|
1,4-Cyclohexanedimethanol Diglycidyl Ether
1,4-Cyclohexanedimethanol diglycidyl ether is an organic chemical in the glycidyl ether family. Its formula is C14H24O4 and the IUPAC name is 2-4-(oxiran-2-ylmethoxymethyl)cyclohexylmethoxymethyloxirane. It has the CAS number of 14228-73-0 and is REACH registered in Europe. An industrial chemical, a key use is in the reduction of the viscosity of epoxy resin systems functioning as a reactive diluent. Synonyms The material is known under various names which include: * 2,2'- ,4-Cyclohexanediylbis(methyleneoxymethylene)is xirane* 1,4-Bis(glycidoxymethyl)cyclohexane * 1,4-Bis(hydroxymethyl)cyclohexane diglycidyl Ether * 1,4-Bis 2,3-epoxypropoxy)methylyclohexane * 1,4-Bis glycidyloxy)methylyclohexane * 1,4-Cyclohexanedimethanol diglycidyl Ether Manufacture The manufacturing process involves reacting cyclohexanedimethanol with epichlorohydrin, using a Lewis acid as catalyst, to form a halohydrin. This is followed by washing with sodium hydroxide in a dehydrochlorination step to f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
International Union Of Pure And Applied Chemistry
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is a member of the International Science Council (ISC). IUPAC is registered in Zürich, Switzerland, and the administrative office, known as the "IUPAC Secretariat", is in Research Triangle Park, North Carolina, United States. IUPAC's executive director heads this administrative office, currently Greta Heydenrych. IUPAC was established in 1919 as the successor of the International Congress of Applied Chemistry for the advancement of chemistry. Its members, the National Adhering Organizations, can be national chemistry societies, national academies of sciences, or other bodies representing chemists. There are fifty-four National Adhering Organizations and three Associate National Adhering Organizations. IUPAC's Inter-divisional Committee ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Hydroxide
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula . It is a white solid ionic compound consisting of sodium cations and hydroxide anions . Sodium hydroxide is a highly corrosive base (chemistry), base and alkali that decomposes lipids and proteins at ambient temperatures and at high concentrations may cause severe chemical burns. It is highly soluble in water, and readily absorbs moisture and carbon dioxide from the air. It forms a series of hydrates . The monohydrate crystallizes from water solutions between 12.3 and 61.8 °C. The commercially available "sodium hydroxide" is often this monohydrate, and published data may refer to it instead of the anhydrous compound. As one of the simplest hydroxides, sodium hydroxide is frequently used alongside neutral water and acidic hydrochloric acid to demonstrate the pH scale to chemistry students. Sodium hydroxide is used in many industries: in the making of wood pulp and paper, tex ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycidol
Glycidol is an organic compound with the formula . The molecule contains both epoxide and alcohol functional groups. Being simple to make and bifunctional, it has a variety of industrial uses. The compound is a colorless, slightly viscous liquid that is slightly unstable and is not often encountered in pure form. Synthesis and applications Glycidol is prepared by the epoxidation of allyl alcohol. A typical catalyst is tungstic acid, and a typical O-atom source is aqueous peroxyacetic acid.Guenter Sienel, Robert Rieth, Kenneth T. Rowbottom "Epoxides" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005. Some useful products derived from glycidol are 2,3-epoxypropyloxy chloroformate (from phosgene) and glycidyl urethanes (by addition of isocyanates): : : Glycidol is used as a chemical intermediate in the synthesis of other glycidyl ethers, esters, and amines. Glycidol can be O-benzylated in the presence of strong base. More typically, such glycidol e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elastomer
An elastomer is a polymer with viscoelasticity (i.e. both viscosity and elasticity) and with weak intermolecular forces, generally low Young's modulus (E) and high failure strain compared with other materials. The term, a portmanteau of ''elastic polymer'', is often used interchangeably with ''rubber'', although the latter is preferred when referring to vulcanisates. Each of the monomers which link to form the polymer is usually a compound of several elements among carbon, hydrogen, oxygen and silicon. Elastomers are amorphous polymers maintained above their glass transition temperature, so that considerable molecular reconformation is feasible without breaking of covalent bonds. Rubber-like solids with elastic properties are called elastomers. Polymer chains are held together in these materials by relatively weak intermolecular bonds, which permit the polymers to stretch in response to macroscopic stresses. Elastomers are usually thermosets (requiring vulcanization ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
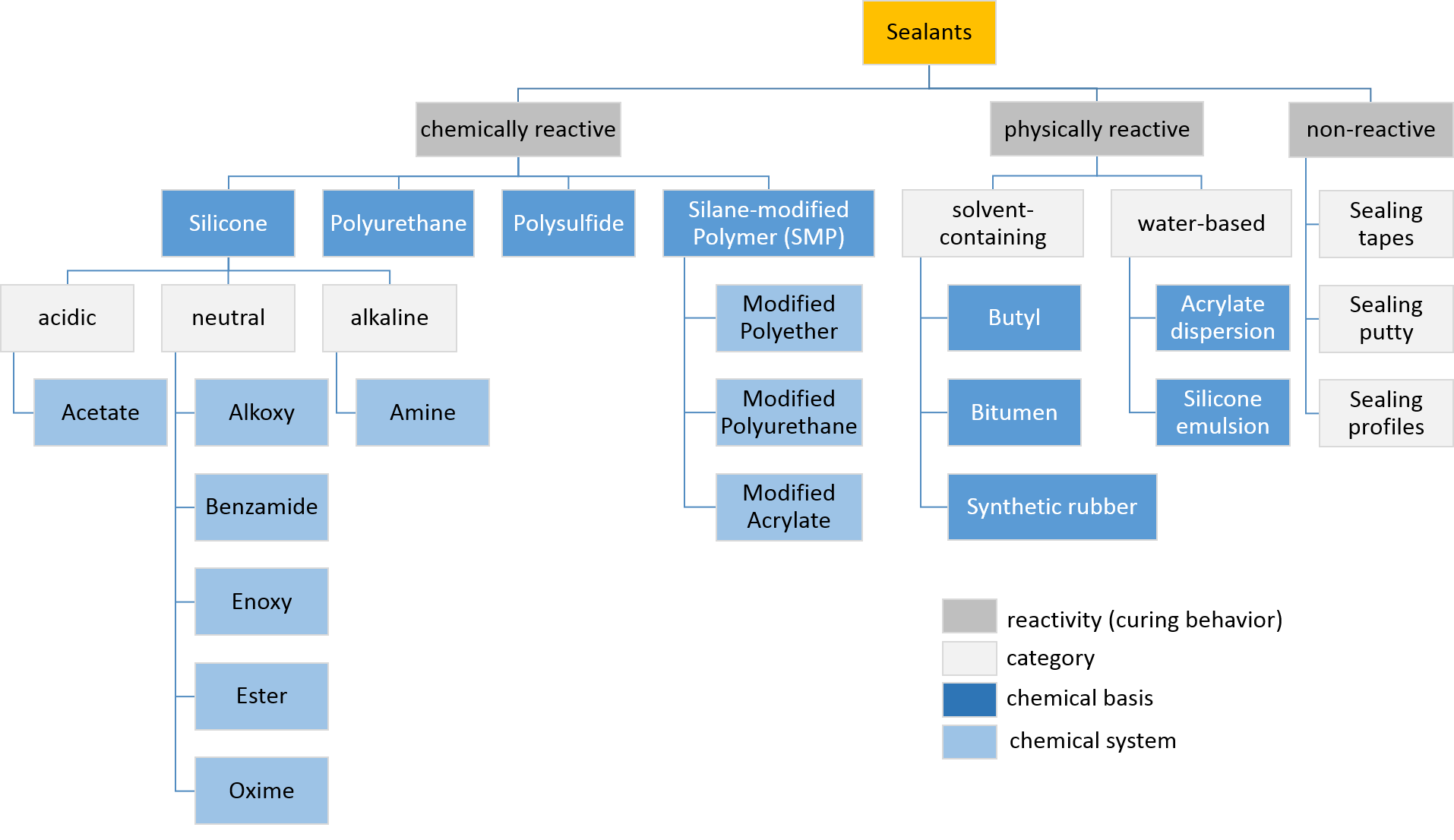
Sealant
Sealant is a substance used to block the passage of fluids through openings in materials, a type of Seal (mechanical), mechanical seal. In building construction ''sealant'' is sometimes synonymous with ''caulk'' (especially if acrylic latex or polyurethane based) and also serve the purposes of blocking dust, sound and heat transmission. Sealants may be weak or strong, flexible or rigid, permanent or temporary. Sealants are not adhesives but some have adhesive qualities and are called ''adhesive-sealants'' or ''structural sealants''. History Sealants were first used in prehistory in the broadest sense as mud, grass and reeds to seal dwellings from the weather such as the daub in wattle and daub and thatching. Natural sealants and adhesive-sealants included plant resins such as pine pitch and birch pitch, bitumen, wax, tar, natural gum, clay (mud) mortar, lime mortar, lead, blood and egg. In the 17th century glazing putty was first used to seal window glass made with linseed oil a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adhesive
Adhesive, also known as glue, cement, mucilage, or paste, is any non-metallic substance applied to one or both surfaces of two separate items that binds them together and resists their separation. The use of adhesives offers certain advantages over other binding techniques such as sewing, mechanical fastenings, and welding. These include the ability to bind different materials together, the more efficient distribution of stress across a joint, the cost-effectiveness of an easily mechanized process, and greater flexibility in design. Disadvantages of adhesive use include decreased stability at high temperatures, relative weakness in bonding large objects with a small bonding surface area, and greater difficulty in separating objects during testing. Adhesives are typically organized by the method of adhesion followed by ''reactive'' or ''non-reactive'', a term which refers to whether the adhesive chemically reacts in order to harden. Alternatively, they can be organized either ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coating
A coating is a covering that is applied to the surface of an object, or substrate. The purpose of applying the coating may be decorative, functional, or both. Coatings may be applied as liquids, gases or solids e.g. powder coatings. Paints and lacquers are coatings that mostly have dual uses, which are protecting the substrate and being decorative, although some artists paints are only for decoration, and the paint on large industrial pipes is for identification (e.g. blue for process water, red for fire-fighting control) in addition to preventing corrosion. Along with corrosion resistance, functional coatings may also be applied to change the surface properties of the substrate, such as adhesion, wettability, or wear resistance.Howarth G.A "Synthesis of a legislation compliant corrosion protection coating system based on urethane, oxazolidine and waterborne epoxy technology" Master of Science Thesis April 1997 Imperial College London In other cases the coating adds a co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epoxy Value
Epoxy value derives from the Epoxy equivalent weight (EEW) or Weight Per Epoxide (WPE) and is a measure of the epoxy content of an epoxy resin or epoxy reactive diluent, or glycidyl ether. This is an important parameter as it allows determination of the correct mix ratio of an epoxy system with a curing agent. The epoxide equivalent weight is usually measured first and done by titration. The standard test method is ASTM D1652 though this has been modified by certain states of the USA. The epoxy equivalent weight (EEW) maybe defined as: the number of grams of epoxy resin required to give 1 mole of epoxy groups. The epoxy value is defined as the number of moles of epoxy group per 100g resin. Example calculations Polyoxypropylene diglycidyl ether, a reactive diluent with the trade name of Diluent F, has an average molecular weight of 1000 and a functionality of 2 so the EEW is 1000/2 = 500. The epoxy value is defined as the number of moles of epoxy group per 100g resin. So as an e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Epoxide
In organic chemistry, an epoxide is a cyclic ether, where the ether forms a three-atom ring: two atoms of carbon and one atom of oxygen. This triangular structure has substantial ring strain, making epoxides highly reactive, more so than other ethers. They are produced on a large scale for many applications. In general, low molecular weight epoxides are colourless and nonpolar, and often volatile. Nomenclature A compound containing the epoxide functional group can be called an epoxy, epoxide, oxirane, and ethoxyline. Simple epoxides are often referred to as oxides. Thus, the epoxide of ethylene (C2H4) is ethylene oxide (C2H4O). Many compounds have trivial names; for instance, ethylene oxide is called "oxirane". Some names emphasize the presence of the epoxide functional group, as in the compound ''1,2-epoxyheptane'', which can also be called ''1,2-heptene oxide''. A polymer formed from epoxide precursors is called an ''epoxy''. However, few if any of the epoxy groups i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dehydrochlorination
In chemistry, dehydrohalogenation is an elimination reaction which removes a hydrogen halide from a substrate. The reaction is usually associated with the synthesis of alkenes, but it has wider applications. Dehydrohalogenation from alkyl halides Traditionally, alkyl halides are substrates for dehydrohalogenations. The alkyl halide must be able to form an alkene, thus halides having no C–H bond on an adjacent carbon are not suitable substrates. Aryl halides are also unsuitable. Upon treatment with strong base, chlorobenzene dehydrohalogenates to give phenol via a benzyne intermediate. Base-promoted reactions to alkenes When treated with a strong base many alkyl chlorides convert to corresponding alkene. It is also called a β-elimination reaction and is a type of elimination reaction. Some prototypes are shown below: :\begin \ce\ &\ce \\ \ce\ &\ce \\ \ce\ &\ce \end Here ethyl chloride reacts with potassium hydroxide, typically in a solvent such as ethanol, giving ethylene. Lik ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halohydrin
In organic chemistry a halohydrin (also a haloalcohol or β-halo alcohol) is a functional group in which a halogen and a hydroxyl are bonded to adjacent carbon atoms, which otherwise bear only hydrogen or hydrocarbyl groups (e.g. 2-chloroethanol, 3-chloropropane-1,2-diol). The term only applies to saturated motifs, as such compounds like 2-chlorophenol would not normally be considered halohydrins. Megatons of some chlorohydrins, e.g. propylene chlorohydrin, are produced annually as precursors to polymers. Halohydrins may be categorized as chlorohydrins, bromohydrins, fluorohydrins or iodohydrins depending on the halogen present. Synthesis From alkenes Halohydrins are usually prepared by treatment of an alkene with a halogen, in the presence of water. The reaction is a form of electrophilic addition, with the halogen acting as electrophile. In that regard, it resembles the halogen addition reaction and proceeds with anti addition, leaving the newly added X and OH groups in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |