|
Intravenous Fluids
Intravenous therapy (abbreviated as IV therapy) is a medical technique that administers fluids, medications and nutrients directly into a person's vein. The intravenous route of administration is commonly used for rehydration or to provide nutrients for those who cannot, or will not—due to reduced mental states or otherwise—consume food or water per os, by mouth. It may also be used to administer pharmaceutical drug, medications or other medical therapy such as blood transfusion, blood products or electrolytes to correct electrolyte imbalances. Attempts at providing intravenous therapy have been recorded as early as the 1400s, but the practice did not become widespread until the 1900s after the development of techniques for safe, effective use. The intravenous route is the fastest way to deliver medications and fluid replacement throughout the body as they are introduced directly into the circulatory system and thus quickly distributed. For this reason, the intravenous route ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cannula
A cannula (; Latin meaning 'little reed'; : cannulae or cannulas) is a tube that can be inserted into the body, often for the delivery or removal of fluid or for the gathering of samples. In simple terms, a cannula can surround the inner or outer surfaces of a trocar needle thus extending the effective needle length by at least half the length of the original needle. Its size mainly ranges from 14 to 26 Needle gauge comparison chart, gauge. Different-sized cannula have different colours as coded. Decannulation is the permanent removal of a cannula (intubation, extubation), especially of a tracheostomy cannula, once a physician determines it is no longer needed for breathing. Medicine Cannulas normally come with a trocar inside. The trocar is a needle, which punctures the body in order to get into the intended space. Intravenous cannulas are the most common in hospital use. A variety of cannulas are used to establish cardiopulmonary bypass in cardiac surgery. A nasal cannula ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Blood Substitutes
A blood substitute (also called artificial blood or blood surrogate) is a substance used to mimic and fulfill some functions of biological blood. It aims to provide an alternative to blood transfusion, which is transferring blood or blood-based products from one person into another. Thus far, there are no well-accepted ''oxygen-carrying'' blood substitutes, which is the typical objective of a red blood cell transfusion; however, there are widely available non-blood volume expanders for cases where only volume restoration is required. These are helping care providers avoid the risks of disease transmission and immune suppression, address the chronic blood donor shortage, and address the concerns of Jehovah's Witnesses and others who have religious objections to receiving transfused blood. The main categories of "oxygen-carrying" blood substitutes being pursued are hemoglobin-based oxygen carriers (HBOC) and perfluorocarbon emulsions. Oxygen therapeutics are in clinical trial ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Buffer Solution
A buffer solution is a solution where the pH does not change significantly on dilution or if an acid or base is added at constant temperature. Its pH changes very little when a small amount of strong acid or base is added to it. Buffer solutions are used as a means of keeping pH at a nearly constant value in a wide variety of chemical applications. In nature, there are many living systems that use buffering for pH regulation. For example, the bicarbonate buffering system is used to regulate the pH of blood, and bicarbonate also acts as a buffer in the ocean. Principles of buffering Buffer solutions resist pH change because of a chemical equilibrium between the weak acid HA and its conjugate base A−: When some strong acid is added to an equilibrium mixture of the weak acid and its conjugate base, hydrogen ions (H+) are added, and the equilibrium is shifted to the left, in accordance with Le Chatelier's principle. Because of this, the hydrogen ion concentration increas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
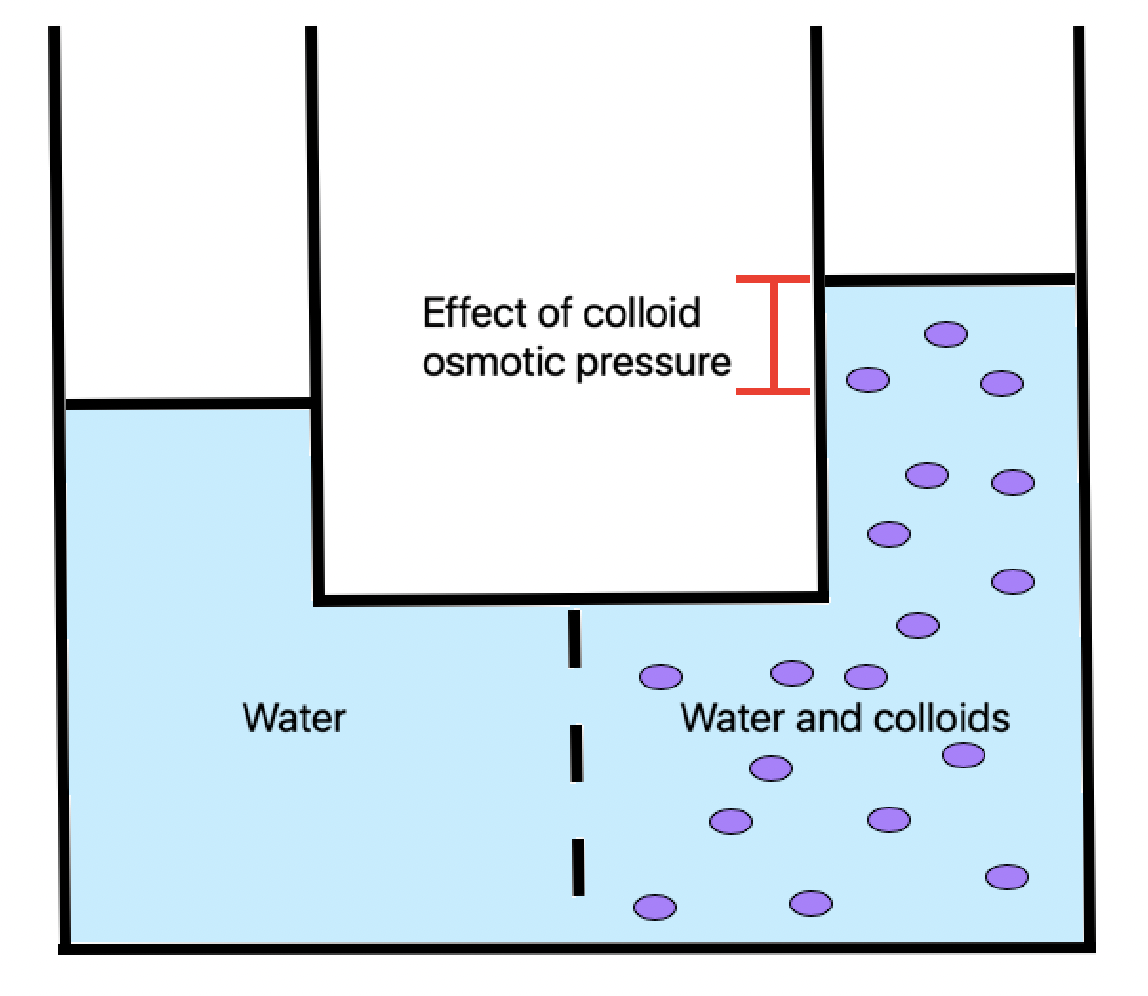
Colloid Osmotic Pressure
Oncotic pressure, or colloid osmotic-pressure, is a type of osmotic pressure induced by the plasma proteins, notably albumin, in a blood vessel's plasma (or any other body fluid such as blood and lymph) that causes a pull on fluid back into the capillary. It has an effect opposing both the hydrostatic blood pressure, which pushes water and small molecules out of the blood into the interstitial spaces at the arterial end of capillaries, and the interstitial colloidal osmotic pressure. These interacting factors determine the partitioning of extracellular water between the blood plasma and the extravascular space. Oncotic pressure strongly affects the physiological function of the circulatory system. It is suspected to have a major effect on the pressure across the glomerular filter. However, this concept has been strongly criticised and attention has shifted to the impact of the intravascular glycocalyx layer as the major player. Etymology The word 'oncotic' by definiti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Burn
A burn is an injury to skin, or other tissues, caused by heat, electricity, chemicals, friction, or ionizing radiation (such as sunburn, caused by ultraviolet radiation). Most burns are due to heat from hot fluids (called scalding), solids, or fire. Burns occur mainly in the home or the workplace. In the home, risks are associated with domestic kitchens, including stoves, flames, and hot liquids. In the workplace, risks are associated with fire and chemical and electric burns. Alcoholism and smoking are other risk factors. Burns can also occur as a result of self-harm or violence between people (assault). Burns that affect only the superficial skin layers are known as superficial or first-degree burns. They appear red without blisters, and pain typically lasts around three days. When the injury extends into some of the underlying skin layer, it is a partial-thickness or second-degree burn. Blisters are frequently present and they are often very painful. Healing can re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hypotonic
In chemical biology, tonicity is a measure of the effective osmotic pressure gradient; the water potential of two solutions separated by a partially-permeable cell membrane. Tonicity depends on the relative concentration of selective membrane-impermeable solutes across a cell membrane which determine the direction and extent of osmotic flux. It is commonly used when describing the swelling-versus-shrinking response of cells immersed in an external solution. Unlike osmotic pressure, tonicity is influenced only by solutes that cannot cross the membrane, as only these exert an effective osmotic pressure. Solutes able to freely cross the membrane do not affect tonicity because they will always equilibrate with equal concentrations on both sides of the membrane without net solvent movement. It is also a factor affecting imbibition. There are three classifications of tonicity that one solution can have relative to another: ''hypertonic'', ''hypotonic'', and ''isotonic''. A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
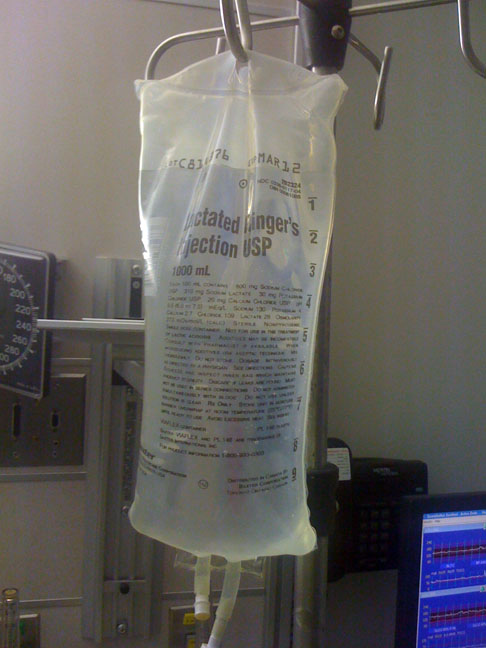
Ringer's Lactate
Ringer's lactate solution (RL), also known as sodium lactate solution, Lactated Ringer's (LR), and Hartmann's solution, is a mixture of sodium chloride, sodium lactate, potassium chloride, and calcium chloride in water. It is used for replacing fluids and electrolytes in those who have low blood volume or low blood pressure. It may also be used to treat metabolic acidosis and to wash the eye following a chemical burn. It is given by intravenous infusion or applied to the affected area. Side effects may include allergic reactions, high blood potassium, hypervolemia, and high blood calcium. It may not be suitable for mixing with certain medications and some recommend against use in the same infusion as a blood transfusion. Ringer's lactate solution has a lower rate of acidosis as compared with normal saline. Use is generally safe in pregnancy and breastfeeding. Ringer's lactate solution is in the Volume expander#Crystalloids, crystalloid family of medications. It i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |