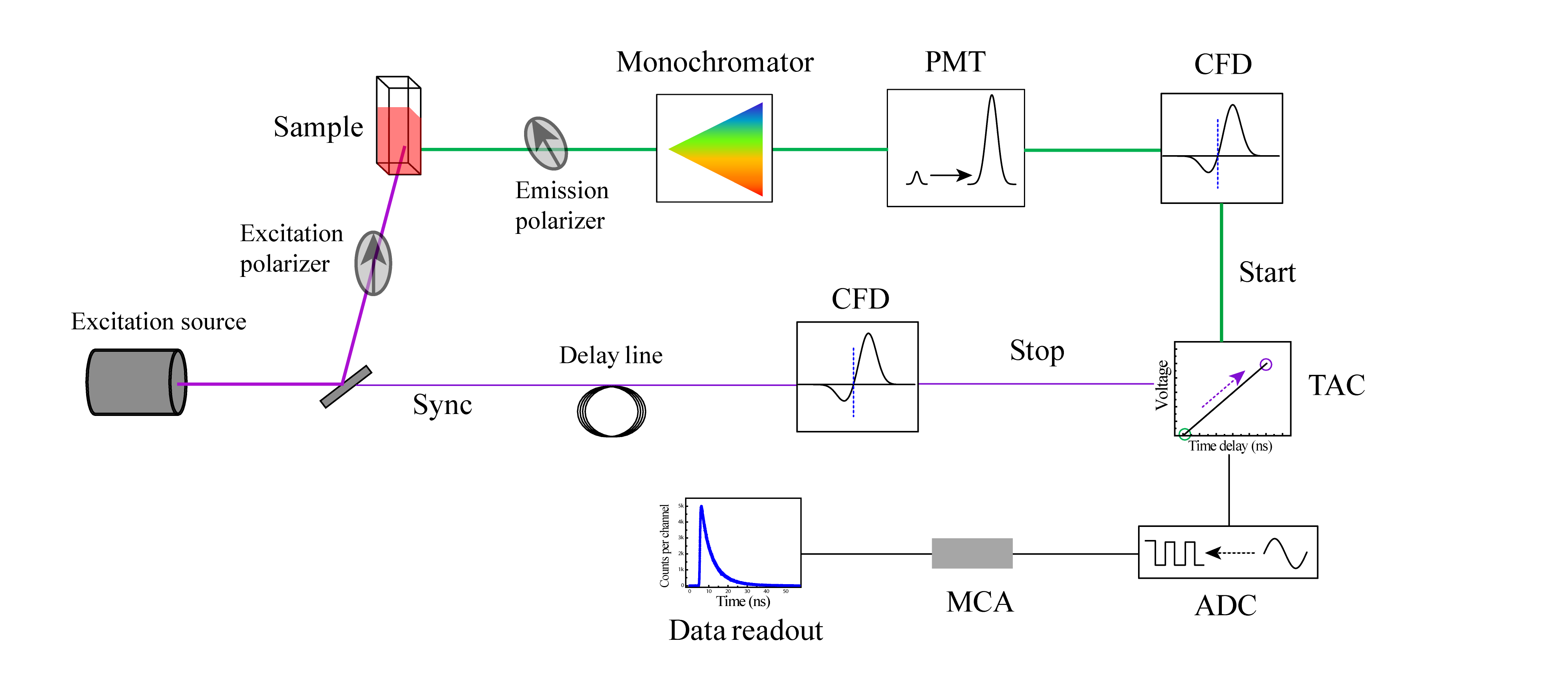
Within the scientific study of a molecule's color, Fluorescence intensity decay shape microscopy (FIDSAM) is a fluorescence microscopy technique, which utilizes the time evolution of fluorescence emission after a pulsed excitation to analyse the decay statistics of an excited chromophore. The main application of FIDSAM is the discrimination of unspecific autofluorescent background signal from the target signal of a dedicated chromophore. Principle The FIDSAM method analyses the number of different molecules contributing to a measured fluorescence signal. Assuming a pure fluorescent dye solution in an isotropic surrounding, the individual emitters are indistinguishable. Accordingly, they obey the same fluorescence emission statistics and the time evolution of the fluorescence emission after a pulsed excitation can be described by a monoexponential decay function according to: :F(t) = A e^ with A = the initial fluorescence intensity after the excitation and \tau = the decay con ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecule
A molecule is a group of two or more atoms held together by attractive forces known as chemical bonds; depending on context, the term may or may not include ions which satisfy this criterion. In quantum physics, organic chemistry, and biochemistry, the distinction from ions is dropped and ''molecule'' is often used when referring to polyatomic ions. A molecule may be homonuclear, that is, it consists of atoms of one chemical element, e.g. two atoms in the oxygen molecule (O2); or it may be heteronuclear, a chemical compound composed of more than one element, e.g. water (molecule), water (two hydrogen atoms and one oxygen atom; H2O). In the kinetic theory of gases, the term ''molecule'' is often used for any gaseous particle regardless of its composition. This relaxes the requirement that a molecule contains two or more atoms, since the noble gases are individual atoms. Atoms and complexes connected by non-covalent interactions, such as hydrogen bonds or ionic bonds, are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Color
Color (American English) or colour (British English) is the visual perceptual property deriving from the spectrum of light interacting with the photoreceptor cells of the eyes. Color categories and physical specifications of color are associated with objects or materials based on their physical properties such as light absorption, reflection, or emission spectra. By defining a color space, colors can be identified numerically by their coordinates. Because perception of color stems from the varying spectral sensitivity of different types of cone cells in the retina to different parts of the spectrum, colors may be defined and quantified by the degree to which they stimulate these cells. These physical or physiological quantifications of color, however, do not fully explain the psychophysical perception of color appearance. Color science includes the perception of color by the eye and brain, the origin of color in materials, color theory in art, and the physics of ele ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorescence
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation. It is a form of luminescence. In most cases, the emitted light has a longer wavelength, and therefore a lower photon energy, than the absorbed radiation. A perceptible example of fluorescence occurs when the absorbed radiation is in the ultraviolet region of the electromagnetic spectrum (invisible to the human eye), while the emitted light is in the visible region; this gives the fluorescent substance a distinct color that can only be seen when the substance has been exposed to UV light. Fluorescent materials cease to glow nearly immediately when the radiation source stops, unlike phosphorescent materials, which continue to emit light for some time after. Fluorescence has many practical applications, including mineralogy, gemology, medicine, chemical sensors ( fluorescence spectroscopy), fluorescent labelling, dyes, biological detectors, cosmic-ray detec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Microscopy
Microscopy is the technical field of using microscopes to view objects and areas of objects that cannot be seen with the naked eye (objects that are not within the resolution range of the normal eye). There are three well-known branches of microscopy: optical, electron, and scanning probe microscopy, along with the emerging field of X-ray microscopy. Optical microscopy and electron microscopy involve the diffraction, reflection, or refraction of electromagnetic radiation/electron beams interacting with the specimen, and the collection of the scattered radiation or another signal in order to create an image. This process may be carried out by wide-field irradiation of the sample (for example standard light microscopy and transmission electron microscopy) or by scanning a fine beam over the sample (for example confocal laser scanning microscopy and scanning electron microscopy). Scanning probe microscopy involves the interaction of a scanning probe with the surface of the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromophore
A chromophore is the part of a molecule responsible for its color. The color that is seen by our eyes is the one not absorbed by the reflecting object within a certain wavelength spectrum of visible light. The chromophore is a region in the molecule where the energy difference between two separate molecular orbitals falls within the range of the visible spectrum. Visible light that hits the chromophore can thus be absorbed by exciting an electron from its ground state into an excited state. In biological molecules that serve to capture or detect light energy, the chromophore is the moiety that causes a conformational change in the molecule when hit by light. Conjugated pi-bond system chromophores Just like how two adjacent p-orbitals in a molecule will form a pi-bond, three or more adjacent p-orbitals in a molecule can form a conjugated pi-system. In a conjugated pi-system, electrons are able to capture certain photons as the electrons resonate along a certain distanc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
TCSPC
Ultrafast laser spectroscopy is a spectroscopic technique that uses ultrashort pulse lasers for the study of dynamics on extremely short time scales (attoseconds to nanoseconds). Different methods are used to examine the dynamics of charge carriers, atoms, and molecules. Many different procedures have been developed spanning different time scales and photon energy ranges; some common methods are listed below. Attosecond-to-picosecond spectroscopy Dynamics on the as to fs time scale are in general too fast to be measured electronically. Most measurements are done by employing a sequence of ultrashort light pulses to initiate a process and record its dynamics. The temporal width (duration) of the light pulses has to be on the same scale as the dynamics that are to be measured or even shorter. Light sources Titanium-sapphire laser Ti-sapphire lasers are tunable lasers that emit red and near-infrared light (700 nm- 1100 nm).Ti-sapphire laser oscillators use Ti doped-sapphir ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorescence Lifetime Imaging Microscopy
Fluorescence-lifetime imaging microscopy or FLIM is an imaging technique based on the differences in the exponential decay rate of the photon emission of a fluorophore from a sample. It can be used as an imaging technique in confocal microscopy, two-photon excitation microscopy, and multiphoton tomography. The fluorescence lifetime (FLT) of the fluorophore, rather than its intensity, is used to create the image in FLIM. Fluorescence lifetime depends on the local micro-environment of the fluorophore, thus precluding any erroneous measurements in fluorescence intensity due to change in brightness of the light source, background light intensity or limited photo-bleaching. This technique also has the advantage of minimizing the effect of photon scattering in thick layers of sample. Being dependent on the micro-environment, lifetime measurements have been used as an indicator for pH, viscosity and chemical species concentration. Fluorescence lifetimes A fluorophore which is excited b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Decay Constant
A quantity is subject to exponential decay if it decreases at a rate proportional to its current value. Symbolically, this process can be expressed by the following differential equation, where is the quantity and ( lambda) is a positive rate called the exponential decay constant, disintegration constant, rate constant, or transformation constant: :\frac = -\lambda N. The solution to this equation (see derivation below) is: :N(t) = N_0 e^, where is the quantity at time , is the initial quantity, that is, the quantity at time . Measuring rates of decay Mean lifetime If the decaying quantity, ''N''(''t''), is the number of discrete elements in a certain set, it is possible to compute the average length of time that an element remains in the set. This is called the mean lifetime (or simply the lifetime), where the exponential time constant, \tau, relates to the decay rate constant, λ, in the following way: :\tau = \frac. The mean lifetime can be looked at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorescence Techniques
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation. It is a form of luminescence. In most cases, the emitted light has a longer wavelength, and therefore a lower photon energy, than the absorbed radiation. A perceptible example of fluorescence occurs when the absorbed radiation is in the ultraviolet region of the electromagnetic spectrum (invisible to the human eye), while the emitted light is in the visible region; this gives the fluorescent substance a distinct color that can only be seen when the substance has been exposed to UV light. Fluorescent materials cease to glow nearly immediately when the radiation source stops, unlike phosphorescent materials, which continue to emit light for some time after. Fluorescence has many practical applications, including mineralogy, gemology, medicine, chemical sensors (fluorescence spectroscopy), fluorescent labelling, dyes, biological detectors, cosmic-ray detection, vacuu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |



.jpeg/1200px-Fall_Leaves_(199582361).jpeg)